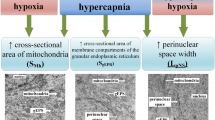
Abstract
Because of their high energetic demand, neurons within the mammalian central nervous system are extremely sensitive to changes in partial pressure of oxygen. Faced with acute hypoxic conditions, an organism must follow a complex and highly dynamic emergency plan to secure survival. Behavioral functions that are not immediately essential for survival are turned off, and critical behaviors (such as breathing) undergo a biphasic response. An augmentation of breathing is initially adaptive, whereas prolonged hypoxic conditions are better served by an energy-saving mode. However, the hypoxic response of an organism depends on many additional factors. Environmental conditions, the animal’s age and health, and the pattern (continuous vs intermittent) and duration (chronic vs acute) of hypoxia greatly determine the specific course of a hypoxic response. Different forms of hypoxia can cause pathology or be used as therapy. Therefore, it is not surprising that the hypoxic response of an organism results from widespread and highly diverse reconfigurations of neuronal network functions in different brain areas that are accomplished by diverse hypoxic changes at all levels of the nervous system (i.e., molecular, cellular, synaptic, neuronal, network). Hypoxia-induced changes in synaptic transmission are generally depressive and result primarily from presynaptic mechanisms, whereas changes in intrinsic properties involve excitatory and inhibitory alterations involving the majority of K+, Na+, and Ca2+ channels. This article reviews the response of the nervous system to hypoxia, accounting for all levels of integration from the cellular to the network level, and postulates that a better understanding of the diversity of cellular events is only possible if cellular and network events are considered in a functional and organismal context.
Similar content being viewed by others
References
Gnaiger E. (2001) Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. Respir. Physiol. 128, 277–297.
Gonzalez C., Almaraz L., Obeso A., and Rigual R. (1994) Carotid body chemoreceptors: from natural stimuli to sensory discharges. Physiol. Rev. 74, 829–898.
Lopez-Barneo J., Pardal R., and Ortega-Saenz P. (2001) Cellular mechanism of oxygen sensing. Annu. Rev. Physiol. 63, 259–287.
Peers C. (1997) Oxygen-sensitive ion channels. Trends Pharmacol. Sci. 18, 405–408.
Prabhakar N. R. (2000) Oxygen sensing by the carotid body chemoreceptors. J. Appl. Physiol. 88, 2287–2295.
Fu X. W., Wang D., Nurse C. A., Dinauer M. C., and Cutz E. (2000) NADPH oxidase is an O2 sensor in airway chemoreceptors: evidence from K+ current modulation in wild-type and oxidase-deficient mice. Proc. Natl. Acad. Sci. USA 97, 4374–4379.
Youngson C., Nurse C., Yeger H., and Cutz E. (1993) Oxygen sensing in airway chemoreceptors. Nature. 365, 153–155.
Thompson R. J. and Nurse C. A. (1998) Anoxia differentially modulates multiple K+ currents and depolarizes neonatal rat adrenal chromaffin cells. J. Physiol. 512, 421–434.
Archer S. L., Huang J., Henry T., Peterson D., and Weir E. K. (1993) A redox-based O2 sensor in rat pulmonary vasculature. Circ. Res. 73, 1100–1112.
Archer S. L., Souil E., Dinh-Xuan A. T., et al. (1998) Molecular identification of the role of voltage-gated K+ channels, Kv1.5 and Kv2.1, in hypoxic pulmonary vasoconstriction and control of resting membrane potential in rat pulmonary artery myocytes. J. Clin. Invest. 101, 2319–2330.
Haddad G. G. and Jiang C. (1993) O2 deprivation in the central nervous system: on mechanisms of neuronal response, differential sensitivity and injury. Prog. Neurobiol. 40, 277–318.
Haddad G. G. and Jiang C. (1997) O2-sensing mechanisms in excitable cells: role of plasma membrane K+ channels. Annu. Rev. Physiol. 59, 23–42.
Henrich M., Hoffmann K., Konig P., et al. (2002) Sensory neurons respond to hypoxia with NO production associated with mitochondria. Mol. Cell. Neurosci. 20, 307–322.
Peña F., Parkis M. A., Tryba A. K., and Ramirez J. M. (2004) Differential contribution of pacemaker properties to the generation of respiratory rhythms during normoxia and hypoxia. Neuron 43, 105–117.
Plant L. D., Kemp P. J., Peers C., Henderson Z., and Pearson H. A. (2002) Hypoxic depolarization of cerebellar granule neurons by specific inhibition of TASK-1. Stroke 33, 2324–2328.
Jiang C. and Haddad G. G. (1994) A direct mechanism for sensing low oxygen levels by central neurons. Proc. Natl. Acad. Sci. USA 91, 7198–7201.
Hammarstrom A. K. and Gage P. W. (2000) Oxygen-sensing persistent sodium channels in rat hippocampus. J. Physiol. 529, 107–118.
Lutz P. L. and Nilsson G. E. (1997) Contrasting strategies for anoxic brain survival—glycolysis up or down. J. Exp. Biol. 200, 411–419.
Bickler P. E. and Buck L. T. (1998) Adaptations of vertebrate neurons to hypoxia and anoxia: maintaining critical Ca2+ concentrations. J. Exp. Biol. 201, 1141–1152.
Bickler P. E., Donohoe P. H., and Buck L. T. (2002) Molecular adaptations for survival during anoxia: lessons from lower vertebrates. Neuroscientist 8, 234–242.
Hochachka P. W. and Lutz P. L. (2001) Mechanism, origin, and evolution of anoxia tolerance in animals. Comp. Biochem. Physiol. B. Biochem. Mol. Biol. 130, 435–459.
Hochachka P. W. (1986) Balancing conflicting metabolic demands of exercise and diving. Fed. Proc. 45, 2948–2952.
Hopfl G., Ogunshola O., and Gassmann M. (2003) Hypoxia and high altitude. The molecular response. Adv. Exp. Med. Biol. 543, 89–115.
Hourdez S. and Weber R. E. (2005) Molecular and functional adaptations in deep-sea hemoglobins. J. Inorg. Biochem. 99, 130–141.
Leblond J. and Krnjevic K. (1989) Hypoxic changes in hippocampal neurons. J. Neurophysiol. 62, 1–14.
Bachevalier J. and Meunier M. (1989) Cerebral ischemia: are the memory deficits associated with hippocampal cell loss? Hippocampus. 6, 553–560.
Hara H., Sukamoto T., and Kogure K. (1993) Mechanism and pathogenesis of ischemia-induced neuronal damage. Prog. Neurobiol. 40, 645–670.
Ramirez J. M., Tryba A. K., and Pena F. (2004) Pacemaker neurons and neuronal networks: an integrative view. Curr. Opin. Neurobiol. 14, 665–674.
Lipton P. and Whittingham T. S. (1979) The effect of hypoxia on evoked potentials in the in vitro hippocampus. J. Physiol. 287, 427–438.
Luhmann H. J. and Heinemann U. (1992) Hypoxia-induced functional alterations in adult rat neocortex. J Neurophysiol. 67, 798–811.
Cummins T. R., Agulian S. K., and Haddad G. G. (1993) Oxygen tension clamp around single neurons in vitro: a computerized method for studies on O2 deprivation. J. Neurosci. Methods 46, 183–189.
Fowler J. C., Gervitz L. M., Hamilton M. E., and Walker J. A. (2003) Systemic hypoxia and the depression of synaptic transmission in rat hippocampus after carotid artery occlusion. J. Physiol. 550, 961–972.
Gervitz L. M., Lutherer L. O., Davies D. G., Pirch J. H., and Fowler J. C. (2001) Adenosine induces initial hypoxic-ischemic depression of synaptic transmission in the rat hippocampus in vivo. Am. J. Physiol. Regul. Integr. Comp. Physiol. 280, R639-R645.
Gervitz L. M., Davies D. G., Omidvar K., and Fowler J. C. (2003) The effect of acute hypoxemia and hypotension on adenosine-mediated depression of evoked hippocampal synaptic transmission. Exp. Neurol. 182, 507–517.
Fujiwara N., Higashi H., Shimoji K., and Yoshimura M. (1987) Effects of hypoxia on rat hippocampal neurones in vitro. J. Physiol. 384, 131–151.
Jiang C. and Haddad G. G. (1992) Differential responses of neocortical neurons to glucose and/or O2 deprivation in the human and rat. J. Neurophysiol. 68, 2165–2173.
Katchman A. N. and Hershkowitz N. (1993) Adenosine antagonists prevent hypoxia-induced depression of excitatory but not inhibitory synaptic currents. Neurosci Lett. 159, 123–126.
Katchman A. N. and Hershkowitz N. (1993) Early anoxia-induced vesicular glutamate release results from mobilization of calcium from intracellular stores. J. Neurophysiol. 70, 1–7.
Fujimura N., Tanaka E., Yamamoto S., Shigemori M., and Higashi H. (1997) Contribution of ATP-sensitive potassium channels to hypoxic hyperpolarization in rat hippocampal CA1 neurons in vitro. J. Neurophysiol. 77, 378–385.
Erdemli G. and Crunelli V. (1998) Response of thalamocortical neurons to hypoxia: a whole-cell patch-clamp study. J Neurosci. 18, 5212–5224.
Hammarstrom A. K. and Gage P. W. (1998) Inhibition of oxidative metabolism increases persistent sodium current in rat CA1 hippocampal neurons. J Physiol. 510, 735–741.
Mironov S. L. and Richter D. W. (1999) Cytoskeleton mediates inhibition of the fast Na+ current in respiratory brainstem neurons during hypoxia. Eur J Neurosci. 11, 1831–1834.
Kulik A., Trapp S., and Ballanyi K. (2000) Ischemia but not anoxia evokes vesicular and Ca(2+)-independent glutamate release in the dorsal vagal complex in vitro. J. Neurophysiol. 83, 2905–2915.
Hida W., Tun Y., Kikuchi Y., Okabe S., and Shirato K. (2002) Pulmonary hypertension in patients with chronic obstructive pulmonary disease: recent advances in pathophysiology and management. Respirology 7, 3–13.
Naughton M. T. and Bradley T. D. (1998) Sleep apnea in congestive heart failure. Clin. Chest Med. 19, 99–113.
Lanfranchi P. and Somers V. K. (2001) Obstructive sleep apnea and vascular disease. Respir. Res. 2, 315–319.
Rossen R., Kabat H., and Anderson J. P. (1943) Acute arrest of cerebral circulation in man. Arch. Neurol. Psych. 50, 510–528.
Lipton P. (1999) Ischemic cell death in brain neurons. Physiol. Rev. 79, 1431–1568.
Hansen A. J., Hounsgaard J., and Jahnsen H. (1982) Anoxia increases potassium conductance in hippocampal nerve cells. Acta. Physiol. Scand. 115, 301–310.
Krnjevic K. and Leblond J. (1989) Changes in membrane currents of hippocampal neurons evoked by brief anoxia. J Neurophysiol. 62, 15–30.
Young J. N. and Somjen G. G. (1992) Suppression of presynaptic calcium currents by hypoxia in hippocampal tissue slices. Brain Res. 21, 70–76.
Neubauer J. A. and Sunderram J. (2004) Oxygen-sensing neurons in the central nervous system. J. Appl. Physiol. 96, 367–374.
Duffy T. E., Kohle S. J., and Vannucci R. C. (1975) Carbohydrate and energy metabolism in perinatal rat brain: relation to survival in anoxia. J. Neurochem. 24, 271–276.
Ferriero D. M., Arcavi L. J., Sagar S. M., McIntosh T. K., and Simon R. P. (1988) Selective sparing of NADPH-diaphorase neurons in neonatal hypoxia-ischemia. Ann. Neurol. 24, 670–676.
Cherubini E., Ben-Ari Y., and Krnjevic K. (1989) Anoxia produces smaller changes in synaptic transmission, membrane potential, and input resistance in immature rat hippocampus. J. Neurophysiol. 62, 882–895.
Haddad G. G. and Donnelly D. F. (1990) O2 deprivation induces a major depolarization in brain stem neurons in the adult but not in the neonatal rat. J. Physiol. 429, 411–428.
Jiang C., Agulian S., and Haddad G. G. (1991) O2 tension in adult and neonatal brain slices under several experimental conditions. Brain Res. 568, 159–164.
Singer D. (1999) Neonatal tolerance to hypoxia: a comparative-physiological approach. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 123, 221–234.
Hicks S. P. (1953) Developmental brain metabolism; effects of cortisone, anoxia, fluoroacetate, radiation, insulin, and other inhibitors on the embryo, newborn, and adult. AMA Arch. Pathol. 55, 302–327.
Ballanyi K., Kuwana S., Volker A., Morawietz G., and Richter D. W. (1992) Developmental changes in the hypoxia tolerance of the in vitro respiratory network of rats. Neurosci. Lett. 148, 141–144.
Choi D. W. and Rothman S. M. (1990) The role of glutamate neurotoxicity in hypoxicischemic neuronal death. Annu. Rev. Neurosci. 13, 171–182.
Friedman J. E. and Haddad G. G. (1993) Major differences in Ca2+i response to anoxia between neonatal and adult rat CA1 neurons: role of Ca2+o and Na+o. J. Neurosci. 13, 63–72.
Kawai S., Yonetani M., Nakamura H., and Okada Y. (1989) Effects of deprivation of oxygen and glucose on the neural activity and the level of high energy phosphates in the hippocampal slices of immature and adult rat. Brain Res. Dev. Brain Res. 48, 11–18.
Bickler P. E., Gallego S. M., and Hansen B. M. (1993) Developmental changes in intracellular calcium regulation in rat cerebral cortex during hypoxia. J. Cereb. Blood. Flow. Metab. 13, 811–819.
Luhmann H. J., Kral T., and Heinemann U. (1993) Influence of hypoxia on excitation and GABAergic inhibition in mature and developing rat neocortex. Exp. Brain Res. 97, 209–224.
Hansen A. J. (1985). Effect of anoxia on ion distribution in the brain. Physiol. Rev. 65, 101–148
Ben-Ari Y. (1992) Effects of anoxia and aglycemia on the adult and immature hippocampus. Biol. Neonate. 62, 225–230.
Kasischke K. A., Vishwasrao H. D., Fisher P. J., Zipfel W. R., and Webb W. W. (2004) Neural activity triggers neuronal oxidative metabolism followed by astrocytic glycolysis. Science 305, 99–103.
Booth R. F., Patel T. B., and Clark J. B. (1980) The development of enzymes of energy metabolism in the brain of a precocial (guinea pig) and non-precocial (rat) species. J. Neurochem. 34, 17–25.
Kreisman N. R., Sick T. J., LaManna J. C., and Rosenthal M. (1981) Local tissue oxygen tension-cytochrome a,a3 redox relationships in rat cerebral cortex in vivo. Brain Res. 218, 161–174.
Erecinska M. and Silver I. A. (1994) Ions and energy in mammalian brain. Prog. Neurobiol. 43, 37–71.
Hong S. S., Gibney G. T., Esquilin M., Yu J., and Xia Y. (2004) Effect of protein kinases on lactate dehydrogenase activity in cortical neurons during hypoxia. Brain Res. 1009, 195–202.
Paschen W. and Djuricic B. (1995) Comparison of in vitro ischemia-induced disturbances in energy metabolism and protein synthesis in the hippocampus of rats and gerbils. J. Neurochem. 65, 1692–1697.
Milusheva E. A., Doda M., Baranyi M., and Vizi E. S. (1996) Effect of hypoxia and glucose deprivation on ATP level, adenylate energy charge and [Ca2+]o-dependent and independent release of [3H]dopamine in rat striatal slices. Neurochem. Int. 28, 501–507.
Marrif H. and Juurlink B. H. (1999) Astrocytes respond to hypoxia by increasing glycolytic capacity. J. Neurosci. Res. 57, 255–260.
Ojala B. E., Page L. A., Moore M. A., and Thompson L. V. (2001) Effects of inactivity on glycolytic capacity of single skeletal muscle fibers in adult and aged rats. Biol. Res. Nurs. 3, 88–95.
Dang C. V. and Semenza G. L. (1999) Oncogenic alterations of metabolism. Trends Biochem. Sci. 24, 68–72.
Robin E. D., Murphy B. J., and Theodore J. (1984) Coordinate regulation of glycolysis by hypoxia in mammalian cells. J. Cell. Physiol. 118, 287–290.
Tian G. F. and Baker A. J. (2000) Glycolysis prevents anoxia-induced synaptic transmission damage in rat hippocampal slices. J. Neurophysiol. 83, 1830–1839.
Schurr A., West C. A., Reid K. H., Tseng M. T., Reiss S. J., and Rigor B. M. (1987) Increased glucose improves recovery of neuronal function after cerebral hypoxia in vitro. Brain Res. 421, 135–139.
Callahan D. J., Engle M. J., and Volpe J. J. (1990) Hypoxic injury to developing glial cells: protective effect of high glucose. Pediatr. Res. 27, 186–190.
Schurr A., West C. A., and Rigor B. M. (1988) Lactate-supported synaptic function in the rat hippocampal slice preparation. Science 240, 1326–1328.
Ballanyi K., Volker A., and Richter D. W. (1996) Functional relevance of anaerobic metabolism in the isolated respiratory network of newborn rats. Pflugers Arch. 432, 741–748.
LaManna J. C., Haxhiu M. A., Kutina-Nelson K. L., et al. (1996) Decreased energy metabolism in brain stem during central respiratory depression in response to hypoxia. J. Appl. Physiol. 81, 1772–1777.
Siesjo B. K., Katsura K., and Kristian T. (1996) Acidosis-related damage. Adv. Neurol. 71, 209–233.
Fujiwara N., Abe T., Endoh H., Warashina A., and Shimoji K. (1992) Changes in intracellular pH of mouse hippocampal slices responding to hypoxia and/or glucose depletion. Brain Res. 572, 335–339.
Pulsinelli W. A., Brierley J. B., and Plum F. (1982) Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann. Neurol. 11, 491–498.
Giffard R. G., Monyer H., Christine C. W., and Choi D. W. (1990) Acidosis reduces NMDA receptor activation, glutamate neurotoxicity, and oxygen-glucose deprivation neuronal injury in cortical cultures. Brain Res. 506, 339–342.
Tombaugh G. C. and Sapolsky R. M. (1990) Mild acidosis protects hippocampal neurons from injury induced by oxygen and glucose deprivation. Brain Res. 506, 343–345.
Krnjevic K. (1993) Central cholinergic mechanisms and function. Prog. Brain Res. 98, 285–292.
Martin L. J., Brambrink A. M., Lehmann C., et al. (1997) Hypoxia-ischemia causes abnormalities in glutamate transporters and death of astroglia and neurons in newborn striatum. Ann. Neurol. 42, 335–348.
Crepel V., Krnjevic K., and Ben-Ari Y. (1992) Developmental and regional differences in the vulnerability of rat hippocampal slices to lack of glucose. Neuroscience 47, 579–587.
Ballanyi K., Doutheil J., and Brockhaus J. (1996) Membrane potentials and microenvironment of rat dorsal vagal cells in vitro during energy depletion. J. Physiol. 495, 769–784.
Ballanyi K., Kuwana S., Volker A., Morawietz G., and Richter D. W. (1992) Developmental changes in the hypoxia tolerance of the in vitro respiratory network of rats. Neurosci. Lett. 148, 141–144.
Donnelly D. F., Jiang C., and Haddad G. G. (1992) Comparative responses of brain stem and hippocampal neurons to O2 deprivation: in vitro intracellular studies. Am. J. Physiol. 262, L549-L554.
O’Reilly J. P., Jiang C., and Haddad G. G. (1995) Major differences in response to graded hypoxia between hypoglossal and neocortical neurons. Brain Res. 683, 179–186.
Cowan A. I. and Martin R. L. (1992) Ionic basis of membrane potential changes induced by anoxia in rat dorsal vagal motoneurones. J. Physiol. 455, 89–109.
Nolan P. C. and Waldrop T. G. (1996) Ventrolateral medullary neurons show age-dependent depolarizations to hypoxia in vitro. Brain Res. Dev. Brain Res. 91, 111–120.
Thoby-Brisson M. and Ramirez J. M. (2000) Role of inspiratory pacemaker neurons in mediating the hypoxic response of the respiratory network in vitro. J. Neurosci. 20, 5858–5866.
Cummins T. R., Donnelly D. F., and Haddad G. G. (1991) Effect of metabolic inhibition on the excitability of isolated hippocampal CA1 neurons: developmental aspects. J. Neurophysiol. 66, 1471–1482.
Belousov A. B., Godfraind J. M., and Krnjevic K. (1995) Internal Ca2+ stores involved in anoxic responses of rat hippocampal neurons. J. Physiol. 486, 547–556.
Hyllienmark L. and Brismar T. (1996) Effect of metabolic inhibition on K+ channels in pyramidal cells of the hippocampal CA1 region in rat brain slices. J Physiol. 496 (Pt 1), 155–164.
Hyllienmark L. and Brismar T. (1999) Effect of hypoxia on membrane potential and resting conductance in rat hippocampal neurons. Neuroscience 91, 511–517.
Erdemli G., Xu Y. Z., and Krnjevic K. (1998) Potassium conductance causing hyperpolarization of CA1 hippocampal neurons during hypoxia. J. Neurophysiol. 80, 2378–2390.
Misgeld U. and Frotscher M. (1982) Dependence of the viability of neurons in hippocampal slices on oxygen supply. Brain Res. Bull. 8, 95–100.
Reiner P. B., Laycock A. G., and Doll C. J. (1990) A pharmacological model of ischemia in the hippocampal slice. Neurosci. Lett. 119, 175–178.
Zhang L. and Krnjevic K. (1993) Whole-cell recording of anoxic effects on hippocampal neurons in slices. J. Neurophysiol. 69, 118–127.
Fung M. L. and Haddad G. G. (1997) Anoxia-induced depolarization in CA1 hippocampal neurons: role of Na+-dependent mechanisms. Brain Res. 762, 97–102.
Rosen A. S. and Morris M. E. (1993) Anoxic depression of excitatory and inhibitory postsynaptic potentials in rat neocortical slices. J. Neurophysiol. 69, 109–117.
Englund M., Hyllienmark L., and Brismar T. (2001) Chemical hypoxia in hippocampal pyramidal cells affects membrane potential differentially depending on resting potential. Neuroscience 106, 89–94.
Godfraind J. M., Kawamaura H., Krnjevic K., and Pumain R. (1971) Actions of dinitrophenol and some other metabolic inhibitors on cortical neurones. J. Physiol. 215, 199–222.
Yamamoto S., Tanaka E., Shoji Y., Kudo Y., Inokuchi H., and Higashi H. (1997) Factors that reverse the persistent depolarization produced by deprivation of oxygen and glucose in rat hippocampal CA1 neurons in vitro. J. Neurophysiol. 78, 903–911.
Zhu P. J. and Krnjevic K. (1997) Endogenous adenosine on membrane properties of CA1 neurons in rat hippocampal slices during normoxia and hypoxia. Neuropharmacology 36, 169–176.
Pascual O., Morin-Surun M. P., Barna B., Denavit-Saubie M., Pequignot J. M., and Champagnat J. (2002) Progesterone reverses the neuronal responses to hypoxia in rat nucleus tractus solitarius in vitro. J. Physiol. 544, 511–520.
Croning M. D., Zetterstrom T. S., Grahame-Smith D. G., and Newberry N. R. (1995) Action of adenosine receptor antagonists on hypoxia-induced effects in the rat hippocampus in vitro. Br. J. Pharmacol. 116, 2113–2119.
Yang J. J., Chou Y. C., Lin M. T., and Chiu T. H. (1997) Hypoxia-induced differential electrophysiological changes in rat locus coeruleus neurons. Life Sci. 61, 1763–1773.
Wang G., Zhou P., Repucci M. A., Golanov E. V., and Reis D. J. (2001) Specific actions of cyanide on membrane potential and voltage-gated ion currents in rostral ventrolateral medulla neurons in rat brainstem slices. Neurosci. Lett. 309, 125–129.
Mercuri N. B., Bonci A., Calabresi P., Stratta F., and Bernardi G. (1994) Responses of rat mesencephalic dopaminergic neurons to a prolonged period of oxygen deprivation. Neuroscience 63, 757–764.
Mercuri N. B., Bonci A., Johnson S. W., Stratta F., Calabresi P., and Bernardi G. (1994) Effects of anoxia on rat midbrain dopamine neurons. J. Neurophysiol. 71, 1165–1173.
Calabresi P., Pisani A., Mercuri N. B, and Bernardi G. (1995) Hypoxia-induced electrical changes in striatal neurons. J. Cereb. Blood Flow Metab. 15, 1141–1145.
Dillon G. H. and Waldrop T. G. (1993) Responses of feline caudal hypothalamic cardiorespiratory neurons to hypoxia and hypercapnia. Exp. Brain Res. 96, 260–272.
Ryan J. W. and Waldrop T. G. (1995) Hypoxia sensitive neurons in the caudal hypothalamus project to the periaqueductal gray. Respir. Physiol. 100, 185–194.
Dillon G. H. and Waldrop T. G. (1992) In vitro responses of caudal hypothalamic neurons to hypoxia and hypercapnia. Neuroscience 51, 941–950.
Horn E. M., Dillon G. H., Fan Y. P., and Waldrop T. G. (1999) Developmental aspects and mechanisms of rat caudal hypothalamic neuronal responses to hypoxia. J. Neurophysiol. 81, 1949–1959.
Watts A. E., Hicks G. A., and Henderson G. (1995) Putative pre- and postsynaptic ATP-sensitive potassium channels in the rat substantia nigra in vitro. J. Neurosci. 15, 3065–3074.
Nieber K., Sevcik J., and Illes P. (1995) Hypoxic changes in rat locus coeruleus neurons in vitro. J. Physiol. 486, 33–46.
Duchen M. R. (1990) Effects of metabolic inhibition on the membrane properties of isolated mouse primary sensory neurones. J. Physiol. 424, 387–409.
Jiang C., Sigworth F. J., and Haddad G. G. (1994) Oxygen deprivation activates an ATP-inhibitable K+ channel in substantia nigra neurons. J. Neurosci. 14, 5590–5602.
Mourre C., Ben Ari Y., Bernardi H., Fosset M., and Lazdunski M. (1989) Antidiabetic sulfony-lureas: localization of binding sites in the brain and effects on the hyperpolarization induced by anoxia in hippocampal slices. Brain Res. 486, 159–164.
Chung I., Zhang Y., Eubanks J. H., and Zhang L. (1998) Attenuation of hypoxic current by intracellular applications of ATP regenerating agents in hippocampal CA1 neurons of rat brain slices. Neuroscience 86, 1101–1107.
Pisani A., Calabresi P., Centonze D., Marfia G. A., and Bernardi G. (1999) Electrophysiological recordings and calcium measurements in striatal large aspiny interneurons in response to combined O2/glucose deprivation. J. Neurophysiol. 81, 2508–2516.
Wan Q., Yao H., and Wang F. (1999) Involvement of K(+) channels in the inhibitory effects of adenosine on anoxia-induced [Ca(2+) ](i) increase in cultured rat hippocampal CA1 neurons. Biol. Signals Recept. 8, 309–315.
Garcia de Arriba S., Franke H., Pissarek M., Nieber K., and Illes P. (1999) Neuroprotection by ATP-dependent potassium channels in rat neocortical brain slices during hypoxia. Neurosci. Lett. 273, 13–16.
Lopez-Barneo J., Lopez-Lopez J. R., Urena J., and Gonzalez C. (1988) Chemotransduction in the carotid body: K+ current modulated by PO2 in type I chemoreceptor cells. Science 241, 580–582.
Lopez-Barneo J. (1994) Oxygen-sensitive ion channels: how ubiquitous are they? Trends Neurosci. 17, 133–135.
Fearon I. M., Palmer A. C., Balmforth A. J., Ball S. G., Varadi G., and Peers C. (1999) Modulation of recombinant human cardiac L-type Ca2+ channel alpha1C subunits by redox agents and hypoxia. J. Physiol. 514, 629–637.
Fleidervish I. A., Gebhardt C., Astman N., Gutnick M. J., and Heinemann U. (2001) Enhanced spontaneous transmitter release is the earliest consequence of neocortical hypoxia that can explain the disruption of normal circuit function. J. Neurosci. 21, 4600–4608.
Zawar C. and Neumcke B. (2000) Differential activation of ATP-sensitive potassium channels during energy depletion in CA1 pyramidal cells and interneurones of rat hippocampus. Pflugers Arch. 439, 256–262.
Shan H. Q. and Cheng J. S. (2000) Effect of adenosine on adenosine triphosphate-sensitive potassium channel during hypoxia in rat hippocampal neurons. Neurosci. Lett. 286, 45–48.
Yamada K., Ji J. J., Yuan H., et al. (2001) Protective role of ATP-sensitive potassium channels in hypoxia-induced generalized seizure. Science. 292, 1543–1546.
Guatteo E., Federici M., Siniscalchi A., Knopfel T., Mercuri N. B., and Bernardi G. (1998) Whole cell patch-clamp recordings of rat midbrain dopaminergic neurons isolate a sulphonylurea- and ATP-sensitive component of potassium currents activated by hypoxia. J. Neurophysiol. 79, 1239–1245.
Kulik A., Brockhaus J., Pedarzani P., and Ballanyi K. (2002) Chemical anoxia activates ATP-sensitive and blocks Ca(2+)-dependent K(+) channels in rat dorsal vagal neurons in situ. Neuroscience 110, 541–554.
Park Y. K., Jung S. J., Yoo J. E., Kwak J., Lim W., and Kim J. (2003) Effect of acute hypoxia on ATP-sensitive potassium currents in substantia gelatinosa neurons of juvenile rats. Pflugers Arch. 446, 600–606.
Mironov S. L., Langohr K., Haller M., and Richter D. W. (1998) Hypoxia activates ATP-dependent potassium channels in inspiratory neurons of neonatal mice. J. Physiol. 509, 755–766.
Mironov S. L. and Richter D. W. (2000) Intracellular signalling pathways modulate K(ATP) channels in inspiratory brainstem neurones and their hypoxic activation: involvement of metabotropic receptors, G-proteins and cytoskeleton. Brain Res. 853, 60–67.
Haller M., Mironov S. L., Karschin A., and Richter D. W. (2001) Dynamic activation of K(ATP) channels in rhythmically active neurons. J. Physiol. 537, 69–81.
Nowicky A. V. and Duchen M. R. (1998) Changes in [Ca2+]i and membrane currents during impaired mitochondrial metabolism in dissociated rat hippocampal neurons. J. Physiol. 507, 131–145.
Liu H., Moczydlowski E., and Haddad G. G. (1999) O(2) deprivation inhibits Ca(2+)-activated K(+) channels via cytosolic factors in mice neocortical neurons. J. Clin. Invest. 104, 577–588.
Buckler K. J. (1997) A novel oxygen-sensitive potassium current in rat carotid body type I cells. J. Physiol. 498, 649–662.
Buckler K. J., Williams B. A., and Honore E. (2000) An oxygen-, acid- and anaesthetic-sensitive TASK-like background potassium channel in rat arterial chemoreceptor cells. J. Physiol. 525, 135–142.
Hartness M. E., Lewis A., Searle G. J., O’Kelly I., Peers C., and Kemp P. J. (2001) Combined antisense and pharmacological approaches implicate hTASK as an airway O(2) sensing K(+) channel. J. Biol. Chem. 276, 26,499–26,508.
Campanucci V. A., Fearon I. M., and Nurse C. A. (2003) A novel O2-sensing mechanism in rat glossopharyngeal neurones mediated by a halothane-inhibitable background K+ conductance. J. Physiol. 548, 731–743.
Crill W. E. (1996) Persistent sodium current in mammalian central neurons. Annu. Rev. Physiol. 58, 349–362.
Ju Y. K., Saint D. A., and Gage P. W. (1996) Hypoxia increases persistent sodium current in rat ventricular myocytes. J. Physiol. 497, 337–347.
Hammarstrom A. K. and Gage P. W. (2002) Hypoxia and persistent sodium current. Eur. Biophys. J. 31, 323–330.
Horn E. M. and Waldrop T. G. (2000) Hypoxic augmentation of fast-inactivating and persistent sodium currents in rat caudal hypothalamic neurons. J. Neurophysiol. 84, 2572–2581.
Cummins T. R., Jiang C., and Haddad G. G. (1993) Human neocortical excitability is decreased during anoxia via sodium channel modulation. J. Clin. Invest. 91, 608–615.
O’Reilly J. P., Cummins T. R., and Haddad G. G. (1997) Oxygen deprivation inhibits Na+ current in rat hippocampal neurones via protein kinase C. J. Physiol. 503, 479–488.
Kawai Y., Qi J., Comer A. M., Gibbons H., Win J., and Lipski J. (1999) Effects of cyanide and hypoxia on membrane currents in neurones acutely dissociated from the rostral ventrolateral medulla of the rat. Brain Res. 830, 246–257.
Mazza E. Jr., Edelman N. H., and Neubauer J. A. (2000) Hypoxic excitation in neurons cultured from the rostral ventrolateral medulla of the neonatal rat. J. Appl. Physiol. 88, 2319–2329.
Lukyanetz E. A., Shkryl V. M., Kravchuk O. V., and Kostyuk P. G. (2003) Action of hypoxia on different types of calcium channels in hippocampal neurons. Biochim. Biophys. Acta. 1618, 33–38.
Lukyanetz E. A., Shkryl V. M., Kravchuk O. V., and Kostyuk P. G. (2003) Effect of hypoxia on calcium channels depends on extracellular calcium in CA1 hippocampal neurons. Brain Res. 980, 128–134.
Sun M. K. and Reis D. J. (1994) Hypoxia-activated Ca2+ currents in pacemaker neurones of rat rostral ventrolateral medulla in vitro. J. Physiol. 476, 101–116.
Mironov S. L. and Richter D. W. (1998) L-type Ca2+ channels in inspiratory neurones of mice and their modulation by hypoxia. J Physiol. 512, 75–87.
Elsen F. P. and Ramirez J. M. (1997) Hypoxia reduces the amplitude of voltage dependent calcium currents in the isolated respiratory system of mice. Soc. Neurosci. Abstr. 27, 495.11
Erdemli G. and Crunelli V. (2000) Release of monoamines and nitric oxide is involved in the modulation of hyperpolarization-activated inward current during acute thalamic hypoxia. Neuroscience 96, 565–574.
Mironov S. L., Langohr K., and Richter D. W. (2000) Hyperpolarization-activated current, Ih, in inspiratory brainstem neurons and its inhibition by hypoxia. Eur. J. Neurosci. 12, 520–526.
Inoue M., Fujishiro N., and Imanaga I. (1999) Na+ pump inhibition and non-selective cation channel activation by cyanide and anoxia in guinea-pig chromaffin cells. J. Physiol. 519, 385–396.
Lees G. J. (1991) Inhibition of sodium-potassium-ATPase: a potentially ubiquitous mechanism contributing to central nervous system neuropathology. Brain Res. Brain Res. Rev. 16, 283–300.
Luhmann H. J. (1996) Ischemia and lesion induced imbalances in cortical function. Prog. Neurobiol. 48, 131–166.
Jha A. and Deshpande S. B. (2003) Aglycemia and ischemia depress spinal synaptic transmission via inhibitory systems involving NMDA receptors. Eur. J. Pharmacol. 481, 189–196.
Deshpande S. B. and Jha A. (2004) Aglycemia and ischemia depress monosynaptic and polysynaptic reflexes in neonatal rat spinal cord in vitro by involving different types of 5-hydroxytryptamine receptors. Neurosci. Lett. 372, 167–172.
Xu Z. C. and Pulsinelli W. A. (1996) Electrophysiological changes of CA1 pyramidal neurons following transient forebrain ischemia: an in vivo intracellular recording and staining study. J. Neurophysiol. 76, 1689–1697.
Hershkowitz N., Katchman A. N., and Veregge S. (1993) Site of synaptic depression during hypoxia: a patch-clamp analysis. J. Neurophysiol. 69, 432–441.
Coelho J. E., de Mendonca A., and Ribeiro J. A. (2000) Presynaptic inhibitory receptors mediate the depression of synaptic transmission upon hypoxia in rat hippocampal slices. Brain Res. 869, 158–165.
Frenguelli B. G., Llaudet E., and Dale N. (2003) High-resolution real-time recording with microelectrode biosensors reveals novel aspects of adenosine release during hypoxia in rat hippocampal slices. J. Neurochem. 86, 1506–1515.
Pearson T. and Frenguelli B. G. (2004) Adrenoceptor subtype-specific acceleration of the hypoxic depression of excitatory synaptic transmission in area CA1 of the rat hippocampus. Eur. J. Neurosci. 20, 1555–1565.
Hentschel S., Lewerenz A., and Nieber K. (2003) Activation of A(3) receptors by endogenous adenosine inhibits synaptic transmission during hypoxia in rat cortical neurons. Restor. Neurol. Neurosci. 21, 55–63.
Centonze D., Saulle E., Pisani A., Bernardi G., and Calabresi P. (2001) Adenosine-mediated inhibition of striatal GABAergic synaptic transmission during in vitro ischaemia. Brain 124, 1855–1865.
Pierrefiche O., Bischoff A. M., Richter D. W., and Spyer K. M. (1997) Hypoxic response of hypoglossal motoneurones in the in vivo cat. J. Physiol. 505, 785–795.
Richter D. W., Bischoff A., Anders K., Bellingham M., and Windhorst U. (1991) Response of the medullary respiratory network of the cat to hypoxia. J. Physiol. 443, 231–256.
Lieske S. P., Thoby-Brisson M., Telgkamp P., and Ramirez J. M. (2000) Reconfiguration of the neural network controlling multiple breathing patterns: eupnea, sighs and gasps. Nat. Neurosci. 3, 600–607.
Hammond C., Crepel V., Gozlan H., and Ben-Ari Y. (1994) Anoxic LTP sheds light on the multiple facets of NMDA receptors. Trends Neurosci. 17, 497–503.
Hellweg R., von Arnim C. A., Buchner M., Huber R., and Riepe M. W. (2003) Neuroprotection and neuronal dysfunction upon repetitive inhibition of oxidative phosphorylation. Exp. Neurol. 183, 346–354.
Rosen A. S. and Morris M. E. (1993) Anoxic depression of excitatory and inhibitory postsynaptic potentials in rat neocortical slices. J. Neurophysiol. 69, 109–117.
Kasa P., Rakonczay Z., and Gulya K. (1997) The cholinergic system in Alzheimer’s disease. Prog. Neurobiol. 52, 511–535.
Porkka-Heiskanen T., Strecker R. E., Thakkar M., Bjorkum A. A., Greene R. W., and McCarley R. W. (1997) Adenosine: a mediator of the sleep-inducing effects of prolonged wakefulness. Science 276, 1265–1268.
Fowler J. C. (1989) Adenosine antagonists delay hypoxia-induced depression of neuronal activity in hippocampal brain slice. Brain Res. 490, 378–384.
Gribkoff V. K., Bauman L. A., and VanderMaelen C. P. (1990) The adenosine antagonist 8-cyclopentyltheophylline reduces the depression of hippocampal neuronal responses during hypoxia. Brain Res. 512, 353–357.
Canhao P., de Mendonca A., and Ribeiro J. A. (1994) 1,3-Dipropyl-8-cyclopentylxanthine attenuates the NMDA response to hypoxia in the rat hippocampus. Brain Res. 661, 265–273.
Dale N., Pearson T., and Frenguelli B. G. (2000) Direct measurement of adenosine release during hypoxia in the CA1 region of the rat hippocampal slice. J. Physiol. 526, 143–155.
Pearson T., Currie A. J., Etherington L. A., et al. (2003) Plasticity of purine release during cerebral ischemia: clinical implications? J. Cell. Mol. Med. 7, 362–375.
Zetterstrom T., Vernet L., Ungerstedt U., Tossman U., Jonzon B., and Fredholm B. B. (1982) Purine levels in the intact rat brain. Studies with an implanted perfused hollow fibre. Neurosci. Lett. 29, 111–115.
Latini S., Bordoni F., Corradetti R., Pepeu G., and Pedata F. (1998) Temporal correlation between adenosine outflow and synaptic potential inhibition in rat hippocampal slices during ischemia-like conditions. Brain Res. 794, 325–328.
de Mendonca A. and Ribeiro J. A. (1997) Contribution of metabotropic glutamate receptors to the depression of excitatory postsynaptic potentials during hypoxia. Neuroreport 8, 3667–3671.
Doherty J. and Dingledine R. (1997) Regulation of excitatory input to inhibitory interneurons of the dentate gyrus during hypoxia. J. Neurophysiol. 77, 393–404.
Cherniack N. S., Edelman N. H., and Lahiri S. (1971) The effect of hypoxia and hypercapnia on respiratory neuron activity and cerebral aerobic metabolism. Chest 59, 29S.
Haddad G. G. and Mellins R. B. (1984) Hypoxia and respiratory control in early life. Annu. Rev. Physiol. 46, 629–643.
Bureau M. A., Zinman R., Foulon P., and Begin R. (1984) Diphasic ventilatory response to hypoxia in newborn lambs. J. Appl. Physiol. 56, 84–90.
St. John W. M. and Bianchi A. L. (1985) Responses of bulbospinal and laryngeal respiratory neurons to hypercapnia and hypoxia. J. Appl. Physiol. 59, 1201–1207.
Fregosi R. F., Knuth S. L., Ward D. K., and Bartlett D. Jr. (1987) Hypoxia inhibits abdominal expiratory nerve activity. J. Appl. Physiol. 63, 211–220.
Maruyama R., Yoshida A., and Fukuda Y. (1989) Differential sensitivity to hypoxic inhibition of respiratory processes in the anesthetized rat. Jpn. J. Physiol. 39, 857–871.
Neubauer J. A., Melton J. E., and Edelman N. H. (1990) Modulation of respiration during brain hypoxia. J. Appl. Physiol. 68, 441–451.
Trippenbach T., Richter D. W., and Acker H. (1990) Hypoxia and ion activities within the brain stem of newborn rabbits. J. Appl. Physiol. 68, 2494–2503.
Kahraman L. and Thach B. T. (2004) Inhibitory effects of hyperthermia on mechanisms involved in autoresuscitation from hypoxic apnea in mice: a model for thermal stress causing SIDS. J. Appl. Physiol. 97, 669–674.
Gozal D., Gozal E., Reeves S. R., and Lipton A. J. (2002) Gasping and autoresuscitation in the developing rat: effect of antecedent intermittent hypoxia. J. Appl. Physiol. 92, 1141–1144.
Deshpande P., Khurana A., Hansen P., Wilkins D., and Thach B. T. (1999) Failure of autoresuscitation in weanling mice: significance of cardiac glycogen and heart rate regulation. J. Appl. Physiol. 87, 203–210.
Harper R. M., Kinney H. C., Fleming P. J., and Thach B. T. (2000) Sleep influences on homeostatic functions: implications for sudden infant death syndrome. Respir. Physiol. 119, 123–132.
Hunt C. E. (1992) The cardiorespiratory control hypothesis for sudden infant death syndrome. Clin. Perinatol. 19, 757–771.
Poets C. F., Meny R. G., Chobanian M. R., and Bonofiglo R. E. (1999) Gasping and other cardiorespiratory patterns during sudden infant deaths. Pediatr. Res. 45, 350–354.
Kara T., Narkiewicz K., and Somers V. K. (2003) Chemoreflexes: physiology and clinical implications. Acta Physiol. Scand. 177, 377–384.
Solomon I. C. (2004) Ionotropic excitatory amino acid receptors in pre-Botzinger complex play a modulatory role in hypoxia-induced gasping in vivo. J. Appl. Physiol. 96, 1643–1650.
Solomon I. C. (2005) Glutamate neurotransmission is not required for, but may modulate, hypoxic sensitivity of pre-Botzinger complex in vivo. J. Neurophysiol. 93, 1278–1284.
Ramirez J. M., Quellmalz U. J., and Richter D. W. (1996) Postnatal changes in the mammalian respiratory network as revealed by the transverse brainstem slice of mice. J. Physiol. 491, 799–812.
Telgkamp P. and Ramirez J. M. (1999) Differential responses of respiratory nuclei to anoxia in rhythmic brain stem slices of mice. J. Neurophysiol. 82, 2163–2170.
Ballanyi K., Volker A., and Richter D. W. (1994) Anoxia induced functional inactivation of neonatal respiratory neurones in vitro. Neuroreport 6, 165–168.
Richter D. W., Schmidt-Garcon P., Pierrefiche O., Bischoff A. M., and Lalley P. M. (1999) Neurotransmitters and neuromodulators controlling the hypoxic respiratory response in anaesthetized cats. J. Physiol. 514, 567–578.
Fazekas J. F., Alexander F. A. D., and Himwich H. E. (1941). Tolerance of the newborn to anoxia. J. Physiol. Lond. 134,282–287
Adolph E. F. (1969) Regulations during survival without oxygen in infant mammals. Respir. Physiol. 7, 356–368.
Jacobi M. S. and Thach B. T. (1989) Effect of maturation on spontaneous recovery from hypoxic apnea by gasping. J. Appl. Physiol. 66, 2384–2390.
Fewell J. E. and Wong V. H. (2002) Interleukin-1beta-induced fever does not alter the ability of 5- to 6-day-old rat pups to autoresuscitate from hypoxia-induced apnoea. Exp. Physiol. 87, 17–24.
Brockhaus J., Ballanyi K., Smith J. C., and Richter D. W. (1993) Microenvironment of respiratory neurons in the in vitro brainstem-spinal cord of neonatal rats. J. Physiol. 462, 421–445.
Voipio J. and Ballanyi K. (1997) Interstitial PCO2 and pH, and their role as chemostimulants in the isolated respiratory network of neonatal rats. J. Physiol. 499, 527–542.
Volker A., Ballanyi K., and Richter D. W. (1995) Anoxic disturbance of the isolated respiratory network of neonatal rats. Exp. Brain Res. 103, 9–19.
Ballanyi K., Onimaru H., and Homma I. (1999) Respiratory network function in the isolated brainstem-spinal cord of newborn rats. Prog. Neurobiol. 59, 583–634.
Greer J. J. and Carter J. E. (1995) Effects of cyanide on the neural mechanisms controlling breathing in the neonatal rat in vitro. Neurotoxicology 16, 211–215.
England S. J., Melton J. E., Douse M. A., and Duffin J. (1995) Activity of respiratory neurons during hypoxia in the chemodenervated cat. J. Appl. Physiol. 78, 856–861.
Morawietz G., Ballanyi K., Kuwana S., and Richter D. W. (1995) Oxygen supply and ion homeostasis of the respiratory network in the in vitro perfused brainstem of adult rats. Exp. Brain Res. 106, 265–274.
Sears T. A. (1964) Investigations on respiratory motoneurones of the thoracic spinal cord. Prog. Brain Res. 12, 259–273.
St. John W. M., Bartlett D. Jr. (1979) Comparison of phrenic motoneuron responses to hypercapnia and isocapnic hypoxia. J. Appl. Physiol. 46, 1096–1102.
Hwang J. C., Bartlett D. Jr., and St. John W. M. (1983) Characterization of respiratory-modulated activities of hypoglossal motoneurons. J. Appl. Physiol. 55, 793–798.
St John W. M. and Wang S. C. (1977) Alteration from apneusis to more regular rhythmic respiration in decerebrate cats. Respir. Physiol. 31, 91–106.
Bergmann F. and Keller B. U. (2004) Impact of mitochondrial inhibition on excitability and cytosolic Ca2+ levels in brainstem motoneurones from mouse. J. Physiol. 555, 45–59.
McCrimmon D. R., Ramirez J. M., Alford S., and Zuperku E. J. (2000) Unraveling the mechanism for respiratory rhythm generation. Bioessays. 22, 6–9.
Smith J. C., Ellenberger H. H., Ballanyi K., Richter D. W., and Feldman J. L. (1991) Pre-Botzinger complex: a brainstem region that may generate respiratory rhythm in mammals. Science 254, 726–729.
Onimaru H. and Homma I. (2003) A novel functional neuron group for respiratory rhythm generation in the ventral medulla. J. Neurosci. 23, 1478–1486
Mellen N. M., Janczewski W. A., Bocchiaro C. M., and Feldman J. L. (2003) Opioid-induced quantal slowing reveals dual networks for respiratory rhythm generation. Neuron 37, 821–826.
Peña F. and Ramirez J. M. (2002) Endogenous activation of serotonin-2A receptors is required for respiratory rhythm generation in vitro. J. Neurosci. 22, 11,055–11,064.
Peña F. and Ramirez J. M. (2004) Substance P-mediated modulation of pacemaker properties in the mammalian respiratory network. J. Neurosci. 24, 7549–7556.
Ogilvie M. D., Gottschalk A., Anders K., Richter D. W., and Pack A. I. (1992) A network model of respiratory rhythmogenesis. Am. J. Physiol. 263, R962-R975.
Ramirez J. M. and Lieske S. P. (2003) Commentary on the definition of eupnea and gasping. Respir. Physiol. Neurobiol. 139, 113–119.
Lieske S. P., Thoby-Brisson M., and Ramirez J. M. (2001) Reconfiguration of the central respiratory network under normoxic and hypoxic conditions. Adv. Exp. Med. Biol. 499, 171–178.
Ramirez J. M., Quellmalz U. J., Wilken B., and Richter D. W. (1998) The hypoxic response of neurones within the in vitro mammalian respiratory network. J. Physiol. 507, 571–582.
Koshiya N. and Smith J. C. (1999) Neuronal pacemaker for breathing visualized in vitro. Nature 400, 360–363.
Del Negro C. A., Morgado-Valle C., Hayes J. A., et al. (2005) Sodium and calcium-mediated pacemaker neurons and respiratory rhythm generation. J. Neurosci. 25, 446–453.
Hill A. (1991) Current concepts of hypoxic-ischemic cerebral injury in the term newborn. Pediatr. Neurol. 7, 317–325.
Latchaw R. E. and Truwit C. E. (1995) Imaging of perinatal hypoxic-ischemic brain injury. Semin. Pediatr. Neurol. 2, 72–89.
Roland E. H. and Hill A. (1995) Clinical aspects of perinatal hypoxic-ischemic brain injury. Semin. Pediatr. Neurol. 2, 57–71.
Suzuki R., Yamaguchi T., Li C. L., and Klatzo I. (1983) The effects of 5-minute ischemia in Mongolian gerbils: II. Changes of spontaneous neuronal activity in cerebral cortex and CA1 sector of hippocampus. Acta Neuropathol. (Berl). 60, 217–222.
Doolette D. J. and Kerr D. I. (1995) Hyperexcitability in CA1 of the rat hippocampal slice following hypoxia or adenosine. Brain Res. 677, 127–137.
Crepel V., Hammond C., Chinestra P., Diabira D., and Ben-Ari Y. (1993) A selective LTP of NMDA receptor-mediated currents induced by anoxia in CA1 hippocampal neurons. J. Neurophysiol. 70, 2045–2055.
Crepel V., Hammond C., Krnjevic K., Chinestra P., and Ben-Ari Y. (1993) Anoxia-induced LTP of isolated NMDA receptor-mediated synaptic responses. J. Neurophysiol. 69, 1774–1778.
Gozlan H., Diabira D., Chinestra P., and Ben-Ari Y. (1994) Anoxic LTP is mediated by the redox modulatory site of the NMDA receptor. J. Neurophysiol. 72, 3017–3022.
Hsu K. S. and Huang C. C. (1997) Characterization of the anoxia-induced long-term synaptic potentiation in area CA1 of the rat hippocampus. Br. J. Pharmacol. 122, 671–681.
Lyubkin M., Durand D. M., and Haxhiu M. A. (1997) Interaction between tetanus long-term potentiation and hypoxia-induced potentiation in the rat hippocampus. J. Neurophysiol. 78, 2475–2482.
Calabresi P., Saulle E., Centonze D., Pisani A., Marfia G. A., and Bernardi G. (2002) Postischaemic long-term synaptic potentiation in the striatum: a putative mechanism for cell type-specific vulnerability. Brain 125, 844–860.
Schmidt-Kastner R. and Freund T. F. (1991) Selective vulnerability of the hippocampus in brain ischemia. Neuroscience 40, 599–636.
Vyskocil F., Kritz N., and Bures J. (1972) Potassium-selective microelectrodes used for measuring the extracellular brain potassium during spreading depression and anoxic depolarization in rats. Brain Res. 39, 255–259.
Muller M. and Somjen G. G. (2000) Na(+) dependence and the role of glutamate receptors and Na(+) channels in ion fluxes during hypoxia of rat hippocampal slices. J. Neurophysiol. 84, 1869–1880.
Leão A. A. P. (1947), Further observations on the spreading depression of activity in the cerebral cortex. J Neurophysiol 10, 409–414.
Marshall W. H. (1959) Spreading cortical depression of Leao. Physiol. Rev. 39, 239–279.
Nicholson C. (1984) Comparative neurophysiology of spreading depression in the cerebellum. An. Acad. Bras. Cienc. 56, 481–494.
Somjen G. G., Aitken P. G., Czeh G. L., Herreras O., Jing J., and Young J. N. (1992) Mechanism of spreading depression: a review of recent findings and a hypothesis. Can. J. Physiol. Pharmacol. 70 Suppl, S248-S254.
Somjen G. G. (2001) Mechanisms of spreading depression and hypoxic spreading depression-like depolarization. Physiol. Rev. 81, 1065–1096.
Bahar S., Fayuk D., Somjen G. G., Aitken P. G., and Turner D. A. (2000) Mitochondrial and intrinsic optical signals imaged during hypoxia and spreading depression in rat hippocampal slices. J. Neurophysiol. 84, 311–324.
Hansen A. J. and Olsen C. E. (1980) Brain extracellular space during spreading depression and ischemia. Acta Physiol. Scand. 108, 355–365.
Perez-Pinzon M. A., Tao L., and Nicholson C. (1995) Extracellular potassium, volume fraction, and tortuosity in rat hippocampal CA1, CA3, and cortical slices during ischemia. J. Neurophysiol. 74, 565–573.
Sykova E., Svoboda J., Polak J., and Chvatal A. (1994) Extracellular volume fraction and diffusion characteristics during progressive ischemia and terminal anoxia in the spinal cord of the rat. J. Cereb. Blood Flow Metab. 14, 301–311.
Chebabo S. R., Hester M. A., Jing J., Aitken P. G., and Somjen G. G. (1995) Interstitial space, electrical resistance and ion concentrations during hypotonia of rat hippocampal slices. J. Physiol. 487 (Pt 3), 685–97.
Jing J., Aitken P. G., and Somjen G. G. (1994) Interstitial volume changes during spreading depression (SD) and SD-like hypoxic depolarization in hippocampal tissue slices. J. Neurophysiol. 71, 2548–2551.
Kreisman N. R., LaManna J. C., Liao S. C., Yeh E. R., and Alcala J. R. (1995) Light transmittance as an index of cell volume in hippocamppal slices: optical differences of interfaced and submerged positions. Brain Res. 693, 179–186.
Turner D. A., Aitken P. G., and Somjen G. G. (1995) Optical mapping of transluence changes in rat hippocampal slices during hypoxia. Neurosci. Lett. 195, 209–213.
Melzian D., Scheufler E., Grieshaber M., and Tegtmeier F. (1996) Tissue swelling and intracellular pH in the CA1 region of anoxic rat hippocampus. J. Neurosci. Methods 65, 183–187.
Kreisman N. R., Soliman S., and Gozal D. (2000) Regional differences in hypoxic depolarization and swelling in hippocampal slices. J. Neurophysiol. 83, 1031–1038.
Hossmann K. A. (1971) Cortical steady potential, impedance and excitability changes during and after total ischemia of cat brain. Exp. Neurol. 32, 163–175.
Korf J., Klein H. C., Venema K., and Postema F. (1988) Increases in striatal and hippocampal impedance and extracellular levels of amino acids by cardiac arrest in freely moving rats. J. Neurochem. 50, 1087–1096.
Astrup J., Siesjo B. K., and Symon L. (1981) Thresholds in cerebral ischemia — the ischemic penumbra. Stroke 12, 723–725.
Choi D. W. (1988) Calcium-mediated neurotoxicity: relationship to specific channel types and role in ischemic damage. Trends Neurosci. 11, 465–469.
Kass I. S. and Lipton P. (1982) Mechanisms involved in irreversible anoxic damage to the in vitro rat hippocampal slice. J. Physiol. 332:459–472.
Roberts E. L. Jr. and Sick T. J. (1988) Calcium-sensitive recovery of extracellular potassium and synaptic transmission in rat hippocampal slices exposed to brief anoxia. Brain Res. 456, 113–119.
Siesjo B. K. (1992) Pathophysiology and treatment of focal cerebral ischemia. Part II: Mechanisms of damage and treatment. J. Neurosurg. 77, 337–354.
Leist M. and Nicotera P. (1998) Calcium and neuronal death. Rev. Physiol Biochem. Pharmacol. 132, 79–125.
Chow E. and Haddad G. G. (1998) Differential effects of anoxia and glutamate on cultured neocortical neurons. Exp. Neurol. 150, 52–59.
Kristian T. and Siesjo B. K. (1996) Calcium-related damage in ischemia. Life Sci. 59, 357–367.
Choi D. W. (1994) Calcium and excitotoxic neuronal injury. Ann. NY Acad. Sci. 747, 162–171.
Tapia R., Medina-Ceja L., and Peña F. (1999) On the relationship between extracellular glutamate, hyperexcitation and neurodegeneration, in vivo. Neurochem. Int. 34, 23–31.
Silver I. and Erecinska M. (1998) Oxygen and ion concentrations in normoxic and hypoxic brain cells. Adv. Exp. Med. Biol. 454, 7–16.
Calabresi P., Centonze D., Pisani A., Cupini L., and Bernardi G. (2003) Synaptic plasticity in the ischaemic brain. Lancet Neurol. 2, 622–629.
LoPachin R. M., Gaughan C. L., Lehning E. J., Weber M. L., and Taylor C. P. (2001) Effects of ion channel blockade on the distribution of Na, K, Ca and other elements in oxygen-glucose deprived CA1 hippocampal neurons. Neuroscience. 103, 971–983.
Peña F. and Tapia R. (1999) Relationships among seizures, extracellular amino acid changes, and neurodegeneration induced by 4-aminopyridine in rat hippocampus: a microdialysis and electroencephalographic study. J. Neurochem. 72, 2006–2014.
Peña F. and Tapia R. (2000) Seizures and neurodegeneration induced by 4-aminopyridine in rat hippocampus in vivo: role of glutamate- and GABA-mediated neurotransmission and of ion channels. Neuroscience 101, 547–561.
Boening J. A., Kass I. S., Cottrell J. E., and Chambers G. (1989) The effect of blocking sodium influx on anoxic damage in the rat hippocampal slice. Neuroscience 33, 263–268.
Haigney M. C., Lakatta E. G., Stern M. D., and Silverman H. S. (1994) Sodium channel blockade reduces hypoxic sodium loading and sodium-dependent calcium loading. Circulation 90, 391–399.
Weber M. L. and Taylor C. P. (1994) Damage from oxygen and glucose deprivation in hippocampal slices is prevented by tetrodotoxin, lidocaine and phenytoin without blockade of action potentials. Brain Res. 664, 167–177.
Fried E., Amorim P., Chambers G., Cottrell J. E., and Kass I. S. (1995) The importance of sodium for anoxic transmission damage in rat hippocampal slices: mechanisms of protection by lidocaine. J. Physiol. 489, 557–565.
Friedman J. E. and Haddad G. G. (1994) Removal of extracellular sodium prevents anoxia-induced injury in freshly dissociated rat CA1 hippocampal neurons. Brain Res. 641, 57–64.
Toner C. C. and Stamford J. A. (1997) Sodium channel blockade unmasks two temporally distinct mechanisms of striatal dopamine release during hypoxia/hypoglycaemia in vitro. Neuroscience 81, 999–1007.
Banasiak K. J., Burenkova O., and Haddad G. G. (2004) Activation of voltage-sensitive sodium channels during oxygen deprivation leads to apoptotic neuronal death. Neuroscience 126, 31–44.
Peers C. and Kemp P. J. (2004) Ion channel regulation by chronic hypoxia in models of acute oxygen sensing. Cell Calcium 36, 341–348.
Semenza G. L. (2000) HIF-1: mediator of physiological and pathophysiological responses to hypoxia. J. Appl. Physiol. 88, 1474–1480.
Semenza G. L. (2000) Oxygen-regulated transcription factors and their role in pulmonary disease. Respir. Res. 1, 159–162.
Sharp F. R. and Bernaudin M. (2004) HIF1 and oxygen sensing in the brain. Nat. Rev. Neurosci. 5, 437–448.
Jiang B. H., Rue E., Wang G. L., Roe R., and Semenza G. L. (1996) Dimerization, DNA binding, and transactivation properties of hypoxia-inducible factor 1. J. Biol. Chem. 271, 17,771–17,778.
Yu A. Y., Frid M. G., Shimoda L. A., Wiener C. M., Stenmark K., and Semenza G. L. (1998) Temporal, spatial, and oxygen-regulated expression of hypoxia-inducible factor-1 in the lung. Am. J. Physiol. 275, L818-L826.
Xia Y., Fung M. L., O’Reilly J. P., and Haddad G. G. (2000) Increased neuronal excitability after long-term O(2) deprivation is mediated mainly by sodium channels. Brain Res. Mol. Brain Res. 76, 211–219.
Hance A. J., Robin E. D., Simon L. M., Alexander S., Herzenberg L. A., and Theodore J. (1980) Regulation of glycolytic enzyme activity during chronic hypoxia by changes in ratelimiting enzyme content. Use of monoclonal antibodies to quantitate changes in pyruvate kinase content. J. Clin. Invest. 66, 1258–1264.
Ptashne K. A., Morin M. E., Hance A., and Robin E. D. (1985) Increased biosynthesis of pyruvate kinase under hypoxic conditions in mammalian cells. Biochim. Biophys. Acta. 844, 19–23.
Xia Y. and Haddad G. G. (1999) Effect of prolonged O2 deprivation on Na+ channels: differential regulation in adult versus fetal rat brain. Neuroscience. 94, 1231–1243.
Neubauer J. A. (2001) Physiological and pathophysiological responses to intermittent hypoxia. J. Appl. Physiol. 90, 1593–1599.
Young A., Home M., Churchward T., Freezer N., Holmes P., and Ho M. (2002) Comparison of sleep disturbance in mild versus severe Parkinson’s disease. Sleep. 25, 573–577.
Montplaisir J., Bedard M. A., Richer F., and Rouleau I. (1992) Neurobehavioral manifestations in obstructive sleep apnea syndrome before and after treatment with continuous positive airway pressure. Sleep. 15, S17-S19.
Beebe D. W. and Gozal D. (2002) Obstructive sleep apnea and the prefrontal cortex: towards a comprehensive model linking nocturnal upper airway obstruction to daytime cognitive and behavioral deficits. J. Sleep. Res. 11, 1–16.
Carpagnano G. E., Kharitonov S. A., Resta O., Foschino-Barbaro M. P., Gramiccioni E., and Barnes P. J. (2002) Increased 8-isoprostane and interleukin-6 in breath condensate of obstructive sleep apnea patients. Chest. 122, 1162–1167.
Macey P. M., Henderson L. A., Macey K. E., et al. (2002) Brain morphology associated with obstructive sleep apnea. Am. J. Respir. Crit. Care Med. 166, 1382–1387.
Belik J., Sienko A., and Light R. B. (1990) The effect of repeated intermittent hypoxia on pulmonary vasoconstriction in the newborn. Can. J. Physiol. Pharmacol. 68, 355–362.
Irlbeck M., Iwai T., Lerner T., and Zimmer H. G. (1997) Effects of angiotensin II receptor blockade on hypoxia-induced right ventricular hypertrophy in rats. J. Mol. Cell Cardiol. 29, 2931–2939.
McGuire M. and Bradford A. (1999) Chronic intermittent hypoxia increases haematocrit and causes right ventricular hypertrophy in the rat. Respir. Physiol. 117, 53–58.
Nattie E. E., Bartlett D. Jr., and Johnson K. (1978) Pulmonary hypertension and right ventricular hypertrophy caused by intermittent hypoxia and hypercapnia in the rat. Am. Rev. Respir. Dis. 118, 653–658.
Nattie E. E. and Doble E. A. (1984) Threshold of intermittent hypoxia-induced right ventricular hypertrophy in the rat. Respir. Physiol. 56, 253–259.
Faridy E. E., Sanii M. R., and Thliveris J. A. (1988) Fetal lung growth: influence of maternal hypoxia and hyperoxia in rats. Respir. Physiol. 73, 225–241.
Schwartz J. E., Kovach A., Meyer J., McConnell C., and Iwamoto H. S. (1998) Brief, intermittent hypoxia restricts fetal growth in Sprague-Dawley rats. Biol. Neonate. 73, 313–319.
Gozal E., Row B. W., Schurr A., and Gozal D. (2001) Developmental differences in cortical and hippocampal vulnerability to intermittent hypoxia in the rat. Neurosci Lett. 2001 305(3), 197–201.
Row B. W., Kheirandish L., Neville J. J., and Gozal D. (2002) Impaired spatial learning and hyperactivity in developing rats exposed to intermittent hypoxia. Pediatr. Res. 52, 449–453.
Li R. C., Row B. W., Gozal E., et al. (2003) Cyclooxygenase 2 and intermittent hypoxia-induced spatial deficits in the rat. Am. J. Respir. Crit. Care Med. 168, 469–475.
Li R. C., Row B. W., Kheirandish L., et al. (2004) Nitric oxide synthase and intermittent hypoxia-induced spatial learning deficits in the rat. Neurobiol. Dis. 17, 44–53.
Xu W., Chi L., Row B. W., et al. (2004) Increased oxidative stress is associated with chronic intermittent hypoxia-mediated brain cortical neuronal cell apoptosis in a mouse model of sleep apnea. Neuroscience. 126, 313–323.
Godukhin O., Savin A., Kalemenev S., and Levin S. (2002) Neuronal hyperexcitability induced by repeated brief episodes of hypoxia in rat hippocampal slices: involvement of ionotropic glutamate receptors and L-type Ca(2+) channels. Neuropharmacology. 42, 459–466.
Guntheroth W. G. and Kawabori I. (1975) Hypoxic apnea and gasping. J. Clin. Invest. 56, 1371–1377.
Fewell J. E., Smith F. G., Ng V. K., Wong V. H., and Wang Y. (2000) Postnatal age influences the ability of rats to autoresuscitate from hypoxic-induced apnea. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279, R39-R46.
Meerson F. Z., Ustinova E. E., and Orlova E. H. (1987) Prevention and elimination of heart arrhythmias by adaptation to intermittent high altitude hypoxia. Clin. Cardiol. 10, 783–789.
Meerson F. Z., Ustinova E. E., and Manukhina E. B. (1989) Prevention of cardiac arrhythmias by adaptation to hypoxia: regulatory mechanisms and cardiotropic effect. Biomed. Biochim. Acta. 48, S83-S88.
Kitaev M. I., Aitbaev K. A., and Liamtsev V. T. (1999) Effect of hypoxic hypoxia on development of atherosclerosis in rabbits Aviakosm. Ekolog. Med. 33, 54–57.
Knaupp W., Khilnani S., Sherwood J., Scharf S., and Steinberg H. (1992) Erythropoietin response to acute normobaric hypoxia in humans. J. Appl. Physiol. 73, 837–840.
Koistinen P. O., Rusko H., Irjala K., et al. (2000) EPO, red cells, and serum transferrin receptor in continuous and intermittent hypoxia. Med. Sci. Sports Exerc. 32, 800–804.
Rutherford C. J., Schneider T. J., Dempsey H., Kirn D. H., Brugnara C., and Goldberg M. A. (1994) Efficacy of different dosing regimens for recombinant human erythropoietin in a simulated perisurgical setting: the importance of iron availability in optimizing response. Am. J. Med. 96, 139–145.
Moore-Gillon J. C. and Cameron I. R. (1985) Right ventricular hypertrophy and polycythaemia in rats after intermittent exposure to hypoxia. Clin. Sci. 69, 595–599.
Cao K. Y., Zwillich C. W., Berthon-Jones M., and Sullivan C. E. (1992) Increased normoxic ventilation induced by repetitive hypoxia in conscious dogs. J. Appl. Physiol. 73, 2083–2088.
Kinkead R., Zhan W. Z., Prakash Y. S., Bach K. B., Sieck G. C., and Mitchell G. S. (1998) Cervical dorsal rhizotomy enhances serotonergic innervation of phrenic motoneurons and serotonin-dependent long-term facilitation of respiratory motor output in rats. J. Neurosci. 18, 8436–8443.
Turner D. L. and Mitchell G. S. (1997) Long-term facilitation of ventilation following repeated hypoxic episodes in awake goats. J. Physiol. 499, 543–550.
Blitz D. M. and Ramirez J. M. (2002) Long-term modulation of respiratory network activity following anoxia in vitro. J. Neurophysiol. 87, 2964–2971.
Coles S. K. and Dick T. E. Neurones in the ventrolateral pons are required for post-hypoxic frequency decline in rats. J. Physiol. 497, 79–94.
Millhorn D. E., Eldridge F. L., and Waldrop T. G. (1980) Prolonged stimulation of respiration by endogenous central serotonin. Respir. Physiol. 42, 171–188.
Hayashi F., Coles S. K., Bach K. B., Mitchell G. S., and McCrimmon D. R. (1993) Time-dependent phrenic nerve responses to carotid afferent activation: intact vs decerebellate rats. Am. J. Physiol. 265, R811-R819.
Fregosi R. F. and Mitchell G. S. (1994) Long-term facilitation of inspiratory intercostal nerve activity following carotid sinus nerve stimulation in cats. J. Physiol. 477, 469–479.
Bach K. B. and Mitchell G. S. (1998) Hypercapnia-induced long-term depression of respiratory activity requires alpha2-adrenergic receptors. J. Appl. Physiol. 84, 2099–2105.
Baker T. L. and Mitchell G. S. (2000) Episodic but not continuous hypoxia elicits long-term facilitation of phrenic motor output in rats. J. Physiol. 529, 215–219.
Baker T. L., Fuller D. D., Zabka A. G., and Mitchell G. S. (2001) Respiratory plasticity: differential actions of continuous and episodic hypoxia and hypercapnia. Respir. Physiol. 129, 25–35.
Babcock M., Shkoukani M., Aboubakr S. E., and Badr M. S. (2003) Determinants of long-term facilitation in humans during NREM sleep. J. Appl. Physiol. 94, 53–59.
Fuller D. D., Bach K. B., Baker T. L., Kinkead R., and Mitchell G. S. (2000) Long term facilitation of phrenic motor output. Respir. Physiol. 121, 135–146.
Powell F. L., Milsom W. K., and Mitchell G. S. (1998) Time domains of the hypoxic ventilatory response. Respir. Physiol. 112, 123–134.
Zabka A. G., Behan M., and Mitchell G. S. (2001) Long term facilitation of respiratory motor output decreases with age in male rats. J. Physiol. 531, 509–514.
Zabka A. G., Behan M., and Mitchell G. S. (2001) Time-dependent hypoxic respiratory responses in female rats are influenced by age and by the estrus cycle. J. Appl. Physiol. 91, 2831–2838.
Mironov S. L., Hartelt N., and Ivannikov M. V. (2005) Mitochondrial K(ATP) channels in respiratory neurons and their role in the hypoxic facilitation of rhythmic activity. Brain Res. 1033, 20–27.
Baker-Herman T. L. and Mitchell G. S. (2002) Phrenic long-term facilitation requires spinal serotonin receptor activation and protein synthesis. J. Neurosci. 22, 6239–6246.
Baker-Herman T. L., Fuller D. D., Bavis R. W., et al. (2004) BDNF is necessary and sufficient for spinal respiratory plasticity following intermittent hypoxia. Nat. Neurosci. 7, 48–55.
Bixler E. O., Vgontzas A. N., Lin H. M., et al. (2001) Prevalence of sleep-disordered breathing in women: effects of gender. Am. J. Respir. Crit. Care Med. 163, 608–613.
Gozal E. and Gozal D. (2001) Respiratory plasticity following intermittent hypoxia: developmental interactions. J. Appl. Physiol. 90, 1995–1999.
Moss I. R. (2000) Respiratory responses to single and episodic hypoxia during development: mechanisms of adaptation. Respir. Physiol. 121, 185–197.
Gaultier C. (2001) Abnormalities of the chemical control of breathing: Clinical correlates in infants and children. Pediatr. Pulmonol. 23, 114–117.
Kinney H. C., Filiano J. J., and White W. F. (2001) Medullary serotonergic network deficiency in the sudden infant death syndrome: review of a 15-year study of a single dataset. J. Neuropathol. Exp. Neurol. 60, 228–247.
Ling L., Fuller D. D., Bach K. B., Kinkead R., Olson E. B. Jr, and Mitchell G. S. (2001) Chronic intermittent hypoxia elicits serotonindependent plasticity in the central neural control of breathing. J. Neurosci. 21, 5381–5388.
Youssef F. F., Addae J. I., McRae A., and Stone T. W. (2001) Long-term potentiation protects rat hippocampal slices from the effects of acute hypoxia. Brain Res. 907, 144–150.
Levin S. and Godukhin O. (2005) Developmental changes in hyperexcitability of CA1 pyramidal neurons induced by repeated brief episodes of hypoxia in the rat hippocampal slices. Neurosci. Lett. 377, 20–24.
Payne R. S., Goldbart A., Gozal D., and Schurr A. (2004) Effect of intermittent hypoxia on long-term potentiation in rat hippocampal slices. Brain Res. 1029, 195–199.
Golder F. J. and Mitchell G. S. (2005) Spinal synaptic enhancement with acute intermittent hypoxia improves respiratory function after chronic cervical spinal cord injury. J. Neurosci. 25, 2925–2932.
Beguin P. C., Joyeux-Faure M., Godin-Ribuot D., Levy P., and Ribuot C. (2005) Acute intermittent hypoxia improves rat myocardium tolerance to ischemia. J. Appl. Physiol., 99, 1064–1069.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peña, F., Ramirez, JM. Hypoxia-induced changes in neuronal network properties. Mol Neurobiol 32, 251–283 (2005). https://doi.org/10.1385/MN:32:3:251
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/MN:32:3:251