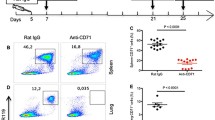
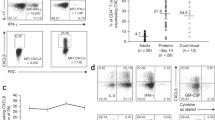
Abstract
The neonate has an increased susceptibility to infection, in part owing to an inability to produce antibody to thymus-independent antigens such as bacterial polysaccharides (PS). This poor response to PS antigens is likely owing to multiple factors. Neonatal B cells are of an immature phenotype, as evidenced by cell-surface marker characteristics and increased susceptibility to tolerance induction. The spleen of the neonate has a different cellular composition, which is most prominent in the marginal zone. Neonatal accessory cells such as macrophages and dendritic cells (DCs) appear to produce less stimulatory cytokines and an overabundance of inhibitory cytokines. This review examines the current data supporting the role of B cells and accessory cells in the neonatal unresponsiveness to PS antigens.
Similar content being viewed by others
References
Adderson EE: Antibody repertoires in infants and adults: effects of T-independent and T-dependent immunizations. Springer Semin Immunopathol 2001;23: 387–403.
Pabst HF, Kreth HW: Ontogeny of the immune response as a basis of childhood disease. J Pediatr 1980;97:519–534.
Bondada S, Garg M: Thymus-independent antigens. InP Snow EC, (ed.), Handbook of B and T Lymphocytes. Academic Press, Inc., San Diego, 1994, pp. 343–370.
Mond JJ, Lees A, Snapper CM: T cell-independent antigens type 2. Annu Rev Immunol 1995;13:655–692.
Snapper CM, Mond JJ: A model for induction of T cell-independent humoral immunity in response to polysaccharide antigens. J Immunol 1996;157:2229–2233.
Snapper CM, Rosas FR, Moorman MA, Mond JJ: Restoration of T cell-independent type 2 induction of Ig secretion by neonatal B cells in vitro. J Immunol 1997;158:2731–2735.
Ambrosino DM, Delaney NR, Shamberger RC: Human polysaccharide-specific B cells are responsive to pokeweed mitogen and IL-6. J Immunol 1990;144: 1221–1226.
Chelvarajan RL, Gilbert NL, Bondada S: Neonatal murine B lymphocytes respond to polysaccharide antigens in the presence of IL-1 and IL-6. J Immunol 1998;161:3315–3324.
Broome CV, Breiman RF: Pneumococcal vaccine—past present and future. N Engl J Med 1991;325:1506–1508.
Rennels MB, Edwards KM, Keyserling HL, Reisinger KS, Hogerman DA, Madore DV, et al. Safety and immunogenicity of heptavalent pneumococcal vaccine conjugated to CRM197 in United States infants. Pediatrics 1998;101:604–611.
Nossal GJ: Cellular mechanisms of immunologic tolerance. Annu Rev Immunol 1983;1:33–62.
Baker PJ: T cell regulation of the antibody response to bacterial polysaccharide antigens: an examination of some general characteristics and their implications. J Infect Dis 1992;165:S44–48.
Mosier DE, Zitron IM, Mond JJ, Ahmed A, Scher I, Paul WE: Surface immunoglobulin D as a functional receptor for a subclass of B lymphocytes. Immunol Rev 1977;37:89–104.
Scher I, Berning AK, Kessler S, Finkelman FD: Development of B lymphocytes in the mouse; studies of the frequency and distribution of surface IgM and IgD in normal and immune-defective CBA/N F1 mice. J Immunol 1980;125:1686–1693.
Carsetti R, Kohler G, Lamers MC: Transitional B cells are the target of negative selection in the B cell compartment. J Exp Med 1995;181:2129–2140.
Gathings WE, Kubagawa H, Cooper MD: A distinctive pattern of B cell immaturity in perinatal humans. Immunol Rev 1981;57:107–126.
Griffioen AW, Franklin SW, Zegers BJ, Rijkers GT: Expression and functional characteristics of the complement receptor type 2 on adult and neonatal B lymphocytes. Clin Immunol Immunopathol 1993;69:1–8.
Griffioen AW, Toebes EA, Zegers BJ, Rijkers GT: Role of CR2 in the human adult and neonatal in vitro antibody response to type 4 pneumococcal polysaccharide. Cell Immunol 1992;143:11–22.
Hamilton AM, Lehuen A, Kearney JF: Immunofluorescence analysis of B-1 cell ontogeny in the mouse. Int Immunol 1994;6:355–361.
Bofill M, Janossy G, Janossa M, Burford GD, Seymour GJ, Wernet P, Kelemen E: Human B cell development. II. Subpopulations in the human fetus. J Immunol 1985;134:1531–1538.
Barrett DJ, Sleasman JW, Schatz DA, Steinitz M: Human anti-pneumococcal polysaccharide antibodies are secreted by the CD5-B cell lineage. Cell Immunol 1992;143:66–79.
Brooks DA, Beckman IG, Bradley J, McNamara PJ, Thomas ME, Zola H: Human lymphocyte markers defined by antibodies derived from somatic cell hybrids. IV. A monoclonal antibody reacting specifically with a suppopulation of human B lymphocytes. J Immunol 1981;126:1373–1377.
Rijkers GT, Dollekamp EG, Zegers BJ: The in vitro B-cell response to pneumococcal polysaccharides in adults and neonates. Scand J Immunol 1987;25:447–452.
Bloem AC, Chand MA, Dollekamp I, Rijkers GT: Functional properties of human B cell subpopulations defined by monoclonal antibodies HB4 and FMC7. J Immunol 1988;140:768–773.
Saito S, Morii T, Umekage H, Makita K, Nishikawa K, Narita N, et al.: Expression of the interleukin-2 receptor gama chain on cord blood mononuclear cells. Blood 1996;87:3344–3350.
Zola H, Fusco M, Macardle PJ, Flego L, Roberton D: Expression of cytokine receptors by human cord blood lymphocytes: comparison with adult blood lymphocytes. Pediatr Res 1995;38:397–403.
Zola H, Fusco M, Weedon H, Macardle PJ, Ridings J, Roberton DM: Reduced expression of the interleukin-2-receptor gamma chain on cord blood lymphocytes: relationship to functional immaturity of the neonatal immune response. Immunology 1996;87:86–91.
Rijkers GT, Dollekamp I, Zegers BJ: 8-Mercaptoguanosine overcomes unresponsiveness of human neonatal B cells to polysaccharide antigens. J Immunol 1988;141: 2313–2316.
Bohnsack JF, Brown EJ: The role of the spleen in resistance to infection. Annu Rev Med 1986;37:49–59.
Goud SN, Muthusamy N, Subbarao B: Differential responses of B cells from the spleen and lymph node to TNP-Ficoll. J Immunol 1988;140:2925–2930.
Humphrey JH, Grennan D: Different macrophage populations distinguished by means of fluorescent polysaccharides. Recognition and properties of marginal-zone macrophages. Eur J Immunol 1981;11:221–228.
Kraal G, Ter Hart H, Meelhuizen C, Venneker G, Claassen E: Marginal zone macrophages and their role in the immune response against T-independent type 2 antigens: modulation of the cells with specific antibody. Eur J Immunol 1989;19:675–680.
Loder F, Mutschler B, Ray RJ, Paige CJ, Sideras P, Torres R, et al. B cell development in the spleen takes place in discrete steps and is determined by the quality of B cell receptor-derived signals. J Exp Med 1999; 190:75–89.
Timens W, Boes A, Rozeboom-Uiterwijk T, Poppema S: Immaturity of the human splenic marginal zone in infancy. Possible contribution to the deficient infant immune response. J Immunol 1989;143:3200–3206.
Rijkers GT, Sanders EA, Breukels MA, Zegers BJ: Infant B cell responses to polysaccharide determinants. Vaccine 1998;16:1396–1400.
Martin F, Oliver AM, Kearney JF: Marginal zone and B1 B cells unite in the early response against T-independent blood-borne particulate antigens. Immunity 2001;14: 617–629.
Guinamard R, Okigaki M, Schlessinger J, Ravetch JV: Absence of marginal zone B cells in Pyk-2-deficient mice defines their role in the humoral response. Nat Immunol 2000;1:31–36.
Halista SM, Johnson-Robbins LA, El-Mohandes AE, Lees A, Mond JJ, Katona IM: Characterization of early activation events in cord blood B cells after stimulation with T cell-independent activators. Pediatr Res 1998;43:496–503.
Haas KM, Hasegawa M, Steeber DA, Poe JC, Zabel MD, Bock CB, et al.: Complement receptors CD21/35 link innate and protective immunity during Streptococcus pneumoniae infection by regulating IgG3 antibody responses. Immunity 2002;17:713–723.
Freedman AS, Freeman G, Whitman J, Segil J, Daley J, Nadler LM: Pre-exposure of human B cells to recombinant IL-1 enhances subsequent proliferation. J Immunol 1988;141:3398–3404.
Bessler H, Komlos L, Punsky I, Ntambi JA, Bergman M, Straussberg R, Sirota L: CD14 receptor expression and lipopolysaccharide-induced cutokine production in preterm and term neonates. Biol Neonate 2001;80: 186–192.
Cohen L, Haziot A, Shen DR, Lin XY, Sia C, Harper R, et al.: CD14-independent responses to LPS require a serum factor that is absent from neonates. J Immunol 1995;155:5337–5342.
Hebra A, Strange P, Egbert JM, Ali M, Mullinax A, Buchanan E: Intracellular cytokine production by fetal and adult monocytes. J Pediatr Surg 2001;36:1321–1326.
Peters AM, Bertram P, Gahr M, Speer CP: Reduced secretion of interleukin-1 and tumor necrosis factor-alpha by neonatal monocytes. Biol Neonate 1993;63:157–162.
Weatherstone KB, Rich EA: Tumor necrosis factor/cachectin and interleukin-1 secretion by cord blood monocytes from premature and term neonates. Pediatr Res 1989;25:342–346.
Glover DM, Brownstein D, Burchett S, Larsen A, Wilson CB: Expression of HLA class II antigens and secretion of interleukin-1 by monocytes and macrophages from adults and neonates. Immunology 1987;61:195–201.
Pillay V, Savage N, Laburn H: Circulating cytokine concentrations and cytokine production by monocytes from newborn babies and adults. Pflugers Arch 1994;428: 197–201.
Schibler KR, Liechty KW, White WL, Rothstein G, Christensen RD: Defective production of interleukin-6 by monocytes: a possible mechanism underlying several host defense deficiencies of neonates. Pediatr Res 1992;31:18–21.
Berner, R, Csorba J, Brandis M: Different cytokine expression in cord blood mononuclear cells after stimulation with neonatal sepsis or colonizing strains of Streptococcus agalactiae. Pediatr Res 2001;49:691–697.
Muller K, Zak M, Nielsen S, Pedersen FK, de Nully P, Bendtzen K: In vitro cytokine production and phenotype expression by blood mononuclear cells from umbilical cords, children and adults. Pediatr Allergy Immunol 1996;7:117–124.
Karlsson H, Hessle C, Rudin A: Innate immune responses of human neonatal cells to bacteria from the normal gastrointestinal flora. Infect Immun 2002;70: 6688–6696.
Bryson YJ, Winter HS, Gard SE, Fischer TJ, Stiehm ER: Deficiency of immune interferon production by leukocytes of normal newborns. Cell Immunol 1980;55:191–200.
Balazs M, Martin F, Zhou T, Kearney J: Blood dendritic cells interact with splenic marginal zone B cells to initiate T-independent immune responses. Immunity 2002;17:341–352.
Langrish CL, Buddle JC, Thrasher AJ, Goldblatt D: Neonatal dendritic cells are intrinsically biased against Th-1 immune responses. Clin Exp Immunol 2002;128: 118–123.
Liu E, Tu W, Law HK, Lau YL: Decreased yield, phenotypic expression and function of immature monocyte-derived dendritic cells in cord blood. Br J Haematol 2001;113:240–246.
Lafontaine M, Landry D, Blanc-Brunat N, Pelletier M, Montplaisir S: IL-1 production by human thymic dendritic cells: studies on the interrelation with DC accessory function. Cell Immunol 1991;135:431–444.
Ismaili J, Rennesson J, Aksoy E, Vekemans J, Vincart B, Amraoui Z, et al.: Monophosphoryl lipid A activates both human dendritic cells and T cells. J Immunol 2002;168:926–932.
Baker PJ, Hiernaux JR, Fauntleroy MB, Prescott B, Cantrell JL, Rudbach JA: Inactivation of suppressor T-cell activity by nontoxic monophosphoryl lipid A. Infect Immun 1988a; 56:1076–1083.
Baker PJ, Hiernaux JR, Fauntleroy MB, Stashak PW, Prescott B, Cantrell JL, Rudbach JA: Ability of monophosphoryl lipid A to augment the antibody response of young mice. Infect Immun 1988b;56:3064–3066.
Garg M, Subbarao B: Immune responses of systemic and mucosal lymphoid organs to Pnu-Imune vaccine as a function of age and the efficacy of monophosphoryl lipid A as an adjuvant. Infect Immun 1992;60:2329–2336.
Krieg AM, Yi AK, Matson S, Waldschmidt TJ, Bishop GA, Teasdale R, et al.: CpG motifs in bacterial DNA trigger direct B-cell activation. Nature 1995;374:546–549.
Chelvarajan RL, Raithatha R, Venkataraman C, Kaul R, Han SS, Robertson DA, Bondada S: CpG oligodeoxynucleotides overcome the unresponsiveness of neonatal B cells to stimulation with the thymus-independent stimuli anti-IgM and TNP-Ficoll. Eur J Immunol 1999; 29:2808–2818.
Kovarik J, Bozzotti P, Tougne C, Davis HL, Lambert PH, Krieg AM, Siegrist CA: Adjuvant effects of CpG oligodeoxynucleotides on responses against T-independent type 2 antigens. Immunology 2001;102:67–76.
Pichyangkul S, Youngvanitchit K, Kum-arb U, Krieg AM, Heppner DG, Walsh DS: Whole blood cultures to assess the immunostimulatory activities of CpG oligodeoxynucleotides. J Immunol Methods 2001;247:83–94.
Hartmann G, Krieg AM: Mechanism and function of a newly identified CpG DNA motif in human primary B cells. J Immunol 2000;164:944–953.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Landers, C.D., Chelvarajan, R.L. & Bondada, S. The role of B cells and accessory cells in the neonatal response to TI-2 antigens. Immunol Res 31, 25–36 (2005). https://doi.org/10.1385/IR:31:1:25
Issue Date:
DOI: https://doi.org/10.1385/IR:31:1:25