Abstract
Background. 5-Fluorouracil remains a key drug in the treatment of colorectal cancer, and the development of a simple and effective test for selecting patients likely to benefit from postoperative adjuvant chemotherapy is an important objective.
Aim of the Study. This study aimed to clarify the feasibility of measuring apoptotic cell rate (AI%) in tumor after short-term oral 5-fluorouracil administration prior to surgery with the objective of establishing a simpler method to test for sensitivity.
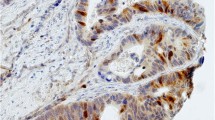
Methods. Forty-five colorectal cancer patients were allocated to two groups, and 21 patients were given oral 5-FU for 3 d prior to surgery. The AI% in surgical specimen, detected by TUNEL staining, was compared in the 5-FU-loaded and control groups. The correlation of AI% with 5-FU metabolic enzyme mRNA levels in tumor was also evaluated.
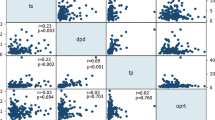
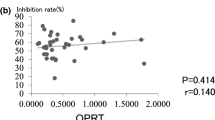
Results. The AI% was significantly higher in the tumor tissue of patients receiving 5-FU than in the control group (p<0.0005). Although insignificant, thymidylate synthase mRNA level and orotate phosphoribosyl transferase mRNA demonstrated a weak positive correlation with AI%.
Conclusions. The AI% measurement in tumor tissue following a 5-FU oral load for 3 d prior to surgery was feasible. It remains to be elucidated if this measurement as a new 5-FU sensitivity test reflects the prognosis with 5-FU-based postoperative adjuvant chemotherapy.
Similar content being viewed by others
References
Popov IP, Jelic SB, Matijasevic MM, Grbic B, Babic DR. Mayo regimen plus three different second-line chemotherapy regimens in sequential therapy in patients with advanced colorectal cancer (ACRC). J Exp Clin Cancer Res 2004;23:395–401.
Kemeny N, Garay CA, Gurtler J, Hochster H, Kennedy P, Benson A, et al. Randomized multicenter phase II trial of bolus plus infusional fluorouracil/leucovorin compared with fluorouracil/leucovorin plus oxaliplatin as third-line treatment of patients with advanced colorectal cancer. J Clin Oncol 2004;22:4753–4761.
Keam SJ, Dunn CJ, Figgitt DP. Oxaliplatin: in operable colorectal cancer. Drugs 2005;65:89–96.
Kubota T. Fluoropyrimidines in Colon Cancer. J Jpn Soc Coloproctol 2000;53:1018–1022.
Ansfield FJ, Schroeder JM, Curreri AR. Five years clinical experience with 5-fluorouracil. JAMA 1962;181:295–299.
Kemeny N. Current approaches to metastatic colorectal cancer. Sem Oncol 1994;21:67–75.
Parker WB, Cheng YC. Metabolism and mechanism of action of 5-fluorouracil. Pharmac Ther 1990;48:381–395.
Danenberg PV. Thymidylate synthetase—a target enzyme in cancer chemotherapy. Biochem. Biophys Acta 1977;473:73–92.
Spiegelman S, Sawyer R, Nayak R, et al. Improving the anti-tumor activity of 5-fluorouracil by increasing its incorporation into RNA via metabolic modulation. Proc Natl Acad Sci USA 1980;77:4966–4970.
Cory JG, Breland JC, Carter GL. Effect of 5-fluorouracil on RNA metabolism in Novikoff hepatoma cells. Cancer Res 1979;39:4905–4913.
Heggie GC, Sommadossi JP, Cross DS, et al. Clinical pharmacokinetics of 5- fluorouracil and its metabolites in plasma, urine, and bile. Cancer Res 1987;47:2203–2206.
Kubota T, Sasano N, Abe O, et al. Potential of the histoculture drug-response assay to contribute to cancer patient survival. Clin Cancer Res 1995;1:1537–1543.
Johnson MR, Wang K, Smith JB, Heslin MJ, Diasio RB. Quantitation of dihydropyrimidine dehydrogenase expression by real-time reverse transcription polymerase chain reaction. Anal Biochem 2000;278:175–184.
Ichikawa W, Uetake H, Shirota Y, et al. Both gene expression for orotate phosphoribosyl transferase and its ratio to dihydropyrimidine dehydrogenase influence outcome following fluoropyrimidine-based chemotherapy for metaststic colorectal cancer. Br J Cancer 2003;89:1486–1492.
Ciccolini J, Mercier C, Blachon MF, Favre R, Durand A, Lacarelle B. A simple and rapid high-performance liquid chromatographic (HPLC) method for 5-fluorouracil (5-FU) assy in plasma and possible detection of patients with impaired dihydropyrimidine dehydrogenase (DPD) activity. J Clin Pharm Ther 2004;29:307–315.
Garg MB, Sevester JC, Sakoff JA, Ackland SP. Simple liquid chromatographic method for the determination of uracil and dihydorouracil plasma levels: a potential pretreatment predicter of 5-fluorouracil toxicity. J Chromatogr B Analyt Technol Biomed Life Sci 2002;774:223–230.
Gamelin E, Boisdron CM, Guerin MV, et al. Correlation between uracil and dihydorouracil plasma ratio, 5-fluorouracil (5-FU) pharmacokinetic parameters, and tolerance in patients with advanced colorectal cancer. A potential interest for predicting 5-FU toxicity and determinig optimal 5-FU dosage. J Clin Oncol 1999;17:1105.
The Colorectal Cancer Chemotherapy Study Group of Japan. Five-year results of a randomized controlled trial of adjuvant chemotherapy for curatively resected colorectal carcinoma. The Colorectal Cancer Chemotherapy Study Group of Japan. Jpn J Clin Oncol 1995;25:91–103.
Izumi S, Hishikawa Y, Shin M, Koji T. Morphological features of apoptosis. The Medical Frontline 2002;57:2454–2459.
Edler D, Glimelius B, Hallstrom M, et al. Thymidylate synthase expression in colorectal cancer: a prognostic and predictive marker of benefit from adjuvant fluorouracil-based chemotherapy. J Clin Oncol 2002;20:1721–1728.
Fukushima M, Nomura H, Murakami Y, Shirasaka T, Aiba K. Estimation of pathways of 5-fluorouracil anabolism in human cancer cells in vitro and vivo. Jpn J Cancer Chemother 1966;23:721–731.
Hamaji M, Nishida T, Miyazaki S, Nishida Y. Dihydropyrimidine dehydrogenase,,ÄÄand thymidylate synthase in colon carcinoma. Jpn J Cancer Chemother 2002;29:889–893.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kawai, K., Watabe, S., Matsuda, M. et al. Correlation between expression of orotate phosphoribosyl transferase and 5-fluorouracil sensitivity, as measured by apoptosis index in colorectal cancer tissue. Int J Gastrointest Canc 35, 197–203 (2005). https://doi.org/10.1385/IJGC:35:3:197
Issue Date:
DOI: https://doi.org/10.1385/IJGC:35:3:197