Abstract
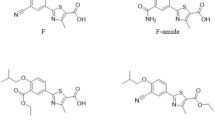
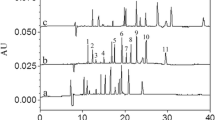
A new micellar electrokinetic capillary chromatographic (MEKC) method has been developed for simultaneous quantitation of atorvastatin (AT) and its related substances. The separation was carried out in an extended light path capillary at applied voltage of 30 kV using a background electrolyte consisting of 10 mM sodium tetraborate buffer pH 9.5, 50 mM sodium dodecyl sulphate and 20% (v/v) methanol. The addition of methanol to the running buffer resulted in a very effective choice to achieve resolution between the peaks of charged substances adjacent to AT as well as the peaks of neutral drug-related substances. Linear calibration curves were established over the concentration range 100–1,200 μg mL−1 for AT and 1.0–12.5 μg mL−1 for related substances. The proposed MEKC procedure has been validated with respect to selectivity, precision, linearity, limits of detection, and quantitation, accuracy and robustness. The method has been successfully applied to the determination of AT and purity evaluation of bulk drug and formulated products.





Similar content being viewed by others
References
Kaufmann DW, Kelly JP, Rosenberg L, Anderson TE, Michell AA (2002) JAMA 287:337–344. doi:10.1001/jama.287.3.337
Mc Taggart F, Jones P (2008) Cardiovasc Drugs Ther 22:321–338. doi:10.1007/s10557-008-6113-z
Nirogi R, Mudigonda K, Kandikere V (2007) J Pharm Biomed Anal 44:379–387. doi:10.1016/j.jpba.2007.02.008
Nováková L, Šatínský D, Solich P (2008) TrAC Trends Anal Chem 27:352–367
Bullen WW, Miller RA, Hayes RN (1999) J Am Soc Mass Spectrom 10:55–66. doi:10.1016/S1044-0305(98)00118-4
Jemal M, Ouyang Z, Chen BC, Teitz D (1999) Rapid Commun Mass Spectrom 13:1003–1015. doi:10.1002/(SICI)1097-0231(19990615)13:11<1003::AID-RCM597>3.0.CO;2-L
Hermann M, Christensen H, Reubsaet JLE (2005) Anal Bioanal Chem 382:1242–1249. doi:10.1007/s00216-005-3266-5
Borek-Dohalsky V, Huclova J, Barrett B, Nemec B, Ulc I, Jelinek I (2006) Anal Bioanal Chem 386:275–285. doi:10.1007/s00216-006-0655-3
Nirogi R, Kandikere VN, Shukla M, Mudigonda K, Maurya S, Boosi R, Anjaneyulu Y (2006) Biomed Chromatogr 20:924–936. doi:10.1002/bmc.622
Ma L, Dong J, Chen XJ, Wang GJ (2007) Chromatographia 65:737–741. doi:10.1365/s10337-007-0236-4
Altuntas TG, Erk N (2004) J Liq Chromatogr Relat Technol 27:83–93. doi:10.1081/JLC-120027087
Pasha MK, Muzeeb S, Basha SJS, Shashikumar D, Mullangi R, Srinivas NR (2006) Biomed Chromatogr 20:282–293. doi:10.1002/bmc.561
Seshachalam U, Kothapally CB (2008) J Liq Chromatogr Relat Technol 31:714–721. doi:10.1080/10826070701854402
Erk N (2004) Crit Rev Anal Chem 34:1–7. doi:10.1080/10408340490273717
Dogan-Topal B, Uslu B, Özkan SA (2007) Comb Chem High Throughput Screen 10:571–582. doi:10.2174/138620707782152407
Erk N (2003) Anal Lett 36:2699–2711. doi:10.1081/AL-120024645
Nagaraj Vipul K, Rajshree M (2007) Anal Sci 23:445–451. doi:10.2116/analsci.23.445
Stanisz B, Rafa W (2008) Chem Anal Warsz 53:417–428
Guihen E, Sisk GD, Scully NM, Glennon JD (2006) Electrophoresis 27:2338–2347. doi:10.1002/elps.200500899
Miller JM, Blackburn AC, Shi Y, Melzak AJ, Ando HY (2002) Electrophoresis 23:2833–2841. doi:10.1002/1522-2683(200209)23:17<2833::AID-ELPS2833>3.0.CO;2-7
European Pharmacopoeia, 6th edn (2007) Council of Europe, Strasbourg
The United States Pharmacopoeia, 31th edn (2007) United States Pharmacopeial Convention. Rockville, MD
Ertürk S, Aktaş ES, Ersoy L, Fiçicioğlu S (2003) J Pharm Biomed Anal 33:1017–1023. doi:10.1016/S0731-7085(03)00408-4
Mohammadi A, Rezanour N, Dogaheh MA, Bidkorbeh FG, Hashem M, Walker RB (2007) J Chromatogr B Analyt Technol Biomed Life Sci 846:215–221. doi:10.1016/j.jchromb.2006.09.007
Chaudhari BG, Patel NM, Shah PB (2007) Chem Pharm Bull (Tokyo) 55:241–246. doi:10.1248/cpb.55.241
Kadav AA, Vora DN (2008) J Pharm Biomed Anal 48:120–126. doi:10.1016/j.jpba.2008.05.018
Petkovska R, Cornett C, Dimitrovska A (2008) Anal Lett 41:992–1009. doi:10.1080/00032710801978566
Altria KD (1996) J Chromatogr A 735:43–56. doi:10.1016/0021-9673(95)00627-3
Altria KD, Marsh A, Sanger-van de Griend C (2006) Electrophoresis 27:2263–2282. doi:10.1002/elps.200600030
Terabe S (2004) Anal Chem 76:240A–246A. doi:10.1021/ac0415859
Nigović B, Vegar I (2008) Croat Chem Acta 81:615–622
Shaha RP, Kumar V, Singh S (2007) Proceedings of 12th ISMAS Symposium Cum Workshop on Mass Spectrometry, Goa, India
International Conference on Harmonization (ICH) (2005) Validation of analytical procedures: text and methodology Q2 (R1)
Mayer BX (2001) J Chromatogr A 907:21–37. doi:10.1016/S0021-9673(00)01057-8
Injac R, Bošković M, Kočevar N, Vovk T (2008) Anal Chim Acta 620:150–161. doi:10.1016/j.aca.2008.05.007
Injac R, Kočevar N, Kreft S (2007) Anal Chim Acta 594:119–127. doi:10.1016/j.aca.2007.05.003
Miller JN, Miller JC (2000) Statistics and chemometrics for analytical chemistry, 4th edn. Pearson Education, Edinburgh
Acknowledgments
This work was supported through a grant (Investigation of new methods in analysis of drugs and bioactive substances) from the Ministry of Science, Education and Sports of the Republic of Croatia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nigović, B., Damić, M., Injac, R. et al. Analysis of Atorvastatin and Related Substances by MEKC. Chroma 69, 1299–1305 (2009). https://doi.org/10.1365/s10337-009-1049-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-009-1049-4