Abstract
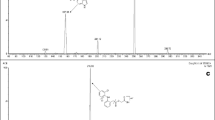
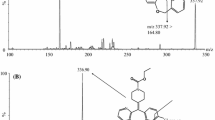
This study presents a high-performance liquid chromatography–electrospray ionization–mass spectrometric (LC–ESI–MS) method for the simultaneous determination of tramadol and acetaminophen in human plasma using phenacetinum as the internal standard. After alkalization with saturated sodium bicarbonate, both compounds were extracted from human plasma with ethyl acetate and were separated by HPLC on a Hanbon LiChrospher CN column with a mobile phase of 10 mM ammonium acetate buffer containing 0.5% formic acid–methanol (40:60, v/v) at a flow rate of 1 mL min−1. Analytes were determined using electrospray ionization in a single quadrupole mass spectrometer. LC–ESI–MS was performed in the positive selected-ion monitoring (SIM) mode using target ions at [M+H]+ m/z 264.3 for tramadol, [M+H]+ m/z 152.2 for acetaminophen and [M+H]+ m/z 180.2 for phenacetinum. Calibration curves were linear over the range of 5–600 ng mL−1 for tramadol and 0.03–16 μg mL−1 for acetaminophen. The inter-run relative standard deviations were less than 14.4% for tramadol and 12.3% for acetaminophen. The intra-run relative standard deviations were less than 9.3% for tramadol and 7.9% for acetaminophen. The mean plasma extraction recovery for tramadol and acetaminophen were in the ranges of 82.7–85.9 and 83.6–85.3%. The method was applied to study the pharmacokinetics of a new formulation of tramadol/acetaminophen tablet in healthy Chinese volunteers.





Similar content being viewed by others
References
Ortho-McNeil Ultracet: tramadol hydrochloride/acetaminophen tablets. Available online
Raffa RB (2001) J Clin Pharmacol Therap 26:257–264
Karen M, Lesley JS (2003) Drugs 63:1079–1086
Campanero MA, Garcia-Quetglas E, Sadaba B, Azanza JR (2004) J Chromatogr A 1031:219–228
Gu Y, Fawcett JP (2005) J Chromatogr B Analyt Technol Biomed Life Sci 821:240–243
Rouini MR, Ardakani YH, Soltani F, Aboul-Enein HY, Foroumadi A (2006) J Chromatogr B Analyt Technol Biomed Life Sci 830:207–211
Sha YF, Shen S, Duan GL (2005) J Pharm Biomed Anal 37:143–147
Leis HJ, Fauler G, Windischhofer W (2004) J Chromatogr B Analyt Technol Biomed Life Sci 804:369–374
Gambaro V, Benvenuti C, De. FL, Dell AL, Fare F (2003) Farmaco 58:947–950
Ho ST, Wan JJ, Liaw WJ, Ho CM, Li JH (1991) J Chromatogr B Biomed Sci Appl 736:89–96
Tao Q, Stone DJJ, Borenstein MR, Jean-Bart V, Codd EE, Coogan TP, Desai-Krieger D, Liao S, Raffa RB (2001) J Chromatogr B Biomed Sci Appl 763:165–171
Musshoff F, Madea B, Stuber F, Stamer UM (2006) J Anal Toxicol 30:463–467
Lan J, Wang J, Wu D, Wang Y, Fawcett JP, Gu J (2006) Rapid Commun Mass Spectrom 20:3309–3312
Zhao LM, Chen XY, Cui JJ, Sunita M, Zhong DF (2004) Yao Xue Xue Bao 39:458–462
Campanero MA, Calahorra B, Garcia-Quetglas E, Lopez-Ocariz A, Honorato J (1999) J Pharm Biomed Anal 20:327–334
Vertzoni MV, Archontaki HA, Galanopoulou P (2003) J Pharm Biomed Anal 32:487–493
Yin QQ, Lam SS, Chow MS (2005) Rapid Commun Mass Spectrom 19:767–774
Chen X, Huang J, Kong Z, Zhong D (2005) J Chromatogr B Analyt Technol Biomed Life Sci 817:263–269
Tan ZR, Ouyang DS, Zhou G, Wang LS (2005) Chin J Pharm Anal 25:795–798
Satinder A, Michael WD (2005) Handbook of pharmaceutical analysis by HPLC, Elsevier, Amsterdam, pp 516
Guidance for industry, bioanalytical method validation, U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Veterinary Medicine (CVM) May 2001
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, T., Ding, L., Guo, X. et al. Simultaneous Determination of Tramadol and Acetaminophen in Human Plasma by LC–ESI–MS. Chroma 66, 171–178 (2007). https://doi.org/10.1365/s10337-007-0268-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-007-0268-9