Abstract
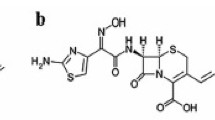
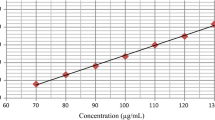
A fast and reliable single method was developed for rapid screening of cephalosporin oral dosage forms aimed to the detection of counterfeit and substandard drugs that might be illegally commercialised. Nine cephalosporin compounds, ceftibuten, cefatrizine, cefadroxil, cefaclor, cefprozil (Z) and (E)-isomers, cefixime, cephalexin and cefradine were separated in a six minutes chromatographic run by using a Symmetry® C18 column (50 × 4.6 mm I.D., 3.5 μm particle size) and an UV detector set at 254 nm. The mobile phase consisted of a mixture of acetonitrile-methanol-phosphate buffer (50 mM) containing 1-pentanesulfonic acid sodium salt (7 mM) adjusted to pH=2.1 with phosphoric acid (9:13:78 v/v/v). Validation of the method showed it to be robust, precise, accurate and linear over the concentration range of analysis.
Similar content being viewed by others
References
The Pharmaceutical Security Institute (1999). PSI general Assembly meeting at Worldwide Headquarters of Merck & co Inc.
World Health Organization (1992). Counterfeit Drugs: Report of a Joint WHO/IFPMA Workshop. WHO/DMP/CFD/92, Geneva
Combating Counterfeit drugs. A Report of the Food an Drug Administration (2004)
McGregor A (1997) The Lancet 350:1690
German Pharma Health Fundation. (1998) Drug Counterfeiting: an Unscroupulous Trade
European Federation of Pharmaceutical Industries and Associations (1999). Position Paper. Response to Commission’s Green Paper on Combating Counterfeiting and Piracy in the Single Market
World Health Organization (1999). Guidelines for the Development of Measures to Combat Counterfeit Drugs. WHO/EDM/QSM/99.1
World Health Organization (2000). WHO Global Strategy for Cointainment of Antimicrobial Resistance. WHO/CDS/CSR/DRS/2000.1
Roderick White E, Zarembo JE (1981) J Antibiotics 34 (7):836–844
Moore CM, Sato K, Katsumata Y (1991) J Chromatogr B 539:215–220
Morsch LM, Bittencourt J, Souza M (2002) J Pharm Biomed Anal 30:643–649
Yun EK, Prince AJ, McMillin JE, Welch E (1998) J Chromatogr B 712:145–152
Farag SA (1998) J AOAC Int 81:381–385
Ting S (1988) J Assoc Off Anal Chem 71:1123–1130
Nishino I, Fujitomo H, Umeda T (2000) J Chromatogr B 749:101–110
Bafeltowska JJ, Buszman E, Mandat K, Hawranek J (2002) J Chromatogr A 976:249–254
Gallo Martinez L, Campins Falco P, Sevillano Cabeza A (2002) J Pharm Biomed Anal 29:405–423
Johnson VM, Allanson JP, Causon RC (2000) J Chromatogr B 740:71–80
Guitton J, Laffont A, Rochet-Mingret L, Bonnefoy M, Bureau J (1998) J Chromatogr B 719:151–157
Nee Ling SS, Yuen KH, Barker SA (2003) J Chromatogr B 783:297–301
Petrauskas AA, Svedas VK (1991) J Chromatogr 585:3–34
Huang HS, Wu JR, Chen ML (1991) J Chromatogr 546:195–203
Pehourcq F, Jarry C (1998) J Chromatogr A 812:159–178
Patel Y P, Shah N, Bhoir I C, Sundaresan M (1998) J Chromatogr A 828:287–290
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Manna, L., Valvo, L. Development and Validation of a Fast Reversed-Phase Ion-Pairing Liquid Chromatographic Method for Simultaneous Determination of Eight Cephalosporin Antibiotics in Pharmaceutical Formulations. Chromatographia 60, 645–649 (2004). https://doi.org/10.1365/s10337-004-0432-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-004-0432-4