Abstract
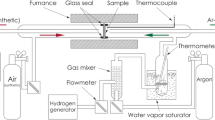
The structural stability of silver (Ag) in dual atmosphere exposure conditions, which are representative of solid oxide fuel cell (SOFC) current collector and gas seals, has been examined in the 600–800 °C temperature range. Experiments conducted on Ag tubular sections exposed to flowing H2-3% H2O (inside the tube) and air (outside the tube) showed extensive porosity formation along the grain boundaries in the bulk metal. Similar tubular sections, when exposed to air only (both inside and outside the tube), showed no bulk porosity or structural changes. It is postulated that the porosity formation in the bulk metal is related to the formation of gaseous H2O bubbles due to simultaneous diffusion of hydrogen and oxygen followed by subsequent interaction resulting in the formation of steam. Thermochemical processes that are responsible for structural degradation are presented and discussed. Based on experimental observations, it is concluded that Ag metal may not provide adequate long-term structural stability under a dual-environment condition that is typical of interconnects or gas seals in intermediate temperature SOFCs.
Similar content being viewed by others
References
W.A. Meulenberg, O. Teller, U. Flesch, H.P. Buchkremer, and D.J. Stover: “Improved Contacting by the Use of Silver in Solid Oxide Fuel Cells Up to an Operating Temperature of 800°C,” J. Mater. Sci. Eng., 2001, 36, pp. 3189–95.
Y. Harufuji: Japan Patent 06231784 (1994).
Y. Harufuji and S. Yoshida: Japan Patent 06084530 (1994).
A.V. Virkar, W.D. Prouse, P.C. Smith, and G.Y. Lin: US Patent 2002048700 (2002).
K.S. Weil and D.M. Paxton: “Development of An Oxidation Resistant Ceramic-to-Metal Braze for Use in YSZ-Based Electrochemical Devices,” Ceram. Eng. Sci. Proc., 2002, 23, pp. 785–92.
X. Xiao, C.Q. Tang, and Z.C. Xia: “Solid Oxide Fuel Cells With Different Cathode Materials,” Dianyuan Jishu, 2002, 26, pp. 128–30.
A.Q. Pham, B.W. Chung, and R.S. Glass: US Patent 2002127460 (2002).
U.D. Diekmann, H. Goebbels, and E. Sigismund: DE 2002014011 (2002).
R.L. Klueh and W.W. Mullins: “Some Observations on Hydrogen Embrittlement of Silver,” Trans. Met. Soc., 1968, 242, pp. 237–44.
G.L. Thomas: “Solubility of Hydrogen in Solid Copper, Silver, and Gold Obtained by a Rapid Quench and Extraction Technique,” Trans. AIME, 1967, 239, pp. 485–90.
W. Siegelin, K.H. Lieser, and H. Witte: Z. Elektrochem., 1957, 61, pp. 359–66.
H. Katsuta and R.B. McLellan: “Diffusivity of Hydrogen in Silver,” Scripta Metall., 1979, 13, pp. 65–66.
W. Eichenauer, H. Kunzig, and A.Z. Pebler: Metallkde, 1958, 49, pp. 220–25.
W. Eichenauer and G. Muller: Z. Metallkde, 1962, 53, pp. 321–25.
E.M. Otto: “Equilibrium Pressure of Oxygen Over Ag-O-Ag at Various Temperatures,” J. Electrochem. Soc., 1966, 113, pp. 643–45.
B. Chalmers, R. King, and R. Shuttleworth: Proc. R. Soc., 1948, A193, p. 465.
T.C. Wei and J. Philips: Adv. Catal., 1996, 41, p. 359.
X. Bao, G. Lehmpfuhl, G. Weinberg, R. Schlogl, and G. Ertl: J. Chem. Soc. Faraday Trans., 1992, 88, p. 865.
E.D. Hondros and A.J.W. Moore: “Evaporation and Thermal Etching,” Acta Metall., 1960, 8, pp. 647–53.
E. Fromm and E. Gebhardt: Gase und Kohlenstoff in Metallen, Springer-Verlag, Berlin/Heidelberg, 1976.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, P., Yang, Z., Viswanathan, V. et al. Observations on the structural degradation of silver during simultaneous exposure to oxidizing and reducing environments. J. of Materi Eng and Perform 13, 287–294 (2004). https://doi.org/10.1361/10599490419261
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1361/10599490419261