Summary
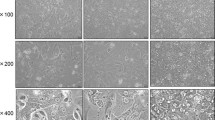
In order to evaluate their suitability for physiological and ecotoxicological studies, hepatocytes were isolated from the common mudpuppy (Necturus maculosus) using a two-step collagenase perfusion. Hepatocytes in primary culture were investigated for 14 d using light and electron microscopy and biochemical analyses. A typical perfusion yielded 1.7×105 viable hepatocytes per gram body weight with an average viability of 86±5%. The majority of isolated cells remained in suspension and formed aggregates. The viability of hepatocytes in primary culture was dependent on a fetal calf serum (FCS) concentration and incubation temperature. Viability was best at 8°C in Leibovitz L-15 medium supplemented with 5% FCS. The ultrastructural characteristics of freshly isolated hepatocytes resembled those of N. maculosus hepatocytes in vivo. Whereas hepatocyte viability remained relatively stable (around 80%) up to 14 d in culture, electron microscopic analyses revealed changes at ultrastructural level. The majority of hepatocytes retained similar structural characteristics to those in vivo up to 4 d. Loss of cellular polarity, fractionation of rough endoplasmic reticulum, formation of autophagosomes, and successive exhaustion of cellular glycogen deposits were observed with increased time in culture. Functional integrity, as estimated by tyrosine aminotransferase induction, decreased during the culture period. Ultrastructural and biochemical analyses indicate the need for further improvement of culture conditions. Nevertheless, isolated hepatocytes in primary culture for up to 4 d can be recommended as a model for physiological and toxicological studies in lower vertebrates.
Similar content being viewed by others
References
Ade, T.; Segner, H.; Hanke, W. Hormonal response of primary hepatocytes of the clawed toad, Xenopus laevis. Exp. Clin. Endocrinol. 103:21–27; 1995.
Agius, L.; Alam, N.; Aiston, S. Short-term regulation by insulin of glucose metabolism in isolated and cultured hepatocytes. In: Berry, M. N.; Edwards, A. M., ed. The hepatocyte review. Dordrecht: Kluwer Academic Publishers; 2000:317–341.
Berry, M. N.; Edwards, A. M. The hepatocyte review. Dordrecht: Kluwer Academic Publishers; 2000.
Berry, M. N.; Edwards, A. M.; Barritt, G. J. Isolated hepatocytes preparation, properties and applications. In: Burdon, R. H.; van Knippenberg, P. H., ed. Laboratory techniques in biochemistry and molecular biology. Vol. 21. Amsterdam: Elsevier; 1991:237–354.
Berry, M. N.; Friend, D. S. High yield preparation of isolated rat liver parenchymal cells. J. Cell Biol. 43:506–520; 1969.
Braunbeck, T.; Segner, H. Isolation and cultivation of teleost hepatocytes. In: Berry, M. N.; Edwards, A. M., ed. The hepatocyte review. Dordrecht: Kluwer Academic Publishers; 2000:49–71.
Braunbeck, T.; Storch, V. Senescence of hepatocytes isolated from rainbow trout (Oncorhynchus mykiss) in primary culture. Protoplasma 170:138–159; 1992.
Chiakulas, J. J.; Scheving, L. E.; Tsai, T. H. The behavior of urodele liver cells in vitro. Acta Hepato-Splenol. 12:257–268; 1965.
Diamondstone, T. I. Assay for tyrosine transaminase activity by conversion of p-hydroxyphenylpyruvate to p-hydroxybenzaldehyde. Anal. Biochem. 16:395–401; 1966.
Gurr, J. A.; Potter, V. R. Independent induction of tyrosine aminotransferase activity by dexamethasone and glucagons in isolated rat liver parenchymal cells in suspension and in monolayer culture in serum-free media. Exp. Cell Res. 126:237–248; 1980.
Haschemeyer, A. E. V.; Mathews, R. W. Temperature dependency of protein synthesis in isolated hepatocytes of Antarctic fish. Physiol. Zool. 56:77–87; 1983.
Hayashi, S.; Ooshiro, Z. Primary culture of the eel hepatocytes in the serum-free medium. Bull. Jap. Soc. Sci. Fish 52:1641–1651; 1986.
Ikeda, T.; Sawada, N.; Satoh, M.; Mori, M. Induction of tyrosine aminotrasferase of primary cultured rat hepatocytes depends on the organization of microtubules. J. Cell. Physiol. 175:41–49; 1998.
Janssens, P. A.; Kleineke, J.; Caine, A. G. Calcium-independent stimulation of glycogenolysis by arginine vasotocin and cateholamines in liver of the axolotl (Ambystoma mexicanum) in vitro. J. Endocrinol. 109:75–84; 1986.
Juillerant, M.; Marceau, N.; Coeytaux, S.; Sierra, F.; Kolodziejczyk, E.; Guigoz, Y. Expression of organ-specific structures and functions in longterm cultures of aggregates from adult rat liver cells. Toxicol. in Vitro 11:57–69; 1997.
Kido, H.; Fukusen, N.; Katunuma, N. Epidermal growth factor as a new regulator of induction of tyrosine aminotrasferase and tryptophan oxygenase by glucocorticoids. FEBS Lett. 223:223–226; 1987.
Klaunig, J.E.; Ruch, R.J.; Goldblatt, P.J. Trout hepatocyte culture: isolation and primary culture. In Vitro Cell. Dev. Biol. 21A:221–228; 1985.
Kloas, W.; Lutz, I.; Einspanier, R. Amphibians as a model to study endocrine disruptors: II. Estrogenic activity of environmental chemicals in vitro and in vivo. Sci. Total Environ. 225:59–68; 1999.
Kreamer, B. L.; Staecker, J. L.; Sawada, N.; Sattler, G. L.; Hsia, M. T. S.; Pitot, H. C. Use of a low-speed, iso-density percoll centrifugation method to increase the viability of isolated rat hepatocyte preparations. In Vitro Cell. Dev. Biol. 22A:201–211; 1986.
LeCluyse, E. L.; Bullock, P. L.; Parkinson, A. Strategies for restoration and maintenance of normal hepatic structure and function in long-term cultures of rat hepatocytes. Adv. Drug Deliv. Rev. 22:133–186; 1996.
Lipsky, M. M.; Sheridan, T. R.; Bennett, R. O.; May, E. B. Comparison of trout hepatocytes on different substrates. In Vitro Cell. Dev. Biol. 22A:360–362; 1986.
Malan-Shibley, L.; Iype P. The influence of culture conditions on cell morphology and tyrosine aminotransferase levels in rat liver epithelial cell lines. Exp. Cell Res. 131:363–371; 1981.
Råbergh, C. M.; Kane, A. S.; Reimschuessel, R.; Lipsky, M. M. Viability and induction of tyrosine aminotransferase in rainbow trout hepatocytes cultured on laminin and polylysine in a serum-free medium. Methods Cell Sci. 17:207–215; 1995.
Rouhani Rankouhi, T.; Sanderson, J. T.; van Holsteijn, I.; van Kooten, P.; Bosveld, A. T. C.; van der Berg, M. Effects of environmental and natural estrogens on vitellogenin production in hepatocytes of the brown frog (Rana temporaria). Aquat. Toxicol. 71:97–101; 2005.
Segner, H. Isolation and primary culture of teleost hepatocytes. Comp. Biochem. Physiol. 120A:71–81; 1998.
Seifter, S.; Dayton, S.; Novic, B.; Muntwyler, E. The estimation of glycogen with anthrone reagent. Arch. Biochem. 25:191–200; 1950.
Stanchfield, J. E.; Yager, J. D. An estrogen responsive primary amphibian liver cell culture system. Exp. Cell Res. 116:239–252; 1978.
Stanchfield, J. E.; Yager, J. D. Insulin effects on protein synthesis and secretion in primary cultures of amphibian hepatocytes. J. Cell. Physiol. 100:279–290; 1979.
Stanchfield, J. E.; Yager, J. D. Primary induction of vitellogenin synthesis in monolayer cultures of amphibian hepatocytes. J. Cell Biol. 84:468–475; 1980.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prelovšek, PM., Batista, U. & Bulog, B. Isolation and primary culture of Necturus maculosus (Amphibia: urodela) hepatocytes. In Vitro Cell.Dev.Biol.-Animal 42, 255–262 (2006). https://doi.org/10.1290/0601008.1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/0601008.1