Abstract
Objective
The aim of this study was to clarify the minimum number of examined lymph nodes (MNELNs) required to ensure the quality of lymph node detection and its impact on long-term survival in distal pancreatectomy for pancreatic ductal adenocarcinoma.
Methods
Clinicopathological characteristics and survival data of patients with resectable pancreatic cancer who underwent distal pancreatectomy between 2004 and 2017 were collected from the Surveillance, Epidemiology, and End Results database. The associations between the number of examined lymph nodes (ELNs) and number of positive lymph nodes (PLNs), stage migration, and overall survival were investigated through adjusted multivariate models with locally weighted scatterplot smoothing smoothing fitting curves and estimation of the structural breakpoints. Kaplan–Meier survival analysis and X-tile software were used to identify the ideal cut-off value for ELNs.
Results
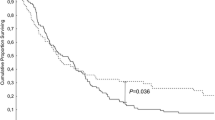
In total, 2610 consecutive patients who underwent distal pancreatectomy between 2004 and 2017 were included in this study. The optimal ELN count according to the associations between the number of ELNs and number of PLNs, odds ratio for stage migration, or hazard ratio for overall survival were 19, 17, and 19, respectively. Furthermore, the optimal division of ELN count for maximum overall survival was divided into three populations (ELN ≤ 8, ELN 9–18, ELN ≥ 19) based on X-tile software.
Conclusion
A minimal count of 19 lymph nodes was demanded to guarantee the quality of lymph node examination in patients with distal pancreatectomy. Long-term survival could be delimited by MNELNs. A sufficient number of ELNs could improve the accuracy of cancer staging and reflect a better overall survival.





Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30.
Huang L, Jansen L, Balavarca Y, et al. Resection of pancreatic cancer in Europe and USA: an international large-scale study highlighting large variations. Gut. 2019;68(1):130–9.
Tol JA, Gouma DJ, Bassi C, et al. Definition of a standard lymphadenectomy in surgery for pancreatic ductal adenocarcinoma: a consensus statement by the International Study Group on Pancreatic Surgery (ISGPS). Surgery. 2014;156(3):591–600.
Fujita T, Nakagohri T, Gotohda N, et al. Evaluation of the prognostic factors and significance of lymph node status in invasive ductal carcinoma of the body or tail of the pancreas. Pancreas. 2010;39(1):e48-54.
Fernández-Cruz L, Johnson C, Dervenis C. Locoregional dissemination and extended lymphadenectomy in pancreatic cancer. Dig Surg. 1999;16(4):313–9.
Miao Y, Lu Z, Yeo CJ, et al. Management of the pancreatic transection plane after left (distal) pancreatectomy: expert consensus guidelines by the International Study Group of Pancreatic Surgery (ISGPS). Surgery. 2020;168(1):72–84.
Huebner M, Kendrick M, Reid-Lombardo KM, et al. Number of lymph nodes evaluated: prognostic value in pancreatic adenocarcinoma. J Gastrointest Surg. 2012;16(5):920–6.
Valsangkar NP, Bush DM, Michaelson JS, et al. N0/N1, PNL, or LNR? The effect of lymph node number on accurate survival prediction in pancreatic ductal adenocarcinoma. J Gastrointest Surg. 2013;17(2):257–66.
Eskander MF, de Geus SWL, Kasumova GG, et al. Evolution and impact of lymph node dissection during pancreaticoduodenectomy for pancreatic cancer. Surgery. 2017;161(4):968–76.
Arrington AK, Price ET, Golisch K, Riall TS. Pancreatic cancer lymph node resection revisited: a novel calculation of number of lymph nodes required. J Am Coll Surg. 2019;228(4):662–9.
Camp RL, Dolled-Filhart M, Rimm DL. X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res. 2004;10(21):7252–9.
Tempero MA, Malafa MP, Al-Hawary M, et al. Pancreatic adenocarcinoma, Version 2.2017, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2017;15(8):1028–61.
Allen PJ, Kuk D, Castillo CF, et al. Multi-institutional validation study of the American Joint Commission on Cancer (8th Edition) changes for T and N staging in patients with pancreatic adenocarcinoma. Ann Surg. 2017;265(1):185–91.
Roalsø M, Aunan JR, Søreide K. Refined TNM-staging for pancreatic adenocarcinoma—real progress or much ado about nothing? Eur J Surg Oncol. 2020;46(8):1554–7.
Warschkow R, Widmann B, Beutner U, et al. The more the better-lower rate of stage migration and better survival in patients with retrieval of 20 or more regional lymph nodes in pancreatic cancer: a population-based propensity score matched and trend SEER analysis. Pancreas. 2017;46(5):648–57.
Ashfaq A, Pockaj BA, Gray RJ, Halfdanarson TR, Wasif N. Nodal counts and lymph node ratio impact survival after distal pancreatectomy for pancreatic adenocarcinoma. J Gastrointest Surg. 2014;18(11):1929–35.
Lahat G, Lubezky N, Gerstenhaber F, et al. Number of evaluated lymph nodes and positive lymph nodes, lymph node ratio, and log odds evaluation in early-stage pancreatic ductal adenocarcinoma: numerology or valid indicators of patient outcome? World J Surg Oncol. 2016;14(1):254.
Faron M, Vuarnesson H, Boher J-M, et al. How to reliably assess nodal status in distal pancreatectomy for adenocarcinoma. Pancreas. 2018;47(3):308–13.
Malleo G, Maggino L, Ferrone CR, et al. Number of examined lymph nodes and nodal status assessment in distal pancreatectomy for body/tail ductal adenocarcinoma. Ann Surg. 2019;270(6):1138–46.
Huang L, Jansen L, Balavarca Y, et al. Significance of examined lymph node number in accurate staging and long-term survival in resected stage I–II pancreatic cancer-more is better? A large international population-based cohort study. Ann Surg. 2019. https://doi.org/10.1097/SLA.0000000000003558.
Park HJ, You DD, Choi DW, Heo JS, Choi SH. Role of radical antegrade modular pancreatosplenectomy for adenocarcinoma of the body and tail of the pancreas. World J Surg. 2014;38(1):186–93.
Latorre M, Ziparo V, Nigri G, Balducci G, Cavallini M, Ramacciato G. Standard retrograde pancreatosplenectomy versus radical antegrade modular pancreatosplenectomy for body and tail pancreatic adenocarcinoma. Am Surg. 2013;79(11):1154–8.
Kitagawa H, Tajima H, Nakagawara H, et al. A modification of radical antegrade modular pancreatosplenectomy for adenocarcinoma of the left pancreas: significance of en bloc resection including the anterior renal fascia. World J Surg. 2014;38(9):2448–54.
Grossman JG, Fields RC, Hawkins WG, Strasberg SM. Single institution results of radical antegrade modular pancreatosplenectomy for adenocarcinoma of the body and tail of pancreas in 78 patients. J Hepato-Biliary-Pancreat Sci. 2016;23(7):432–41.
Abe T, Ohuchida K, Miyasaka Y, Ohtsuka T, Oda Y, Nakamura M. Comparison of surgical outcomes between radical antegrade modular pancreatosplenectomy (RAMPS) and standard retrograde pancreatosplenectomy (SPRS) for left-sided pancreatic cancer. World J Surg. 2016;40(9):2267–75.
Chun YS. Role of radical antegrade modular pancreatosplenectomy (RAMPS) and pancreatic cancer. Ann Surg Oncol. 2018;25(1):46–50.
Sahakyan MA, Haugvik SP, Rosok BI, et al. Can standardized pathology examination increase the lymph node yield following laparoscopic distal pancreatectomy for ductal adenocarcinoma? HPB (Oxford). 2018;20(2):175–81.
Noone A-M, Lund JL, Mariotto A, et al. Comparison of SEER treatment data with medicare claims. Med Care. 2016;54(9):e55–64.
Funding
This study was supported by the Shanghai Medical Engineering and Collaborative Innovation Center (10-20-308-40).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosure
Weishen Wang, Ziyun Shen, Jun Zhang, Hao Chen, Xiaxing Deng, Chenghong Peng, Junjie Xie, Zhiwei Xu, and Baiyong Shen have declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.


Rights and permissions
About this article
Cite this article
Wang, W., Shen, Z., Zhang, J. et al. A Novel Criterion for Lymph Nodes Dissection in Distal Pancreatectomy for Ductal Adenocarcinoma: A Population Study of the US SEER Database. Ann Surg Oncol 29, 1533–1539 (2022). https://doi.org/10.1245/s10434-021-10797-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-10797-2