Abstract
Background
This study aimed to analyze timing and sites of recurrence for patients receiving neoadjuvant chemotherapy for gastric cancer. Neoadjuvant chemotherapy followed by surgical resection is the standard treatment for locally advanced gastric cancer in the West, but limited information exists as to timing and patterns of recurrence in this setting.
Methods
Patients with clinical stage 2 or 3 gastric cancer treated with neoadjuvant chemotherapy followed by curative-intent resection between January 2000 and December 2015 were analyzed for 5-year recurrence-free survival (RFS) as well as timing and site of recurrence.
Results
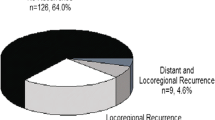
Among 312 identified patients, 121 (38.8%) experienced recurrence during a median follow-up period of 46 months. The overall 5-year RFS rate was 58.9%, with RFS rates of 95.8% for ypT0N0, 81% for ypStage 1, 77.4% for ypStage 2, and 22.9% for ypStage 3. The first site of recurrence was peritoneal for 49.6%, distant (not peritoneal) for 45.5%, and locoregional for 11.6% of the patients. The majority of the recurrences (84.3%) occurred within 2 years. Multivariate analysis showed that ypT4 status was an independent predictor for recurrence within 1 year after surgery (odds ratio, 2.58; 95% confidence interval, 1.10–6.08; p = 0.030).
Conclusions
The majority of the recurrences for patients with clinical stage 2 or 3 gastric cancer who received neoadjuvant chemotherapy and underwent curative resection occurred within 2 years. After neoadjuvant chemotherapy, pathologic T stage was a useful risk predictor for early recurrence.



Similar content being viewed by others
References
Ajani JA, Mansfield PF, Janjan N, et al. Multi-institutional trial of preoperative chemoradiotherapy in patients with potentially resectable gastric carcinoma. J Clin Oncol. 2004;22:2774–80.
Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectablegastroesophageal cancer. N Engl J Med. 2006;355:11–20.
Wang X, Zhao L, Liu H, et al. A phase II study of a modified FOLFOX6 regimen as neoadjuvant chemotherapy for locally advanced gastric cancer. Br J Cancer. 2016;114:1326–33.
Al-Batran SE, Homann N, Pauligk C, et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): a randomised, phase 2/3 trial. Lancet. 2019;393:1948–57.
Coccolini F, Nardi M, Montori G, et al. Neoadjuvant chemotherapy in advanced gastric and esophago-gastric cancer: meta-analysis of randomized trials. Int J Surg. 2018;51:120–7.
Miao ZF, Liu XY, Wang ZN, et al. Effect of neoadjuvant chemotherapy in patients with gastric cancer: a PRISMA-compliant systematic review and meta-analysis. BMC Cancer. 2018;18:118.
Mokadem I, Dijksterhuis WPM, van Putten M, et al. Recurrence after preoperative chemotherapy and surgery for gastric adenocarcinoma: a multicenter study. Gastric Cancer. 2019;22:1263–73.
Ikoma N, Chen HC, Wang X, et al. Patterns of initial recurrence in gastric adenocarcinoma in the era of preoperative therapy. Ann Surg Oncol. 2017;24:2679–87.
Seyfried F, von Rahden BH, Miras AD, et al. Incidence, time course, and independent risk factors for metachronous peritoneal carcinomatosis of gastric origin: a longitudinal experience from a prospectively collected database of 1108 patients. BMC Cancer. 2015;15:73.
Wu CW, Lo SS, Shen KH, et al. Incidence and factors associated with recurrence patterns after intended curative surgery for gastric cancer. World J Surg. 2003;27:153–8.
Spolverato G, Ejaz A, Kim Y, et al. Rates and patterns of recurrence after curative intent resection for gastric cancer: a United States multi-institutional analysis. J Am Coll Surg. 2014;219:664–75.
D’Angelica M, Gonen M, Brennan MF, Turnbull AD, Bains M, Karpeh MS. Patterns of initial recurrence in completely resected gastric adenocarcinoma. Ann Surg. 2004;240:808–16.
Chou HH, Kuo CJ, Hsu JT, et al. Clinicopathologic study of node-negative advanced gastric cancer and analysis of factors predicting its recurrence and prognosis. Am J Surg. 2013;205:623–30.
Deng J, Liang H, Wang D, Sun D, Pan Y, Liu Y. Investigation of the recurrence patterns of gastric cancer following a curative resection. Surg Today. 2011;41:210–5.
Eom BW, Yoon H, Ryu KW, et al. Predictors of timing and patterns of recurrence after curative resection for gastric cancer. Dig Surg. 2010;27:481–6.
Liu D, Lu M, Li J, et al. The patterns and timing of recurrence after curative resection for gastric cancer in China. World J Surg Oncol. 2016;14:305.
Sakar B, Karagol H, Gumus M, et al. Timing of death from tumor recurrence after curative gastrectomy for gastric cancer. Am J Clin Oncol. 2004;27:205–9.
Yoo CH, Noh SH, Shin DW, Choi SH, Min JS. Recurrence following curative resection for gastric carcinoma. Br J Surg. 2000;87:236–42.
Lee JH, Chang KK, Yoon C, Tang LH, Strong VE, Yoon SS. Lauren histologic type is the most important factor associated with pattern of recurrence following resection of gastric adenocarcinoma. Ann Surg. 2018;267:105–13.
Amin MB, Greene FL, Edge SB, et al. The Eighth Edition AJCC Cancer Staging Manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67:93–9.
Chappell R. Competing risk analyses: how are they different and why should you care? Clin Cancer Res. 2012;18:2127–9.
Smyth EC, Fassan M, Cunningham D, et al. Effect of pathologic tumor response and nodal status on survival in the Medical Research Council Adjuvant Gastric Infusional Chemotherapy trial. J Clin Oncol. 2016;34:2721–7.
Harrison LE, Karpeh MS, Brennan MF. Proximal gastric cancers resected via a transabdominal-only approach: Results and comparisons to distal adenocarcinoma of the stomach. Ann Surg. 1997;225:678–83 (discussion 683–675).
Piso P, Werner U, Lang H, Mirena P, Klempnauer J. Proximal versus distal gastric carcinoma: what are the differences? Ann Surg Oncol. 2000;7:520–5.
Higuchi K, Koizumi W, Tanabe S, Saigenji K, Ajani JA. Chemotherapy is more active against proximal than distal gastric carcinoma. Oncology. 2004;66:269–74.
Schwarz RE, Zagala-Nevarez K. Recurrence patterns after radical gastrectomy for gastric cancer: Prognostic factors and implications for postoperative adjuvant therapy. Ann Surg Oncol. 2002;9:394–400.
Ajani JA, D’Amico TA. NCCN Clinical Practice Guidelines in Oncology: Gastric Cancer v 2.2020. Retrieved at https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf. Accessed 30 Dec 2000.
Japanese Gastric Cancer A. Japanese gastric cancer treatment guidelines 2014 (version 4). Gastric Cancer. 2014;2017(20):1–19.
Aurello P, Petrucciani N, Antolino L, Giulitti D, D’Angelo F, Ramacciato G. Follow-up after curative resection for gastric cancer: is it time to tailor it? World J Gastroenterol. 2017;23:3379–87.
Jackson C, Cunningham D, Oliveira J, Group EGW. Gastric cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2009;20(Suppl 4):34–6.
Acknowledgments
This research was supported in part by the NIH/NCI Cancer Center Support Grant P30 CA008748. The authors acknowledge Jessica Moore, MS, staff editor at MSK, for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Dr. Yelena Y. Janjigian has received research funding provided to the institution from Rgenix, Boehringer Ingelheim, Bayer, Genentech/Roche, Bristol-Myers Squibb, Eli Lilly, and Merck, has served on advisory boards for Rgenix, Merck Serono, Bristol-Myers Squibb, Eli Lilly, Pfizer, Bayer, Imugene, Merck, Daiichi-Sankyo, and AstraZeneca. GYK, and has received honoraria and research funding from Merck, Bristol-Myers Squibb, and Pieris and research funding from AstraZeneca, Zymeworks, and Daiichi Sankyo. Dr. David H. Ilson has received research funding from and served on advisory boards for Astellas, Eli Lilly, Pieris, and Taiho, and has served on advisory boards for Astra-Zeneca, Amgen, Bayer, Bristol-Myers Squibb, and Roche. Dr. Steven B. Maron has received research funding from Genentech and travel expenses from Merck and Bayer. The remaining authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nakauchi, M., Vos, E., Tang, L.H. et al. Outcomes of Neoadjuvant Chemotherapy for Clinical Stages 2 and 3 Gastric Cancer Patients: Analysis of Timing and Site of Recurrence. Ann Surg Oncol 28, 4829–4838 (2021). https://doi.org/10.1245/s10434-021-09624-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-09624-5