Abstract
Background
The routine use of external beam radiotherapy (EBRT) is not recommended for parathyroid carcinoma (PC). However, case series have demonstrated a potential benefit in preventing local recurrence with EBRT. We aimed to characterize the patient population treated with EBRT and identify any impact of EBRT on overall survival (OS) in parathyroid carcinoma.
Methods
Patients who underwent surgery for PC from 2004 to 2016 were identified from the National Cancer Database. Clinicopathologic variables and OS were compared between patients based on treatment with EBRT. Multivariable logistic and Cox regression models were performed with propensity scores and inverse-probability-weighting (IPW) adjustment to reduce treatment-selection bias in the OS analysis.
Results
A total of 885 patients met the inclusion criteria, with 126 (14.2%) undergoing EBRT. Demographics were similar between the two cohorts (EBRT vs. no EBRT). However, patients treated with EBRT had a higher frequency of regionally extensive disease, nodal metastases, and residual microscopic disease (all p < 0.05). On multivariable analysis, Black race, regional tumor extension, nodal metastasis, and treatment at an urban facility were independently associated with EBRT. The 5-year OS was 85.3% with a median follow-up of 60.8 months. EBRT was not associated with a difference in OS in crude, multivariable, or IPW models. More importantly, 10.5% of patients with completely resected localized disease (M0, N0 or Nx) underwent EBRT without a benefit in OS (p = 0.183).
Conclusions
EBRT is not associated with any survival benefit in the treatment of PC. Therefore, it may be overutilized, particularly in patients with localized disease and complete surgical resection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
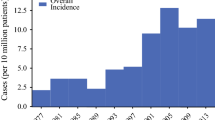
Parathyroid carcinoma (PC) is a rare malignancy, accounting for approximately 11 cases per 10 million people in the United States, and less than 1% of all patients with primary hyperparathyroidism1,2,3,4,5,6 Preoperative diagnosis is difficult, and the majority of patients are diagnosed at the time of surgery or postoperatively on final pathology. Patients with PC typically suffer from multiple recurrences, undergo numerous surgical resections, and eventually develop metastases.7,8,9 The 5-year and 10-year overall survival (OS) rates range from 78 to 86% and 49 to 77%, respectively.3,10 Disease-related mortality is usually secondary to uncontrolled hypercalcemia.14
The mainstay of treatment for PC is surgery. Complete surgical resection with en-bloc excision of the ipsilateral thyroid lobe and any involved structures is generally recommended.15,16 Microscopically negative margins are considered the best chance for cure.17 Chemotherapy has not yet shown benefit, and is generally considered an ineffective therapy in PC.8,14,17 PC is also usually considered radio-resistant, and therefore adjuvant external beam radiotherapy (EBRT) has not traditionally been deemed effective.4,14,17 The routine use of EBRT is not currently recommended by the American Association of Endocrine Surgeons (AAES).16 However, a few very small case series have documented encouraging results for a potential role of adjuvant EBRT in locally invasive PC, when microscopic residual disease is present, or after multiple recurrences.11,18 Therefore, using a large national database, we aimed to characterize the patient population undergoing EBRT, and investigate any potential contribution of EBRT towards improved OS in patients with PC.
Methods
This study was designated as exempt by the Institutional Review Board at Weill Cornell Medicine, given the use of publicly available de-identified data from the American College of Surgeons National Cancer Database (NCDB). The NCDB is the largest cancer registry in the world, representing more than 70% of newly diagnosed cancer cases in the United States, and containing data from over 1500 Commission on Cancer (CoC)-accredited facilities.21 Reported variables include patient demographics, comorbidity severity, pathologic factors, treatments provided, and treating facility characteristics.22 The size of the database and variety of variables reported make the NCDB an ideal database to study rare malignancies and investigate potential prognostic factors.
Patient Population
The 2016 NCDB Participant Use Data File was used for analysis. All patients treated for PC from 2004 to 2016 were identified using the primary site code C75.0 and International Classification of Disease for Oncology, 3rd Edition (ICD-O3) morphologic codes 8000 (neoplasm), 8140 (adenocarcinoma, NOS), 8010 (carcinoma, NOS), and 8290 (oxyphilic adenocarcinoma), and an ICD-O3 behavior code indicating invasion or micro-invasion.23 Patients were excluded if all treatment was performed outside of the reporting facility (n = 7), or if they did not undergo surgery (n = 15). Patients were subsequently divided into two groups based on treatment with EBRT. The use and impact of EBRT was further assessed in two distinct subgroups:1 in patients with definitive PC demonstrated by factors that supplement histologic diagnosis—lymph node metastasis, distant metastasis, or local extension, and2 in patients with localized and completely resected disease.
Dependent and Independent Variables
The primary outcomes of interest were rate of adjuvant EBRT administration among the entire cohort and overall survival of patients in each group. Secondarily, demographics, tumor characteristics, treatment facility data, and surgical outcomes were compared between groups to identify factors associated with use of EBRT. Administration of EBRT was determined based on the radiation treatment summary defined by the NCDB. Chemotherapy included both single and multi-drug therapies, and radical surgery was defined as partial or total removal of the parathyroid with resection in continuity of at least part of another organ. The R system was used to evaluate success of surgical resection, where R0 represents complete microscopic tumor resection, R1 complete macroscopic resection, and R2 incomplete macroscopic resection.24 In terms of patient demographics, median income references the median household income for the patient’s residential zip code based on the 2016 American Community Survey data.
Statistical Analysis
Descriptive statistics were performed using Student’s t test and Chi squared tests for continuous parametric and categorical variables, respectively. Univariable and multivariable logistic regressions were used to identify variables independently associated with EBRT administration. Only variables with p < 0.1 on univariable analysis were included in the multivariable model. Survival analyses by means of Kaplan–Meier estimates with subsequent multivariable Cox proportional regression models were performed. Variables were included in the multivariable Cox regression regardless of result in the univariable analysis in order to adequately assess factors previously reported in the literature to impact OS.1,9,12,13,25 In addition, to help account for the nonrandom treatment assignment of EBRT, inverse probability weighting (IPW) using propensity scores was performed to reduce the effects of confounders. Individual propensities of undergoing EBRT were estimated using a multivariable logistic regression model including the covariates age, comorbidity score, treatment facility county and type, tumor size and extension, nodal and distant metastasis, type of procedure, and success of surgical resection. The Hosmer–Lemeshow test was used to ensure proper fit of the model. The association of EBRT and mortality was then estimated by a Cox regression model using the propensity-score IPW trimmed at the 1st centile.26 The NCDB excludes data on vital status for patients diagnosed in 2016 due to limited follow-up; therefore, these patients were excluded from all survival analyses and propensity score calculations. A p-value of < 0.05 was considered statistically significant. All statistical analyses were performed using Stata software, version 15.1 (Stata Corp. College Station, TX).
Results
Cohort Characteristics and Factors Associated with EBRT
A total of 885 patients underwent surgical resection for parathyroid carcinoma during the study period. Adjuvant EBRT was used in 126 (14.2%) patients. Demographics were similar between the EBRT and no EBRT groups. However, patients who had EBRT were more often treated at urban (p = 0.005) and non-academic centers (p = 0.010) compared with those who did not undergo EBRT. No rural centers administered EBRT (Table 1). Patients who underwent EBRT (vs no EBRT) had a higher rate of regional tumor extension and nodal metastasis, and were less likely to have had an R0 resection (Table 2). Of note, only 3 patients in the entire cohort had an R2 resection, and only 11 had distant metastasis at the time of diagnosis. On multivariable analysis, Black race, treatment at an urban facility (vs metropolitan), regionally extensive disease, pathologically positive lymph nodes, and residual microscopic disease (R1) were independently associated with EBRT (Fig. 1). When EBRT was used, 40–70 Gy was the most common dose (87%), with 8% and 2% of patients getting a higher and lower dose, respectively (3% had an unknown dose).
Analysis of Overall Survival Between All Cohorts
In this patient population, the 5-year and 10-year OS was 85.4% (95% CI 82.4–87.9%) and 67.1% (95% CI 61.7–72.0%), respectively, with a median follow-up of 60.8 months. After accounting for demographics, comorbidities, facility details, and tumor characteristics on multivariable analysis, local median income above the 50th percentile and traveling an intermediate distance to a treatment facility were associated with a better prognosis. Factors that portended a worse OS included older age, Black race, Asian race, a Charlson-Deyo score of at least one, and distant metastasis. Presence of distant metastasis was the most predictive, with a greater than sixfold [HR 6.2 (95% CI 2.5–15.5), p < 0.001] increased risk in mortality (Table 3). Extent of surgical resection was not independently associated with OS.
Impact of EBRT on Overall Survival
No difference in OS was seen with EBRT utilization in the crude unadjusted analysis [HR 1.3 (95% CI 0.9–2.0) p = 0.170] or the multivariable analysis accounting for demographics, comorbidities, facility details, and tumor characteristics [HR 1.3 (95% CI 0.8–2.1) p = 0.320] (Table 3). To reduce treatment-selection bias in the OS analysis and balance confounders between treatment groups, an IPW adjustment according to propensity scores was performed. In a cox regression using IPW, no association of EBRT with OS was identified [HR 1.6 (95% CI 0.9–2.6), p = 0.098], congruent with the aforementioned analyses (Table 4).
Sub-analysis of Patients with Local Extension or Metastasis
Histologically, PC can be difficult to distinguish from atypical parathyroid adenomas or benign neoplasms, and thus pathologic diagnosis often relies on consideration of the overall picture (i.e., intraoperative signs of invasion, or presence of metastasis).16 Therefore, to address this potential limitation in the NCDB, patients with evidence of regional extension, or either nodal or distant metastasis were considered unequivocally to have parathyroid carcinoma (n = 298) in a subset analysis. EBRT was used in 23% (n = 70) of these patients but was not associated with an OS benefit [HR 0.9 (95% CI 0.5–1.7), p = 0.854].
Sub-analysis of Patients with Localized and Completely Resected Disease
Among the entire cohort, the majority of patients (58%, n = 514) had localized (N0 or Nx, and M0), and completely resected (R0) disease. Within this subset, 10.5% (n = 54, 6.1% of entire cohort) were treated with adjuvant EBRT (Fig. 2a). Table 5 compares the demographics, facility details, tumor characteristics, and treatments between the two groups. No rural facilities administered EBRT to these patients, and those who had EBRT were more often treated at non-academic centers. On multivariable analysis, urban facility (vs metropolitan) was predictive of EBRT [OR 2.6 (95% CI 1.2–5.5), p = 0.013], and being treated at an academic/research program (vs community program) was associated with a decreased likelihood of EBRT [OR 0.2 (95% CI 0.05–0.5), p = 0.003]. EBRT use in this population did not confer a survival advantage (log-rank p = 0.085). When controlling for demographics, comorbidities, facility details, and tumor characteristics on univariable and subsequent multivariable analyses, EBRT again was not associated with a prognostic benefit [HR 1.7 (95% CI 0.9–3.1), p = 0.094] (Fig. 2b).
Among the entire cohort, (a) the proportion of patients with localized disease and complete surgical resection, stratified by use of external beam radiation therapy (EBRT) is graphically represented, and (b) independent factors associated with overall survival (OS) in this population (n = 485) is shown. Variables were included in the multivariable model if p < 0.1 on univariable analysis
Discussion
PC is a rare disease characterized by slow progression, frequent recurrences, and difficult-to-control hypercalcemia; consequently, patients often undergo repeated surgical resections. The AAES published guidelines for the definitive management of primary hyperparathyroidism in 2016, recommending against routine EBRT use in PC, but to reserve it for palliative care.16 However this recommendation (recommendation 12-5) was based on low-quality evidence available at the time.16 With the use of a large cancer database, we aimed to characterize the utilization of EBRT and identify any associated survival benefit. We found that over 14% of patients diagnosed with PC undergo EBRT. Factors associated with its use include Black race, treatment at an urban facility (vs metropolitan), and signs of advanced disease—invasive lesions, R1 resection, and nodal metastasis. Despite the considerable use of EBRT, no survival benefit was associated with this therapy, even after accounting for treatment-selection bias using a propensity-score-based analysis. Furthermore, over 10% of patients with localized and completely resected disease also underwent EBRT, despite the lack of a documented survival benefit.
We found a 5-year OS of 85%, consistent with the current literature.3,10,27 Distant metastasis is the most frequently reported marker of poor prognosis, with hazard ratios ranging from 4.7 to 11.6.3,9,13,25 This is similarly seen in our data, with a sixfold increase in mortality independently associated with metastasis. Several prior studies have noted larger tumor size, greater than 3 cm1 or greater than 4 cm,12 and positive lymph nodes13,27 are associated with worse OS. However, neither tumor size (greater or equal to 3 cm) nor pathologically positive lymph nodes impacted survival in this study. Similar conflicting results were found by Asare et al. who identified a 2% increased risk of death with each centimeter increase in tumor size, but neither tumor size categorized >/< 4 cm nor nodal metastasis was associated with OS in their study.12 These discrepancies between studies highlight the difficulty in determining a prognostic staging system for PC.
Surgery is the standard of therapy for PC, although the extent of surgical resection is highly debated. The usual recommendation is complete tumor resection with an ipsilateral hemithyroidectomy with or without central lymphadenectomy regardless of gross tumor involvement.8 Wachter et al. showed that more aggressive surgery (i.e., parathyroidectomy with ipsilateral hemithyroidectomy and central lymph node dissection) compared with parathyroidectomy alone, resulted in an increase in disease-specific survival from 18 to 143 months.2 However, the majority of reports have not shown an association between the extent of surgery and OS—and the data presented here corroborate these previous findings.,1,8,25,28 Thus, the data bring into question the benefit of prophylactic aggressive resection in the absence of gross invasion.
As previously stated, adjuvant EBRT is not recommended as standard therapy for PC. No reports, including this study, have shown a survival advantage with EBRT use.3,12,13,17 Therefore, being able to tease out which patient populations may benefit from EBRT as opposed to those in whom it is unnecessary is important. Tumor recurrence is a staple of this disease, with rates of 33–78% often within the first 2–5 years after initial surgery.8,11,17,28 Four case series have shown improvement in disease recurrence with EBRT compared with historical rates. Munson et al. and Busaidy et al. reported only one local relapse in a total of 10 patients with total follow-up of 20–228 months who were treated with 69 Gy and 60 Gy EBRT, respectively, after non-standardized surgical resections.11,19 In both Chow et al. and Christakis et al.’s studies, all 10 patients underwent the standard oncologic resection with adjuvant EBRT (40 Gy). No recurrences were identified over a follow-up of 1 to over 12 years.18,20 Although these results are statistically underpowered, they are provocative, and potentially identify a benefit of EBRT in the treatment of PC. This may account for the relatively high number of patients undergoing EBRT reported in the NCDB, despite the traditional notion that PC is radio-resistant.
As a consequence of these reports, some have proposed that adjuvant EBRT should be used in patients at higher risk of local relapse—patients with residual disease or tumor present within 2 mm of the resection margin, breakage of the tumor pseudocapsule, or when nodal metastasis is present.4 These guidelines encompass the indications for EBRT use in each of the aforementioned case series. Therefore, it is concerning that the NCDB data shows that more than one tenth of patients with R0, M0, N0/Nx disease underwent adjuvant EBRT, few of whom would meet any of the proposed criteria for EBRT treatment and are even less likely to be palliative. Among these patients, those diagnosed at academic centers were 83% less likely to receive EBRT compared with those treated at community centers, which suggests that the potential overuse of EBRT may be facilitated by the rarity of the disease and, therefore, inconsistent familiarity with the most up-to-date treatment consensus across institutions.
The AAES guidelines recommend reserving EBRT for palliative treatment because of the difficultly a radiated field poses for subsequent surgery. The consensus treatment for PC recurrence is surgery in order to remove tumor bulk and alleviate associated hypercalcemia. Most patients require multiple operations, with each intervention predisposing them to increased risk.14,17 Therefore, EBRT may be a hazard to those patients who recur, and likely should only be considered in patients with refractory disease who are not candidates for re-operation.
Limitations of this study arise from the use of the NCDB, a retrospective database that relies on the accurate coding of a specific set of clinical and oncologic variables. In particular, the histology of PC can vary and requires histologic identification of unequivocal angioinvasion.16 A pathologic review cannot be performed within the NCDB to ensure there is no overlap with atypical parathyroid lesions. Given this NCDB limitation, a sub-analysis was performed in patients considered to definitively have PC—those with regional tumor extension or metastasis (nodal or distant). The OS results for this subgroup were consistent with the main analysis. The NCDB does not have the granularity to identify all indications for EBRT use—for example, the exact proximity of the tumor to the surgical margin, and operative details such as tumor capsule integrity. Patient calcium and parathyroid hormone levels are also unavailable, which may influence prognosis as well as influence management decisions. Data on local recurrence and number of operative resections would also be valuable to augment our analysis of overall survival. Despite these limitations, the size of the NCDB allows for a thorough analysis of an uncommon treatment in a rare disease.
In conclusion, PC is a rare malignancy characterized by moderate OS. Although a few case series have shown a decreased recurrence rate in high risk patients after adjuvant EBRT therapy, an overall survival benefit has yet to be seen, which supports the AAES recommendations against the routine use of EBRT. Nevertheless, we discovered that a significant percentage of patients undergo EBRT despite these standardized guidelines. Therefore, adjuvant EBRT may be overutilized, particularly in patients with localized disease and complete surgical resection. Multi-institutional studies with standardized EBRT regimens are needed to assess whether adjuvant EBRT truly has a role in preventing PC recurrence and to identify which patients should be considered for this therapy.
References
Lo WM, Good ML, Nilubol N, Perrier ND, Patel DT. Tumor size and presence of metastatic disease at diagnosis are associated with disease-specific survival in parathyroid carcinoma. Ann Surg Oncol. 2018;25(9):2535-2540 10.1245/s10434-018-6559-6
Wächter S, Holzer K, Manoharan J, et al. Surgical treatment of parathyroid carcinoma: does the initial en bloc resection improve the prognosis? Chirurg. 2019;90(11):905-912 10.1007/s00104-019-1007-0
Lee PK, Jarosek SL, Virnig BA, Evasovich M, Tuttle TM. Trends in the incidence and treatment of parathyroid cancer in the United States. Cancer. 2007;109(9):1736-1741 10.1002/cncr.22599
Lumachi F, Basso SMM, Basso U. Parathyroid cancer: etiology, clinical presentation and treatment. Anticancer Res. 2006;26(6C): 4803-4807.
Ferraro V, Sgaramella LI, Di Meo G, et al. Current concepts in parathyroid carcinoma: a single centre experience. BMC Endocr Disord. 2019;19(Suppl 1):46 10.1186/s12902-019-0368-1
Shane E. Clinical review 122: Parathyroid carcinoma. J Clin Endocrinol Metab. 2001;86(2):485-493 10.1210/jcem.86.2.7207
Silva-Figueroa AM, Hess KR, Williams MD, et al. Prognostic scoring system to risk stratify parathyroid carcinoma. J Am Coll Surg. 2017 10.1016/j.jamcollsurg.2017.01.060
Al-Kurd A, Mekel M, Mazeh H. Parathyroid carcinoma. Surg Oncol. 2014;23(2):107–114 10.1016/j.suronc.2014.03.005
Asare EA, Silva-Figueroa A, Hess KR, et al. Risk of distant metastasis in parathyroid carcinoma and its effect on survival: a retrospective review from a high-volume center. Ann Surg Oncol. 2019;26(11):3593–3599 10.1245/s10434-019-07451-3
Hundahl SA, Fleming ID, Fremgen AM, Menck HR. Two hundred eighty-six cases of parathyroid carcinoma treated in the a National Cancer Data Base Report. Cancer. 1999;86(3): 538-544 10.1002/(sici)1097-0142(19990801)86:3 < 538::aid-cncr25 > 3.0.co;2-k
Busaidy NL, Jimenez C, Habra MA, et al. Parathyroid carcinoma: a 22-year experience. Head Neck. 2004;26(8):716–726 10.1002/hed.20049
Asare EA, Sturgeon C, Winchester DJ, et al. Parathyroid carcinoma: an update on treatment outcomes and prognostic factors from the national cancer data base (NCDB). Ann Surg Oncol. 2015;22(12):3990–3995 10.1245/s10434-015-4672-3
Harari A, Waring A, Fernandez-Ranvier G, et al. Parathyroid carcinoma: a 43-year outcome and survival analysis. J Clin Endocrinol Metab. 2011;96(12):3679-3686 10.1210/jc.2011-1571
Givi B, Shah JP. Parathyroid carcinoma. Clin Oncol (R Coll Radiol). 2010;22(6):498-507 10.1016/j.clon.2010.04.007
Salcuni AS, Cetani F, Guarnieri V, et al. Parathyroid carcinoma. Best Pract Res Clin Endocrinol Metab. 2018;32(6): 877–889 10.1016/j.beem.2018.11.002
Wilhelm SM, Wang TS, Ruan DT, et al. The American Association of Endocrine Surgeons guidelines for definitive management of primary hyperparathyroidism. JAMA Surg. 2016;151(10):959–968 10.1001/jamasurg.2016.2310
Wei CH, Harari A. Parathyroid carcinoma: update and guidelines for management. Curr Treat Options Oncol. 2012;13(1):11–23 10.1007/s11864-011-0171-3
Chow E, Tsang RW, Brierley JD, Filice S. Parathyroid carcinoma—the Princess Margaret Hospital experience. Int J Radiat Oncol Biol Phys. 1998;41(3): 569–572 10.1016/s0360-3016(98)00098-4
Munson ND, Foote RL, Northcutt RC, et al. Parathyroid carcinoma: is there a role for adjuvant radiation therapy? Cancer. 2003;98(11):2378–2384 10.1002/cncr.11819
Christakis I, Silva AM, Williams MD, et al. Postoperative local-regional radiation therapy in the treatment of parathyroid carcinoma: the MD Anderson experience of 35 years. Pract Radiat Oncol. 2017;7(6):e463–e470 10.1016/j.prro.2017.05.009
National Cancer Database. https://www.facs.org/quality-programs/cancer/ncdb. Accessed December 13, 2018.
https://www.facs.org/-/media/files/quality-programs/cancer/ncdb/puf_data_dictionary.ashx. Accessed December 13, 2018.
International Classification of Diseases for Oncology. http://codes.iarc.fr/codegroup/2. Accessed December 11, 2018.
Amin MB, American Joint Committee On Cancer, Society AC. AJCC cancer staging manual. 8th ed. Chicago IL: Springer; 2017:1032.
Young S, Wu JX, Li N, Yeh MW, Livhits MJ. More extensive surgery may not improve survival over parathyroidectomy alone in parathyroid carcinoma. Ann Surg Oncol. 2016;23(9):2898–2904 10.1245/s10434-016-5256-6
Sato T, Matsuyama Y. Marginal structural models as a tool for standardization. Epidemiology. 2003;14(6):680–686 10.1097/01.EDE.0000081989.82616.7d
Sadler C, Gow KW, Beierle EA, et al. Parathyroid carcinoma in more than 1000 patients: a population-level analysis. Surgery. 2014;156(6):1622–9 10.1016/j.surg.2014.08.069
Hu Y, Bi Y, Cui M, et al. The influence of surgical extent and parafibromin staining on the outcome of parathyroid carcinoma: 20-year experience from a single institute. Endocr Pract. 2019;25(7):634-641 10.4158/EP-2018-0538
Acknowledgments
This project was accepted as a quick-shot oral presentation at the American Association of Endocrine Surgeons annual meeting on April 4-6th, 2020 in Birmingham, AL that was cancelled due to the COVID-19 pandemic.
Funding
No funding was received for this project
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Limberg, J., Stefanova, D., Ullmann, T.M. et al. The Use and Benefit of Adjuvant Radiotherapy in Parathyroid Carcinoma: A National Cancer Database Analysis. Ann Surg Oncol 28, 502–511 (2021). https://doi.org/10.1245/s10434-020-08825-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08825-8