Abstract
Background
Postsurgical recurrence is common in early-stage hepatocellular carcinoma (HCC). Prolonged time to surgery (TTS) may lead to tumor progression. However, the impact of TTS on HCC prognosis is controversial in Western studies and unknown in China. We aim to investigate the impact of TTS on the prognosis of Chinese HCC patients at Barcelona Clinic Liver Cancer (BCLC) stage 0-A who underwent surgery.
Patients and Methods
We retrospectively enrolled 967 BCLC 0-A HCC patients who underwent surgery at three tertiary centers in China. Primary outcomes were recurrence-free survival (RFS) and overall survival (OS). Restricted cubic spline (RCS) was used to select the cutoff value of TTS. Propensity score matching (PSM) was performed to reduce confounding bias, and a time-dependent Cox model was utilized to investigate factors influencing TTS.
Results
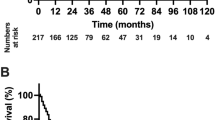
The median TTS of BCLC 0-A HCC patients was 13 days (interquartile range: 10–21 days). For patients with TTS ≤ 70 days, the cutoff value of TTS was 13 days according to RCS. After PSM, corresponding 1-, 3-, and 5-year RFS of the TTS > 13 days and TTS ≤ 13 days groups were 75.6%, 55.3%, 46.4% and 71.2%, 52.3%, 38.8%, respectively (P = 0.103). Corresponding 1-, 3-, and 5-year OS of TTS > 13 days and TTS ≤ 13 days groups were 93.7%, 82.8%, 69.6% and 92.4%, 78.5%, 68.4%, respectively (P = 0.580). Time-dependent Cox analysis revealed that age and tumor size were factors influencing TTS.
Conclusions
Our study suggests that, for patients with TTS ≤ 70 days, prolonged TTS had no impact on BCLC 0-A Chinese HCC patients receiving surgery.



Similar content being viewed by others
References
Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet 2018; 391:1301–14.
Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015; 65:87–108.
Bruix J, Reig M, Sherman M. Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology 2016; 150:835–53.
Yeung YP, Lo CM, Liu CL, et al. Natural history of untreated nonsurgical hepatocellular carcinoma. Am J Gastroenterol 2005; 100:1995–2004.
Bialecki ES, Di Bisceglie AM. Clinical presentation and natural course of hepatocellular carcinoma. Eur J Gastroenterol Hepatol 2005; 17:485–9.
Huo TI, Huang YH, Chiang JH, et al. Survival impact of delayed treatment in patients with hepatocellular carcinoma undergoing locoregional therapy: is there a lead-time bias? Scand J Gastroenterol 2007; 42:485–92.
Singal AG, Waljee AK, Patel N, et al. Therapeutic delays lead to worse survival among patients with hepatocellular carcinoma. J Natl Compr Canc Netw 2013; 11:1101–8.
Lim C, Bhangui P, Salloum C, et al. Impact of time to surgery in the outcome of patients with liver resection for BCLC 0-A stage hepatocellular carcinoma. J Hepatol 2018; 68:100–8.
Xu K, Watanabe-Galloway S, Rochling FA, et al. Surgical delay is associated with improved survival in hepatocellular carcinoma: results of the National Cancer Database. J Gastrointest Surg 2019; 23:933–43.
Park JW, Chen M, Colombo M, et al. Global patterns of hepatocellular carcinoma management from diagnosis to death: the BRIDGE study. Liver Int 2015; 35:2155–66.
Tanaka M, Katayama F, Kato H, et al. Hepatitis B and C virus infection and hepatocellular carcinoma in China: a review of epidemiology and control measures. J Epidemiol 2011; 21:401–16.
Li XJ, Chen MS, Wang ZH, et al. Forgone care among middle aged and elderly with chronic diseases in China: evidence from the China Health and Retirement Longitudinal Study Baseline Survey. BMJ Open 2018; 8:1–10.
Xiong XL, Zhang ZG, Ren J, et al. Impact of universal medical insurance system on the accessibility of medical service supply and affordability of patients in China. PLoS ONE 2018; 13:1–20.
Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018; 67:358–80.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004; 240:205–13.
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien–Dindo classification of surgical complications: five-year experience. Ann Surg 2009; 250:187–96.
Cucchetti A, Vivarelli M, Piscaglia F, et al. Tumor doubling time predicts recurrence after surgery and describes the histological pattern of hepatocellular carcinoma on cirrhosis. J Hepatol 2005; 43:310–16.
Durrleman S, Simon R. Flexible regression models with cubic splines. Stat Med 1989; 8:551–61.
D’Agostino RB, Jr. Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Stat Med 1998; 17:2265–81.
Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol 2006; 163:1149–56.
Austin PC. Some methods of propensity-score matching had superior performance to others: results of an empirical investigation and Monte Carlo simulations. Biometrical J 2009; 51:171–84.
McCaffrey DF, Griffin BA, Almirall D, et al. A tutorial on propensity score estimation for multiple treatments using generalized boosted models. Stat Med 2013; 32:3388–414.
Croome KP, Chudzinski R, Hanto DW. Increasing time delay from presentation until surgical referral for hepatobiliary malignancies. HPB (Oxford) 2010; 12:644–8.
Willatt JM, Hussain HK, Adusumilli S, et al. MR imaging of hepatocellular carcinoma in the cirrhotic liver: challenges and controversies. Radiology 2008; 247:311–30.
Bilimoria KY, Ko CY, Tomlinson JS, et al. Wait times for cancer surgery in the United States: trends and predictors of delays. Ann Surg 2011; 253:779–85.
Shin DW, Cho J, Kim SY, et al. Delay to curative surgery greater than 12 weeks is associated with increased mortality in patients with colorectal and breast cancer but not lung or thyroid cancer. Ann Surg Oncol 2013; 20:2468–76.
Hanna SJ, Muneer A, Khalil KH. The 2-week wait for suspected cancer: time for a rethink? Int J Clin Pract 2005; 59:1334–9.
Stec AA, Coons BJ, Chang SS, et al. Waiting time from initial urological consultation to nephrectomy for renal cell carcinoma: does it affect survival? J Urol 2008; 179:2152–7.
Jooste V, Dejardin O, Bouvier V, et al. Pancreatic cancer: wait times from presentation to treatment and survival in a population-based study. Int J Cancer 2016; 139:1073–80.
Helewa RM, Turner D, Park J, et al. Longer waiting times for patients undergoing colorectal cancer surgery are not associated with decreased survival. J Surg Oncol 2013; 108:378–84.
Acknowledgment
This work was supported by the National Natural Science Foundation of China (grant nos. 81801703, 81770608), the National Science Fund for Distinguished Young Scholars (grant no. 81825013), the Natural Science Foundation of Guangdong Province (grant no. 2018A030310282), Science and Technology Program of Guangzhou, China (grant no. 201704020215), and the National high level talents special support plan— “Ten thousand plan”—Young top-notch talent support program (grant no. not available).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2020_8499_MOESM2_ESM.tiff
Supplementary material 2 The comparison of RFS (Supplementary figure 2a) and OS (Supplementary figure 2b) between TTS > 13 days and TTS ≤ 13 days groups before PSM (TIFF 104 kb)
Rights and permissions
About this article
Cite this article
Wei, M., Chen, S., Li, J. et al. Prognostic Role of Time to Surgery in Hepatocellular Carcinoma at Barcelona Clinic Liver Cancer Stage 0-A. Ann Surg Oncol 27, 3740–3753 (2020). https://doi.org/10.1245/s10434-020-08499-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08499-2