Abstract
Background
Many surgeons advocate the use of neoadjuvant treatment for resectable pancreatic cancer, however little is known about variation in the utilization of neoadjuvant therapy (NAT) at the hospital level.
Methods
The National Cancer Data Base was used to identify patients undergoing resection for pancreatic cancer between 2006 and 2014 at high-volume centers. Hospitals were grouped by NAT utilization using standard deviations (SD) from the mean as follows: high neoadjuvant utilizers (> 2 SDs above the mean, > 40% of patients receiving NAT); medium–high (1–2 SDs, 27–40%), medium (0–1 SD, 14–26%); or low (− 1.1 to 0 SDs, < 14%). Overall survival (OS) was compared across NAT utilization groups.
Results
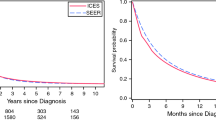
Among 107 high-volume centers, 20,119 patients underwent resection. The proportion of patients receiving NAT varied widely among hospitals, ranging from 0 to 74%, with only five centers using NAT in > 40% of patients. These five hospitals had the longest median OS at 28.9 months, compared with 21.1 months for low neoadjuvant utilizers (p < 0.001). On multivariable analysis, high and medium–high NAT utilization predicted improved OS, with a hazard ratio (HR) of 0.68 (95% confidence interval [CI] 0.56–0.83, p < 0.001) and 0.80 (95% CI 0.68–0.95, p = 0.010), respectively, compared with low utilizers. After excluding patients who underwent NAT, there remained an association of improved OS with high NAT utilization (HR 0.74, 95% CI 0.60–0.93, p = 0.009).
Conclusion
High-volume hospitals that more commonly utilize NAT demonstrated longer survival for all patients treated at those centers. In addition to altering patient selection for surgery, high neoadjuvant utilization may be a marker of institutional factors that contribute to improved outcomes.

Similar content being viewed by others
References
Dimou F, Sineshaw H, Parmar AD, Tamirisa NP, Jemal A, Riall TS. Trends in receipt and timing of multimodality therapy in early-stage pancreatic cancer. J Gastrointest Surg. 2016;20:93–103.
Le A-T, Huang B, Hnoosh D, et al. Effect of complications on oncologic outcomes after pancreaticoduodenectomy for pancreatic cancer. J Surg Res. 2017;214:1–8.
Aloia TE, Lee JE, Vauthey J-N, et al. Delayed recovery after pancreaticoduodenectomy: a major factor impairing the delivery of adjuvant therapy? J Am Coll Surg. 2007;204:347–55.
Matsumoto I, Murakami Y, Shinzeki M, et al. Proposed preoperative risk factors for early recurrence in patients with resectable pancreatic ductal adenocarcinoma after surgical resection: a multi-center retrospective study. Pancreatology. 2015;15:674–80.
Xia BT, Habib DA, Dhar VK, et al. Early recurrence and omission of adjuvant therapy after pancreaticoduodenectomy argue against a surgery-first approach. Ann Surg Oncol. 2016;23:4156–64.
Sugiura T, Uesaka K, Kanemoto H, et al. Serum CA19-9 is a significant predictor among preoperative parameters for early recurrence after resection of pancreatic adenocarcinoma. J Gastrointest Surg. 2012;16:977–85.
Gillen S, Schuster T, Meyer Zum Büschenfelde C, Friess H, Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010;7:e1000267.
Dhir M, Malhotra GK, Sohal DPS, et al. Neoadjuvant treatment of pancreatic adenocarcinoma: a systematic review and meta-analysis of 5520 patients. World J Surg Oncol. 2017;15:183.
National Comprehensive Cancer Network. NCCN Guidelines Version 2.2017 Pancreatic Adenocarcinmoa. https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf (2017). Accessed 13 Jun 2017.
Golcher H, Brunner TB, Witzigmann H, et al. Neoadjuvant chemoradiation therapy with gemcitabine/cisplatin and surgery versus immediate surgery in resectable pancreatic cancer. Strahlentherapie und Onkol. 2015;191:7–16.
Casadei R, Di Marco M, Ricci C, et al. Neoadjuvant chemoradiotherapy and surgery versus surgery alone in resectable pancreatic cancer: a single-center prospective, randomized, controlled trial which failed to achieve accrual targets. J Gastrointest Surg. 2015;19:1802–12.
Palmer DH, Stocken DD, Hewitt H, et al. A randomized phase 2 trial of neoadjuvant chemotherapy in resectable pancreatic cancer: gemcitabine alone versus gemcitabine combined with cisplatin. Ann Surg Oncol. 2007;14:2088–96.
Artinyan A, Anaya DA, McKenzie S, Ellenhorn JDI, Kim J. Neoadjuvant therapy is associated with improved survival in resectable pancreatic adenocarcinoma. Cancer. 2011;117:2044–49.
Parmar AD, Vargas GM, Tamirisa NP, Sheffield KM, Riall TS. Trajectory of care and use of multimodality therapy in older patients with pancreatic adenocarcinoma. Surgery. 2014;156:280–9.
de Geus SWL, Eskander MF, Bliss LA, et al. Neoadjuvant therapy versus upfront surgery for resected pancreatic adenocarcinoma: a nationwide propensity score matched analysis. Surgery. 2017;161:592–601.
Mokdad AA, Minter RM, Zhu H, et al. Neoadjuvant therapy followed by resection versus upfront resection for resectable pancreatic cancer: a propensity score matched analysis. J Clin Oncol. 2017;35:515–22.
Birkmeyer JD, Warshaw AL, Finlayson SR, Grove MR, Tosteson AN. Relationship between hospital volume and late survival after pancreaticoduodenectomy. Surgery. 1999;126:178–83.
Fong Y, Gonen M, Rubin D, Radzyner M, Brennan MF. Long-term survival is superior after resection for cancer in high-volume centers. Ann Surg. 2005;242:540-4-7.
Birkmeyer JD, Siewers AE, Finlayson EVA, et al. Hospital volume and surgical mortality in the United States. N Engl J Med. 2002;346:1128–37.
Ghaferi AA, Birkmeyer JD, Dimick JB. Variation in hospital mortality associated with inpatient surgery. N Engl J Med. 2009;361:1368–75.
StataCorp LP. STATA multilevel mixed-effects reference manual. Release 13. College Station, TX. StataCorp. LLC. https://www.stata.com/manuals13/me.pdf. Accessed 8 Jan 2018.
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC cancer staging manual. 7th ed. New York, NY: Springer; 2010.
Piperdi M, McDade TP, Shim JK, et al. A neoadjuvant strategy for pancreatic adenocarcinoma increases the likelihood of receiving all components of care: lessons from a single-institution database. HPB (Oxford). 2010;12:204–10.
Oettle H, Post S, Neuhaus P, et al. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA. 2007;297:267–77.
Oettle H, Neuhaus P, Hochhaus A, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer. JAMA. 2013;310:1473.
Neoptolemos JP, Stocken DD, Friess H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004;350:1200–10.
Katz MHG, Wang H, Fleming JB, et al. Long-term survival after multidisciplinary management of resected pancreatic adenocarcinoma. Ann Surg Oncol. 2009;16:836–47.
Itchins M, Arena J, Nahm CB, et al. Retrospective cohort analysis of neoadjuvant treatment and survival in resectable and borderline resectable pancreatic ductal adenocarcinoma in a high volume referral centre. Eur J Surg Oncol. 2017;43:1711–17.
Petrelli F, Coinu A, Borgonovo K, et al. FOLFIRINOX-based neoadjuvant therapy in borderline resectable or unresectable pancreatic cancer. Pancreas. 2015;44:515–21.
Pingpank JF, Hoffman JP, Ross EA, et al. Effect of preoperative chemoradiotherapy on surgical margin status of resected adenocarcinoma of the head of the pancreas. J Gastrointest Surg. 2001;5:121–30.
Varadhachary GR, Wolff RA, Crane CH, et al. Preoperative gemcitabine and cisplatin followed by gemcitabine-based chemoradiation for resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3487–95.
Evans DB, Varadhachary GR, Crane CH, et al. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3496–502.
Acknowledgment
AVF’s position as a research fellow is supported by a National Institutes of Health Surgical Oncology Training Grant (T32 CA090217). SMW and DEA are supported by the resources and use of facilities at the William S. Middleton Memorial Veterans Hospital, Madison, WI, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Alexander V. Fisher, Daniel E. Abbott, Manasa Venkatesh, Glen E. Leverson, Stephanie A. Campbell-Flohr, Sean M. Ronnekleiv-Kelly, Caprice C. Greenberg, Emily R. Winslow, and Sharon M. Weber declare no conflicts of interest or financial disclosures.
Rights and permissions
About this article
Cite this article
Fisher, A.V., Abbott, D.E., Venkatesh, M. et al. The Impact of Hospital Neoadjuvant Therapy Utilization on Survival Outcomes for Pancreatic Cancer. Ann Surg Oncol 25, 2661–2668 (2018). https://doi.org/10.1245/s10434-018-6650-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6650-z