Abstract
Background
Muscle depletion is a poor prognostic indicator in colorectal cancer (CRC) patients, but there were no data assessing comparative temporal body composition changes following elective CRC surgery. We examined patient skeletal muscle index trajectories over time after surgery and determined factors that may contribute to those alterations.
Methods
Patients diagnosed with CRC undergoing elective surgical resection between 2006 and 2013 were included in this study. Image analysis of serial computed tomography (CT) scans was used to calculate lumbar skeletal muscle index (LSMI). A multilevel mixed-effect linear regression model was applied using STATA (version 12.0) using the xtmixed command to fit growth curve models (GCM) for LSMI and time.
Results
In 856 patients, a total of 2136 CT images were analyzed; 856 (38.2 %) were preoperative. A quadratic GCM with random intercept and random slope for patients’ LSMI was identified that demonstrated laparoscopy produces a positive change on the LSMI curve [estimate = 0.17 cm2/m2, standard error (SE) 0.06 cm2/m2; p = 0.03], whereas Union for International Cancer Control (UICC) stage III + IV disease contributed to a negative curve change (estimate = −0.19 cm2/m2, SE 0.09 cm2/m2; p = 0.03). Older age (p < 0.01), female gender (p < 0.01), higher American Society of Anesthesiologists (ASA) score (p < 0.01), and altered systemic inflammatory response [SIR] (p = 0.03) were factors significantly associated with lower values of LSMI over time.
Conclusion
In patients undergoing CRC surgery, laparoscopy and the absence of a significantly elevated SIR favored preservation and restoration of skeletal muscle, postoperatively. These emerging data may permit the development of new treatment protocols whereby monitoring and modification of body composition has therapeutic potential.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
It is becoming increasingly clear that a variety of body composition changes occur in cancer patients, and that muscle depletion is a common, albeit in most, occult feature. Muscle depletion is characterized by reduction in muscle size (myopenia) and an increased infiltration by inter- and intramuscular fat, described as myosteatosis.1,2 The incidence of muscle depletion varies from 15 to 70 % for patients treated for CRC,3 and evidence also demonstrates that muscle depletion is associated with poorer outcomes in patients treated for cancer.4,5 For CRC patients treated surgically, myopenia negatively impacts short-term outcomes, including mortality, morbidity, and functional recovery.3 Muscle depletion, estimated from CTBC analysis, has also been found to be a prognostic factor for developing severe toxicity in CRC patients receiving chemotherapy.6 Finally, emerging data suggest that myopenia can be an independent predictor of poorer survival after CRC treatment; however, neither the point of onset of muscle depletion nor the patterns of muscle alterations over time and their precipitants are known. Therefore, there is a need to identify whether body composition changes assessable by surveillance imaging after CRC can be related to specific clinicopathological or treatment factors that, once identified, might allow muscle depletion to be contained or modified.
Growth curve modeling (GCM) is an advanced method for demonstrating within-patient and between-patient variation in outcomes measured across different time points over a follow-up period.7 The GCM approach has allowed researchers to overcome problems in studies that assess comparisons of the intraindividual changes over time. More traditional methods examining changes of time, such as analysis of variance and analysis of covariance, are problematic and are limited, mandating accuracy in equal group sizes, a condition that is very difficult to meet.8,9
Using the GCM approach, we aimed to not only examine how patient body composition, as determined by skeletal muscle index trajectories, varied over time after elective surgery for CRC but also to determine specific factors that may contribute to alterations over time.
Methods
Patient Population
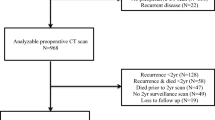
Overall, 1477 consecutive patients undergoing CRC surgery at St Mark’s Hospital, London, between January 2006 and December 2013, were identified from a prospective database. Patients with recorded height data, laboratory blood test data within 4 weeks of staging computed tomography (CT) scan and preoperative staging, and surveillance CT images stored and retrievable in an electronic format suitable for image analysis were included in the study. Exclusions were patients with disease recurrence confirmed preoperatively or at surgery, and emergency operations. All prospectively recorded clinical and pathological data were revalidated from medical and histopathology records. Data collected prospectively during the perioperative period (within 30 days of surgery) included age, sex, body mass index (BMI), American Society of Anesthesiologists (ASA) physical status classification system, tumor site, TNM stage [Union for International Cancer Control (UICC) 5 version] and surgical approach. Laboratory blood test data collected included preoperative neutrophil and lymphocyte counts. The neutrophil to lymphocyte ratio (NLR) was derived as a valid reflection of the host systemic inflammatory response (SIR);10 a high NLR [HNLR] was defined as >3.0.
Body Composition Analysis
Serial CT scan images performed as part of the CRC follow-up protocol were retrieved from digital storage in the picture archiving and communication system (PACS). CT image analysis Slice-O-Matic V4.3 software (Tomovision, Montreal, QC, Canada) was performed as described previously.11 Briefly, total skeletal muscle surface areas (cm2) were evaluated on a single image at the third lumbar vertebrae (L3) using Hounsfield unit (HU) thresholds of −29 to 150 for skeletal muscle, −50 to 150 for visceral adipose tissue, and −190 to −30 for subcutaneous adipose tissues. The sum of skeletal cross-sectional muscle areas was normalized for stature (m2) and reported as lumbar skeletal muscle index (LSMI) [cm2m−2].
Data Analysis
A non-parametric Mann–Whitney U test was used to determine significant differences between the LSMI from baseline demographic and clinicopathological characteristics (a p value < 0.05 was regarded as significant), and a multi-level, mixed-effect GCM was applied using STATA version 12.0 (StataCorp LP, College Station, TX, USA).12 GCM is a special case of random–coefficient models where the coefficient of time varies randomly between subjects. Growth trajectories can take a variety of shapes. A flexible approach to model possible non-linear growth in Y ij is to use a pth degree polynomial function of time \({t}_{\text{ij}} ,_{{}} {Y}_{{{\text{ij}} }} = {\text{b1 }}\left( {\text{constant}} \right) \, + {\text{b2}}.{t}_{\text{ij}} \left( {\text{linear}} \right)_{{}} + {\text{ b3}}.{t}^{ 2}_{\text{ij}} \left( {\text{quadratic}} \right) \, + \ldots + {\text{ b}}.{\text{p }} + { 1t}^{\text{p}}_{\text{ij}} + {\varvec{\upxi}}_{\text{ij}}\).
Using the xtmixed command, we modeled the shape of trajectories of the dependent variable (LSMI) over time and how these trajectories varied due to time- and patient-level covariates. A number of steps were considered in specifying a repeated measures analysis using a GCM approach, as described by Rabe-Hesketh and Skrondal.12 Penalized-likelihood information criteria, such as Akaike’s information criterion (AIC) and the Bayesian information criterion (BIC) were used for model selection. Heteroscedasticity checks were also performed. Heteroscedasticity refers to the circumstance in which the observed variance is independent of the variable mean.13
Results
Study Population
The clinicopathological characteristics of the 856 elective colorectal cancer (CRC) resection cases that fulfilled the selection criteria are provided in Table 1. The median age at operation was 67 years [interquartile range (IQR) 58–76]. Overall, 63.1 % of patients were treated laparoscopically [intention-to-treat] (63.1 % laparoscopic vs. 36.9 % open); 238 (27.8 %) were rectal cancers. The majority of the operations were performed or supervised by one of two consultant colorectal surgeons (RHK and JTJ) who were designated CRC surgeons and were both trained in laparoscopic colorectal surgery, with the laparoscopic technique being standardized between surgeons. These two surgeons performed both open and laparoscopic colorectal resections. Additional cases were performed by three other surgeons using mainly open techniques or by laparoscopy, with mentoring by the two laparoscopic colorectal surgeons. The selection for open or laparoscopic surgery reflected each individual surgeon’s expertise during the study period, and all patients were part of an enhanced recovery after surgery (ERAS) protocol for recovery. A total of 2136 CT images were analyzed, of which 856 (38.2 %) were pretreatment scans.
Body Composition Analysis
The median LSMI was 42.9 (IQR 37.4–49.5) cm2m2, and men had a higher LSMI compared with women (p < 0.001). Elderly patients, patients with a high ASA or high NLR preoperatively, or patients with a colon cancer had significantly lower LSMI median values compared with patients aged <65 years (p < 0.001), ASA I + II (p < 0.001), NLR < 3.0 (p < 0.001), and patients with rectal cancers (p = 0.016), respectively. No differences were noted between the preoperative LSMI and the type of surgical approach (laparoscopic vs. open; p = 0.710) or the UICC stage (p = 0.056). The CT-derived LSMI values and their associations with different clinicopathological variables are summarized in Table 2.
Model Fit Non-linear Growth
Patient LSMI change was non-linear over time when the observed growth trajectories were plotted. As the relationship between LSMI over time was non-linear, we included a quadratic term for time in our model; both time terms in this model were statistically significant (if the time2 term had not been statistically significant, we could have only included a linear term for time in our model). The estimated standard deviation (SD) of the random intercept was 8.53 (95 % confidence interval [CI] 8.09–9.00) and the estimated SD of the error was 3.22 (95 % CI 3.09–3.36). We then included a random slope on time, to permit variability between patients in relation to overall rates of LSMI change. The SD of the random coefficient on time was 0.27 (95 % CI 0.14–0.53), indicating heterogeneity between the rates of change in LSMI. In addition, the estimated SD of the error term decreased from 3.22 to 3.17, indicating a better fit of the model.
Quadratic Growth for Patients’ LSMI that Includes Patient-Level Covariates
Gender
At any given time, we estimated that a female patient’s LSMI was 7.95 cm2/m2 [standard error (SE) 0.58 cm2/m2] less than a male patient’s LSMI. The coefficients of both time and time2 were significant at the 5 % level. Gender did not exert any effect on the slope of the curve trajectory as the estimate for gender*time variable was not significant from the two-stage model formation.
Age
Elderly patients (>65 years of age) had an LSMI, on average, of 3.79 cm2/m2 (SE 0.11 cm2/m2) less than younger patients (< 65years of age). Inclusion of the age*time variable in a two-stage formation model was not significant and the fact that the AIC and BIC values were in favor of the polynomial model; the two-stage formation model was omitted.
Preoperative Systemic Inflammatory Response
Elevated preoperative SIR, expressed as NLR > 3.0 (HNLR), had a significant effect on the LSMI trajectory over time. Patients with HNLR at any given time point had a lower LSMI at an average of −2.28 cm2/m2 (SE 0.79 cm2/m2) compared with the patients with an NLR < 3.0 (low NLR). The two-stage formation model had the best-fit values, but NLR did not have an impact on the slope of the trajectory.
American Society of Anesthesiologists
The two-stage formation model was also the best-fit model when ASA was considered as a patient-level covariate. Patients with a higher ASA score (III + IV) had a significantly lower LSMI (estimate = −2.68 cm2/m2; SE 0.62 cm2/m2) than patients with an ASA score of I or II.
Surgical Approach
At any given time, for patients who underwent laparoscopic resection we estimated that their LSMI was not statistically different than the LSMI of patients who had an open procedure. The coefficients of time2 were significant at the 5 % level. The surgical approach had an impact on the slope of LSMI trajectory as the estimate for surgical approach*time variable was significant from the two-stage model formation. Of interest, patients who underwent a laparoscopic resection had a positive change to the slope of their LSMI trajectory compared with the open approach group (estimate = +0.15 cm2/m2; SE 0.06 cm2/m2). No significant differences were observed between the preoperative BMI (p = 0.61), ASA (p = 0.09), age (p = 0.21), NLR (p = 0.91), and the type of surgical approach.
Tumor Site
The two-stage formation model was the best-fit model when tumor location was considered as the patient-level covariate. Patients who underwent surgery for a rectal tumor had a negative change to the slope of their LSMI trajectory compared with the colon group (estimate = −0.13 cm2/m2; SE 0.05 cm2/m2).
Union for International Cancer Control (UICC) Stage
Patients with a higher stage of disease (UICC stage III + IV) had a negative change to the slope of their LSMI trajectory compared with patients with UICC stage I + II (estimate = −0.20 cm2/m2; SE 0.09 cm2/m2).
Heteroscedasticity
Heteroscedasticity checks were performed for each of the patient-level covariates under investigation, and no evidence of heteroscedasticity was identified.
Figure 1 shows the mean trajectory and 95 % range of patient-specific trajectories for the different patient-level covariates. Table 3 summarizes the maximum likelihood estimates for quadratic models for the patients’ LSMI.
Multivariate Quadratic Growth with Random Intercept and Random Slope for Patients’ LSMI
To adjust for all patient-level covariates and all statistically significant time covariates, interactions were considered for the formulation of the final model. Older age, female gender, high preoperative SIR (HNLR) and higher ASA were significantly associated with lower LSMI values over time. Laparoscopy and a more advanced UICC stage had a significant effect on the slope of the LSMI trajectory. Laparoscopy offered a positive change on the LSMI slope (estimate = 0.17 cm2/m2; SE 0.06 cm2/m2), whereas UICC stage III + IV contributed to a negative slope change (estimate = −0.19 cm2/m2; SE 0.09 cm2/m2). The coefficients of time and time2 were significant at the 5 % level. The log-likelihood and values for AIC and BIC were also improved, suggestive of a model of better fit. Table 4 shows the estimates of the multivariate quadratic growth with random intercept and random slope for patients’ LSMI.
Discussion
This study applied a flexible method for modeling the non-linear and asymmetric relationships between body composition and time for patients treated surgically for CRC, specifically addressing muscle mass as represented by LSMI. Using the multilevel GCM approach, we found that the LSMI–time relation followed a quadratic trajectory over the postoperative follow-up period of up to 60 months. We identified that patients with older age, female gender, high preoperative SIR (reported as HNLR and high ASA) have, on average, a low LSMI over time compared with their opposite groups. We also demonstrated that laparoscopy offered a positive change to the LSMI over time, whereas UICC stage III + IV was associated with a negative change, inferring that muscle mass is augmented after laparoscopy and depleted with higher cancer stages. Loss of skeletal mass due to aging is a well-recognized process, but muscle depletion can also be a consequence of chronic diseases such as cancer.14 The presence of comorbidities and the effect of the sex hormones are recognized factors that contribute to muscle metabolism.15,16 Richards et al. previously demonstrated that CT-derived body composition parameters vary between the two genders, but subgroup analysis of this cohort of patients did not reveal any different LSMI trajectories compared with the whole of the cohort;17 however, the pattern of muscle changes after treatment for cancer has not, to our knowledge, been examined in this way before.
We have previously demonstrated that, in patients undergoing CRC surgery, a low NLR favors maintenance of muscle mass postoperatively. Recent advancements in the investigation of the pathophysiology of skeletal muscle depletion and cachexia in cancer patients have suggested that inflammation could be considered the common link.18 Inflammation plays a vital role in the metabolic and body composition changes in cancer though five key domains: systemic inflammation, central energy balance, control of muscle metabolism/function, control of adipose tissue metabolism/function, and regulation of appetite.19 An ongoing state of low-grade inflammation that involves stimulation of various acute-phase proteins such as C-reactive protein and proinflammatory cytokines that enhance autophagy in skeletal muscle and inhibit the synthesis of myofibrillar proteins, may be the background mechanism of the effect of systemic inflammation on the muscle trajectory postoperatively.19 Our findings further support the assertion that resolution of the SIR is a potential approach to develop more effective therapies against muscle depletion and cancer cachexia.
We identified that laparoscopic resection has a positive impact on restoration of patient muscle mass postoperatively. Multiple randomized trials have confirmed that laparoscopy for CRC produces equivalent oncological outcomes as open surgery, and also produces benefits from decreased complications and hospital stay, decreased postoperative narcotic analgesia use, a faster return of bowel function, and improved cosmesis.20,21 We have now identified that additional benefits include maintenance and restoration of muscle mass. The associated tissue injuries from surgical trauma induce immunologic alterations in the patient that depend on the extent of the injury. Laparoscopy, as opposed to open surgery, reduces the systemic inflammatory changes of surgery and this, along with reduced complications, may be the mechanism for preservation of muscle.22 The relative preservation of health during the first year after laparoscopic surgery may be the mechanism underlying reports of improved cancer outcomes compared with conventional open colorectal resection. Recent work from our group (Malietzis et al., BJS in print) showed that laparoscopy and myopenia were both independent predictors of survival in CRC patients treated surgically when adjusted for confounding factors such as BMI, age, UICC, and visceral adiposity. This finding, combined with the results of this study, may be extrapolated that increased adoption of laparoscopy for CRC surgery may have a positive indirect impact upon cancer survival.
GCM, the statistical methodology used, is an advanced technique to determine individual growth profiles and to address questions of stability over time, with a number of advantages by comparison with other analytical methods. First, it provides a more flexible way to analyse unbalanced data with measurements that are inconsistent over time; second, it allows investigators to analyse both intra- and intersubject differences in the growth parameters (e.g. slopes and intercepts); third, the effects of predictors at higher levels and other predictors on individual growth can flexibly be added in the GCM; and, finally, the GCM approach is more powerful in examining the effects associated with measures over time as it models the covariance matrix (i.e. fitting the true covariance structure to the data), rather than imposing a certain type of structure as is commonly used in traditional univariate and multivariate approaches.7
Limitations of this study include the fact that this was an uncontrolled study despite comprising a relatively large and homogenous data set. Electronic records of CT scans were not available before 2007 as the PACS was introduced in February that year and has contributed to the exclusion from the study of a proportion of the St Mark’s early cohort. BMI and other pathological markers, such as grade of differentiation and lymphovascular invasion, were not included in the analysis as previous work from our group did not identify any significant relationships between these parameters and the presence of muscle depletion in CRC patients.23 Finally, the effect of the postoperative outcomes on the LSMI trajectory, and that of additional treatments such as adjuvant chemotherapy, was not interrogated. To avoid confusion with regard to the impact of the postoperative major morbidity events, we analyzed CT scans requested only for CRC follow-up purposes. The time point of the initiation and duration of the adjuvant chemotherapy regimen made it extremely difficult to account for, in an analysis that focused mainly on preoperative factors and their impact on the muscle trajectory.
Conclusions
In patients undergoing CRC surgery, laparoscopy and the absence of a significantly elevated SIR favors preservation and restoration of muscle mass, postoperatively. The emerging data from our study may permit the development of new treatment protocols whereby monitoring and modifying body composition has therapeutic potential.
References
Fearon K, Evans WJ, Anker SD. Myopenia: a new universal term for muscle wasting. J Cachexia Sarcopenia Muscle. 2011;2(1):1–3.
Miljkovic I, Zmuda JM. Epidemiology of myosteatosis. Curr Opin Clin Nutr Metab Care. 2010;13(3):260–4.
Malietzis G, Aziz O, Bagnall NM, et al. The role of body composition evaluation by computerized tomography in determining colorectal cancer treatment outcomes: a systematic review. Eur J Surg Oncol. 2015;41(2):186–96.
Prado CM, Lieffers JR, McCargar LJ, et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 2008;9(7):629–35.
Martin L, Birdsell L, Macdonald N, et al. Cancer cachexia in the age of obesity: skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol. 2013;31(12):1539–47.
Prado CM, Baracos VE, McCargar LJ, et al. Body composition as an independent determinant of 5-fluorouracil-based chemotherapy toxicity. Clin Cancer Res. 2007;13(11):3264–8.
Shek DT, Ma CM. Longitudinal data analyses using linear mixed models in SPSS: concepts, procedures and illustrations. Sci World J 2011; 11:42–76.
Francis DJ, Fletcher JM, Stuebing KK, et al. Analysis of change: modeling individual growth. J Consult Clin Psychol. 1991;59(1):27–37.
Singer JD, Willett JB. Applied longitudinal data analysis: modeling change and event occurrence. Oxford: Oxford University Press; 2003.
Malietzis G, Giacometti M, Askari A, et al. A preoperative neutrophil to lymphocyte ratio of 3 predicts disease-free survival after curative elective colorectal cancer surgery. Ann Surg. 2014;260(2):287–92.
Mourtzakis M, Prado CM, Lieffers JR, et al. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl Physiol Nutr Metab. 2008;33(5):997–1006.
Rabe-Hesketh S, Skrondal A, Stata Corporation. Multilevel and longitudinal modeling using stata: continuous responses. vol 1, 3rd ed; College Station (TX): Stata Press Publication; 2012.
Brehm MA, Scholtes VA, Dallmeijer AJ, et al. The importance of addressing heteroscedasticity in the reliability analysis of ratio-scaled variables: an example based on walking energy-cost measurements. Dev Med Child Neurol. 2012;54(3):267–73.
Biolo G, Cederholm T, Muscaritoli M. Muscle contractile and metabolic dysfunction is a common feature of sarcopenia of aging and chronic diseases: from sarcopenic obesity to cachexia. Clin Nutr. 2014;33(5):737–48.
Biolo G, Ciocchi B, Stulle M, et al. Calorie restriction accelerates the catabolism of lean body mass during 2 wk of bed rest. Am J Clin Nutr. 2007;86(2):366–72.
Ji L, Meng H, Dong B. Factors associated with poor nutritional status among the oldest-old. Clin Nutr. 2012;31(6):922–6.
Richards CH, Roxburgh CS, MacMillan MT, et al. The relationships between body composition and the systemic inflammatory response in patients with primary operable colorectal cancer. PLoS One. 2012;7(8):e41883.
Seelaender M, Batista M Jr, Lira F, et al. Inflammation in cancer cachexia: to resolve or not to resolve (is that the question?). Clin Nutr. 2012;31(4):562–6.
Tan BH, Ross JA, Kaasa S, et al. Identification of possible genetic polymorphisms involved in cancer cachexia: a systematic review. J Genet. 2011;90(1):165–77.
Kennedy RH, Francis EA, Wharton R, et al. Multicenter randomized controlled trial of conventional versus laparoscopic surgery for colorectal cancer within an enhanced recovery programme: EnROL. J Clin Oncol. 2014;32(17):1804–11.
Vlug MS, Wind J, Hollmann MW, et al. Laparoscopy in combination with fast track multimodal management is the best perioperative strategy in patients undergoing colonic surgery: a randomized clinical trial (LAFA-study). Ann Surg. 2011;254(6):868–75.
Watt DG, Horgan PG, McMillan DC. Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: a systematic review. Surgery. 2015;157(2):362–80.
Malietzis G, Johns N, Al-Hassi HO, et al. Low muscularity and myosteatosis is related to the host systemic inflammatory response in patients undergoing surgery for colorectal cancer. Ann Surg. 2016;263(2):320–5.
Acknowledgment
The authors thank R. Baldwin for retrieving and preparing for analysis the CT images.
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Malietzis, G., Currie, A.C., Johns, N. et al. Skeletal Muscle Changes After Elective Colorectal Cancer Resection: A Longitudinal Study. Ann Surg Oncol 23, 2539–2547 (2016). https://doi.org/10.1245/s10434-016-5188-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5188-1