Abstract
Background
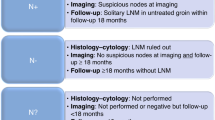
In patients with primary vulvar cancer and bilateral sentinel lymph node (SLN) biopsy, bilateral complete inguino-femoral lymphadenectomy (LAE) is recommended, even in cases with only unilaterally positive SLN by most guidelines. The risk of contralateral non-SLN metastasis is unclear.
Methods
All patients with primary vulvar cancer receiving an SLN dissection with radioactive tracer ± blue dye at the University Medical Center Hamburg-Eppendorf between 2001 and 2013 were retrospectively evaluated. Median follow-up was 33 months.
Results
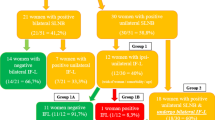
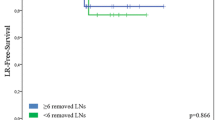
A total of 140 patients were included; 124 with bilateral and 16 with unilateral SLN dissection. A median number of two SLNs (range 1–7) per groin were dissected. Overall, 53 (53/140, 37.9 %) patients received a complete inguino-femoral LAE, 41 of whom (77.4 %) had previously presented with a positive SLN (33 unilaterally, 8 bilaterally). Of the 33 patients with unilaterally positive SLN, 28 (84.9 %) underwent complete bilateral inguino-femoral LAE despite a contralateral negative SLN. Of these patients, none presented a contralateral non-SLN metastasis (0/28, 0 %) in full dissection; however, one developed groin recurrence in the initially SLN-negative, fully dissected groin after 19 months (1/28, 3.6 %).
Conclusion
In case of bilateral SLN biopsy for clinically node-negative disease and only unilaterally positive SLN, the risk for contralateral non-SLN metastases appears to be low. These data support the omission of contralateral LAE to reduce surgical morbidity.

Similar content being viewed by others
References
Mahner S, Jueckstock J, Hilpert F, et al. Adjuvant therapy in lymph node-positive vulvar cancer: The AGO-CaRE-1 Study. J Natl Cancer Inst. 2015;107:dju426.
Van der Zee AG, Oonk MH, De Hullu JA, et al. Sentinel node dissection is safe in the treatment of early-stage vulvar cancer. J Clin Oncol. 2008;26:884–9.
Burke TW, Levenback C, Coleman RL, et al. Surgical therapy of T1 and T2 vulvar carcinoma: further experience with radical wide excision and selective inguinal lymphadenectomy. Gynecol Oncol. 1995;57:215–20.
Katz A, Eifel PJ, Jhingran A, Levenback CF. The role of radiation therapy in preventing regional recurrences of invasive squamous cell carcinoma of the vulva. Int J Radiat Oncol Biol Phys. 2003;57:409–18.
National German Guideline S2k (2015) Diagnosis, therapy, and follow-up care of vulvar cancer and its precursors. AWMF Registry No. 015/059.
Hinten F, van den Einden LC, Hendriks JC, et al. Risk factors for short- and long-term complications after groin surgery in vulvar cancer. Br J Cancer. 2011;105:1279–87.
Magrina JF, Webb MJ, Gaffey TA, Symmonds RE. Stage I squamous cell cancer of the vulva. Am J Obstet Gynecol. 1979;134:453–9.
Iversen T, Aas M. Lymph drainage from the vulva. Gynecol Oncol. 1983;16:179–89.
Hoffman MS, Roberts WS, Finan MA, et al. A comparative study of radical vulvectomy and modified radical vulvectomy for the treatment of invasive squamous cell carcinoma of the vulva. Gynecol Oncol. 1992;45:192–7.
Hacker NF, Berek JS, Lagasse LD, et al. Management of regional lymph nodes and their prognostic influence in vulvar cancer. Obstet Gynecol. 1983;61:408–12.
Andrews SJ, Williams BT, DePriest PD, et al. Therapeutic implications of lymph nodal spread in lateral T1 and T2 squamous cell carcinoma of the vulva. Gynecol Oncol. 1994;55:41–46.
Hacker NF, Berek JS, Lagasse LD, et al. Individualization of treatment for stage I squamous cell vulvar carcinoma. Obstet Gynecol. 1984;63:155–62.
Jeppesen JT, Sell A, Skjoldborg H. Treatment of cancer of the vulva. Acta Obstet Gynecol Scand. 1972;51:101–7.
Rutledge F, Smith JP, Franklin EW. Carcinoma of the vulva. Am J Obstet Gynecol. 1970;106:1117–30.
Krupp PJ, Bohm JW. Lymph gland metastases in invasive squamous cell cancer of the vulva. Am J Obstet Gynecol. 1978;130:943–52.
Gonzalez Bosquet J, Magrina JF, Magtibay PM, et al. Patterns of inguinal groin metastases in squamous cell carcinoma of the vulva. Gynecol Oncol. 2007;105:742–6.
Beller U, Quinn MA, Benedet JL, et al. Carcinoma of the vulva. FIGO 26th annual report on the results of treatment in gynecological cancer. Int J Gynaecol Obstet. 2006;95(Suppl 1):S7–27.
Benedet JL, Bender H, Jones H 3rd, et al. FIGO staging classifications and clinical practice guidelines in the management of gynecologic cancers. FIGO Committee on Gynecologic Oncology. Int J Gynaecol Obstet. 2000;70:209–262.
Stehman FB, Bundy BN, Thomas G, et al. Groin dissection versus groin radiation in carcinoma of the vulva: a Gynecologic Oncology Group study. Int J Radiat Oncol Biol Phys. 1992;24:389–96.
Cormio G, Loizzi V, Carriero C, et al. Groin recurrence in carcinoma of the vulva: management and outcome. Eur J Cancer Care (Engl). 2010;19:302–7.
Stehman FB, Bundy BN, Dvoretsky PM, Creasman WT. Early stage I carcinoma of the vulva treated with ipsilateral superficial inguinal lymphadenectomy and modified radical hemivulvectomy: a prospective study of the Gynecologic Oncology Group. Obstet Gynecol. 1992;79:490–7.
Funding
This study was funded by internal departmental sources.
Conflicts of interest
Linn Woelber, Christine Eulenburg, Donata Grimm, Fabian Trillsch, Inga Bohlmann, Eike Burandt, Jan Dieckmann, Susanne Klutmann, Barbara Schmalfeldt, Sven Mahner, and Katharina Prieske declare that there are no conflicts of interest involved with the presented data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Woelber, L., Eulenburg, C., Grimm, D. et al. The Risk of Contralateral Non-sentinel Metastasis in Patients with Primary Vulvar Cancer and Unilaterally Positive Sentinel Node. Ann Surg Oncol 23, 2508–2514 (2016). https://doi.org/10.1245/s10434-016-5114-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5114-6