Abstract
Background
This study aimed to investigate the association of epidermal growth factor receptor (EGFR) mutation status with treatment outcome for patients with stage 3 non-small cell lung cancer (NSCLC) who had undergone a complete (R0) resection.
Methods
The study identified 3445 NSCLC patients tested for EGFR mutations between September 2001 and December 2011 at the Sun Yat-Sen University Cancer Center. Of these patients, 224 were stage 3 patients who had undergone R0 resections.
Results
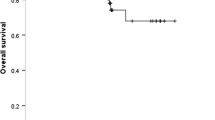
These 224 R0-resected, pathologic stage 3A and 3B patients included 150 patients with wild-type EGFR and 74 patients with EGFR mutations. During a median follow-up period of 42 months (range, 4–133 months), pathologic stage was shown to be the only prognostic factor. The 3-year overall survival (OS) rates did not differ significantly from the OS rates for the wild-type and mutant EGFR groups (62.0 vs 67.2 %; p = 0.789). Multivariate analyses indicated that the patients in the mutant EGFR group with EGFR exon 19 mutations had a better OS rate (73.0 vs 61.1 %; p = 0.026).
Conclusions
Cancer stage remained the significant prognostic factor in R0-resected stage 3 NSCLC patients. The presence of an EGFR mutation is more likely to be a predictive marker for the response to treatment with tyrosine kinase inhibitors. In the EGFR mutant group, the patients with an exon 19 mutation had better 3-year OS rates. These findings might be considered in future study designs.


Similar content being viewed by others
References
Arriagada R, Bergman B, Dunant A, Le Chevalier T, Pignon JP, Vansteenkiste J. Cisplatin-based adjuvant chemotherapy in patients with completely resected non-small cell lung cancer. N Engl J Med. 2004;4:351–60.
Pignon JP, Tribodet H, Scagliotti GV, et al. Lung adjuvant cisplatin evaluation: a pooled analysis by the LACE Collaborative Group. J Clin Oncol. 2008;26:3552–9. doi:10.1200/JCO.2007.13.9030.
Strauss GM, Herndon JN, Maddaus MA, et al. Adjuvant paclitaxel plus carboplatin compared with observation in stage IB non-small cell lung cancer: CALGB9633 with the Cancer and Leukemia Group B, Radiation Therapy Oncology Group, and North Central Cancer Treatment Group study groups. J Clin Oncol. 2008;31:5043–51.
Winton T, Livingston R, Johnson D, et al. Vinorelbine plus cisplatin Vs: observation in resected non-small cell lung cancer. N Engl J Med. 2005;25:2589–97.
Douillard JY, Rosell R, De Lena M, et al. Adjuvant vinorelbine plus cisplatin versus observation in patients with completely resected stage IB-IIIA non-small cell lung cancer (Adjuvant Navelbine International Trialist Association [ANITA]): a randomised controlled trial. Lancet Oncol. 2006;9:719–27.
Detterbeck FC, Boffa DJ, Tanoue LT. The new lung cancer staging system. Chest. 2009;1: 260–71.
Bell DW, Lynch TJ, Haserlat SM, et al. Epidermal growth factor receptor mutations and gene amplification in non-small cell lung cancer: molecular analysis of the IDEAL/INTACT Gefitinib trials. J Clin Oncol. 2005;31: 8081–92.
Eberhard DA, Johnson BE, Amler LC, et al. Mutations in the epidermal growth factor receptor and in KRAS are predictive and prognostic indicators in patients with non-small cell lung cancer treated with chemotherapy alone and in combination with erlotinib. J Clin Oncol. 2005;25: 5900–9.
Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;3:239–46.
Fukuoka M, Wu YL, Thongprasert S, et al. Biomarker analyses and final overall survival results from a phase 3, randomized, open-label, first-line study of gefitinib versus carboplatin/paclitaxel in clinically selected patients with advanced non-small cell lung cancer in Asia (IPASS). J Clin Oncol. 2011;21:2866–74.
Zhou C, Wu YL, Chen G, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011;8:735–42.
Zhang Z, Wang T, Zhang J, et al. Prognostic value of epidermal growth factor receptor mutations in resected non-small cell lung cancer: a systematic review with meta-analysis. Plos One. 2014;8:e106053.
Janjigian YY, Park BJ, Zakowski MF, et al. Impact on disease-free survival of adjuvant erlotinib or gefitinib in patients with resected lung adenocarcinomas that harbor EGFR mutations. J Thorac Oncol. 2011;3:569–75.
Smith IE, O’Brien ME, Talbot DC, et al. Duration of chemotherapy in advanced non-small cell lung cancer: a randomized trial of three versus six courses of mitomycin, vinblastine, and cisplatin. J Clin Oncol. 2001;5:1336–43.
Baggstrom MQ, Stinchcombe TE, Fried DB, Poole C, Hensing TA, Socinski MA. Third-generation chemotherapy agents in the treatment of advanced non-small cell lung cancer: a meta-analysis. J Thorac Oncol. 2007;9:845–53.
Kato M, Onishi H, Matsumoto K, et al. Preoperative chemoradiotherapy using cisplatin plus S-1 can induce downstaging in patients with locally advanced (stage III) non-small cell lung cancer. Anticancer Res. 2012;11:5099–104.
Ramnath N, Dilling TJ, Harris LJ, et al. Treatment of stage III non-small cell lung cancer: diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2013;143(Suppl 5):e314S–40S.
Dahabreh IJ, Linardou H, Siannis F, Kosmidis P, Bafaloukos D, Murray S. Somatic EGFR mutation and gene copy gain as predictive biomarkers for response to tyrosine kinase inhibitors in non-small cell lung cancer. Clin Cancer Res. 2010;1:291–303.
Han SW, Kim TY, Hwang PG, et al. Predictive and prognostic impact of epidermal growth factor receptor mutation in non-small cell lung cancer patients treated with gefitinib. J Clin Oncol. 2005;11:2493–501.
D’Angelo SP, Janjigian YY, Ahye N, et al. Distinct clinical course of EGFR-mutant resected lung cancers: results of testing of 1118 surgical specimens and effects of adjuvant gefitinib and erlotinib. J Thorac Oncol. 2012;12:1815–22.
Kim YT, Seong YW, Jung YJ, et al. The presence of mutations in epidermal growth factor receptor gene is not a prognostic factor for long-term outcome after surgical resection of non-small cell lung cancer. J Thorac Oncol. 2013;2:171–8.
Sun HB, Ou W, Li Y, et al. Epidermal growth factor receptor mutation status and adjuvant chemotherapy in resected advanced non-small cell lung cancer. Clin Lung Cancer. 2013;4:376–82.
Luo YH, Chen YM. Influence of chemotherapy on EGFR mutation status. Transl Lung Cancer Res. 2013;6:442–4.
Disclosure
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Song Ran Liu, Bo Qiu and Hong Yang have contributed equally to this article.
Lan Feng Lin is the co-corresponding author of this article.
Rights and permissions
About this article
Cite this article
Liu, S.R., Qiu, B., Yang, H. et al. Epidermal Growth Factor Receptor Mutation Status and Treatment Outcome for R0-Resected Patients with Stage 3 Non-small Cell Lung Cancer. Ann Surg Oncol 23, 2115–2122 (2016). https://doi.org/10.1245/s10434-015-5076-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-5076-0