Abstract
Background
Liver kinase B1 (LKB1) loss by gene mutation, loss of heterozygosity, and promoter methylation rarely occurs in colorectal cancer. We wondered whether LKB1 loss could be deregulated at the transcriptional level to promote tumor progression and poor outcome in colorectal cancer.
Methods
Mechanistic studies were performed in two each of p53 wild-type (HCT116, LoVo) and p53-mutated (SW480, HT29) colon cancer cells to explore whether LKB1 loss could be deregulated by NKX2-1-mediated p53 pathway. LKB1 and NK2 homeobox 1 (NKX2-1) expressions in colorectal tumors were determined by immunohistochemistry, and the prognostic value of both molecules was assessed by Kaplan–Meier test and Cox regression model.
Results
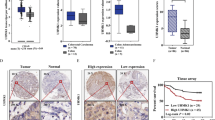
Mechanistically, LKB1 loss at the transcriptional level due to alteration of the NKX2-1-mediated p53 pathway promotes invasiveness in colon cancer cells. The cell invasiveness induced by LKB1 loss was nearly suppressed by mammalian target of rapamycin (mTOR) inhibitor (rapamycin and everolimus) and mTOR/AKT dual inhibitor Palomid 529 (P529). Among patients, low LKB1 tumors exhibited shorter overall survival (OS) and relapse-free survival periods than high LKB1 tumors. The highest hazard ratio value for OS and relapse-free survival was observed in wild-type p53 with low LKB1/low NKX2-1 tumors and in mutated p53 with low LKB1/high NKX2-1 tumors when wild-type p53 with high LKB1/high NKX2-1 and mutated p53 with high LKB1/low NKX2-1 tumors were used as references.
Conclusions
LKB1 loss at the transcriptional level via alteration of the NKX2-1/p53 axis promotes cell invasion, consequently resulting in poor outcome in colorectal cancer patients.



Similar content being viewed by others
References
Hemminki A, Markie D, Tomlinson I, Avizienyte E, Roth S, Loukola A, et al. A serine/threonine kinase gene defective in Peutz-Jeghers syndrome. Nature. 1998;391(6663):184–7.
Wang HH, Xie NN, Li QY, Hu YQ, Ren JL, Guleng B. Exome sequencing revealed novel germline mutations in Chinese Peutz-Jeghers syndrome patients. Dig Dis Sci. 2014;59:64–71.
Gao Y, Zhang FM, Huang S, Wang X, Zhang P, Huang XD, et al. A De Novo mutation of STK11 gene in a Chinese patient with Peutz-Jeghers syndrome. Dig Dis Sci. 2010;55:1032–6.
Zheng B, Pan J, Wang Y, Li M, Lian M, Zheng Y, et al. Analysis of STK11 gene variant in five Chinese patients with Peutz-Jeghers syndrome. Dig Dis Sci. 2013;58:2868–72.
Yoon KA, Ku JL, Choi HS, Heo SC, Jeong SY, Park YJ, et al. Germline mutations of the STK11 gene in Korean Peutz-Jeghers syndrome patients. Br J Cancer. 2000;82:1403–6.
Launonen V, Avizienyte E, Loukola A, Laiho P, Salovaara R, Järvinen H, et al. No evidence of Peutz-Jeghers syndrome gene LKB1 involvement in left-sided colorectal carcinomas. Cancer Res. 2000;60:546–8.
Avizienyte E, Roth S, Loukola A, Hemminki A, Lothe RA, Stenwig A, et al. Somatic mutations in LKB1 are rare in sporadic colorectal and testicular tumors. Cancer Res. 1998;58:2087–90.
Avizienyte E, Loukola A, Roth S, Hemminki A, Tarkkanen M, Salovaara R, et al. LKB1 somatic mutations in sporadic tumors. Am J Pathol. 1999;154:677–81.
Trojan J, Brieger A, Raedle J, Esteller M, Zeuzem S. 5′-CpG island methylation of the LKB1/STK11 promoter and allelic loss at chromosome 19p13.3 in sporadic colorectal cancer. Gut. 2000;47:272–6.
Forster LF, Defres S, Goudie DR, Baty DU, Carey FA. An investigation of the Peutz-Jeghers gene (LKB1) in sporadic breast and colon cancers. J Clin Pathol. 2000;53:791–3.
Saigusa S, Inoue Y, Tanaka K, Toiyama Y, Kawamura M, Okugawa Y, et al. Significant correlation between LKB1 and LGR5 gene expression and the association with poor recurrence-free survival in rectal cancer after preoperative chemoradiotherapy. J Cancer Res Clin Oncol. 2013;139:131–8.
Launonen V. Mutations in the human LKB1/STK11 gene. Hum Mutat. 2005;26:291–7.
Shen Z, Wen XF, Lan F, Shen ZZ, Shao ZM. The tumor suppressor gene LKB1 is associated with prognosis in human breast carcinoma. Clin Cancer Res. 2002;8:2085–90.
Gu Y, Lin S, Li JL, Nakagawa H, Chen Z, Jin B, et al. Altered LKB1/CREB-regulated transcription co-activator (CRTC) signaling axis promotes esophageal cancer cell migration and invasion. Oncogene. 2012;31:469–79.
Ji H, Ramsey MR, Hayes DN, Fan C, McNamara K, Kozlowski P, et al. LKB1 modulates lung cancer differentiation and metastasis. Nature. 2007;448(7155):807–10.
Scott KD, Nath-Sain S, Agnew MD, Marignani PA. LKB1 catalytically deficient mutants enhance cyclin D1 expression. Cancer Res. 2007;67:5622–7.
Feng Y, Wang Y, Wang Z, Fang Z, Li F, Gao Y, et al. The CRTC1-NEDD9 signaling axis mediates lung cancer progression caused by LKB1 loss. Cancer Res. 2012;72:6502–11.
Gao Y, Xiao Q, Ma H, Li L, Liu J, Feng Y, et al. LKB1 inhibits lung cancer progression through lysyl oxidase and extracellular matrix remodeling. Proc Natl Acad Sci USA. 2010;107:18892–7.
Upadhyay S, Liu C, Chatterjee A, Hoque MO, Kim MS, Engles J, et al. LKB1/STK11 suppresses cyclooxygenase-2 induction and cellular invasion through PEA3 in lung cancer. Cancer Res. 2006;66:7870–9.
Takeda H, Miyoshi H, Kojima Y, Oshima M, Taketo MM. Accelerated onsets of gastric hamartomas and hepatic adenomas/carcinomas in Lkb1+/−p53−/− compound mutant mice. Oncogene. 2006;25:1816–20.
Miyoshi H, Nakau M, Ishikawa TO, Seldin MF, Oshima M, Taketo MM. Gastrointestinal hamartomatous polyposis in Lkb1 heterozygous knockout mice. Cancer Res. 2002;62:2261–6.
Duivenvoorden WC, Beatty LK, Lhotak S, Hill B, Mak I, Paulin G, et al. Underexpression of tumour suppressor LKB1 in clear cell renal cell carcinoma is common and confers growth advantage in vitro and in vivo. Br J Cancer. 2013;108:327–33.
Liu W, Monahan KB, Pfefferle AD, Shimamura T, Sorrentino J, Chan KT, et al. LKB1/STK11 inactivation leads to expansion of a prometastatic tumor subpopulation in melanoma. Cancer Cell. 2012;21:751–64.
Morton JP, Jamieson NB, Karim SA, Athineos D, Ridgway RA, Nixon C, et al. LKB1 haploinsufficiency cooperates with Kras to promote pancreatic cancer through suppression of p21-dependent growth arrest. Gastroenterology. 2010;139:586–97, e581–6.
Huang YH, Chen ZK, Huang KT, Li P, He B, Guo X, et al. Decreased expression of LKB1 correlates with poor prognosis in hepatocellular carcinoma patients undergoing hepatectomy. Asian Pac J Cancer Prev. 2013;14:1985–8.
Tsai LH, Chen PM, Cheng YW, Chen CY, Sheu GT, Wu TC, et al. LKB1 loss by alteration of the NKX2-1/p53 pathway promotes tumor malignancy and predicts poor survival and relapse in lung adenocarcinomas. Oncogene. In press.
Ko JL, Chiao MC, Chang SL, Lin P, Lin JC, Sheu GT, et al. A novel p53 mutant retained functional activity in lung carcinomas. DNA Repair (Amst). 2002;1:755–62.
Chen PM, Wu TC, Wang YC, Cheng YW, Sheu GT, Chen CY, et al. Activation of NF-kappaB by SOD2 promotes the aggressiveness of lung adenocarcinoma by modulating NKX2-1-mediated IKKbeta expression. Carcinogenesis. 2013;34:2655–63.
Wu DW, Lee MC, Wang J, Chen CY, Cheng YW, Lee H. DDX3 loss by p53 inactivation promotes tumor malignancy via the MDM2/Slug/E-cadherin pathway and poor patient outcome in non-small-cell lung cancer. Oncogene. 2014;33:1515–26.
Chen TH, Huang CC, Yeh KT, Chang SH, Chang SW, Sung WW, et al. Human papilloma virus 16 E6 oncoprotein associated with p53 inactivation in colorectal cancer. World J Gastroenterol. 2012;18:4051–8.
Esteller M, Avizienyte E, Corn PG, Lothe RA, Baylin SB, Aaltonen LA, et al. Epigenetic inactivation of LKB1 in primary tumors associated with the Peutz-Jeghers syndrome. Oncogene. 2000;19:164–8.
Feng Z, Zhang H, Levine AJ, Jin S. The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci USA. 2005;102:8204–9.
Shaw RJ, Bardeesy N, Manning BD, Lopez L, Kosmatka M, DePinho RA, et al. The LKB1 tumor suppressor negatively regulates mTOR signaling. Cancer Cell. 2004;6:91–9.
Zhong D, Liu X, Khuri FR, Sun SY, Vertino PM, Zhou W. LKB1 is necessary for Akt-mediated phosphorylation of proapoptotic proteins. Cancer Res. 2008;68:7270–7.
Acknowledgment
This work was supported by Grants from the National Science Council (NSC101-2632-B-040-001-MY3), Taiwan, and Taipei Medical University (TMU101-AE1-B10), Taipei, Taiwan.
Disclosures
The authors declare that they have no conflict of none.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tsung-Ying He and Lung-Hung Tsai have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, TY., Tsai, LH., Huang, CC. et al. LKB1 Loss at Transcriptional Level Promotes Tumor Malignancy and Poor Patient Outcomes in Colorectal Cancer. Ann Surg Oncol 21 (Suppl 4), 703–710 (2014). https://doi.org/10.1245/s10434-014-3824-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3824-1