Abstract
Background
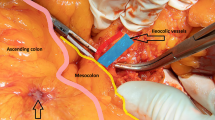
A concept of complete mesocolic excision (CME) and central vascular ligation for colonic cancer has been recently introduced. The aim of this study was to evaluate and compare perioperative and oncologic outcomes after laparoscopic-assisted CME (LCME) and open CME (OCME) for right-sided colon cancers.
Methods
The study group included 128 patients who underwent an LCME and 137 patients who underwent an OCME for right-sided colon cancer between June 2006 and December 2008. The propensity scoring matching for sex, body mass index, tumor location, and pathologic T and TNM stage produced 85 matched pairs.
Results
The median time to soft diet (LCME 6 days vs. OCME 7 days, p < 0.001) and the possible length of stay (7 vs. 13 days, p < 0.001) were significantly shorter in the laparoscopic group. The median operation time (179 vs. 194 minutes, p = 0.862) and number of harvested lymph nodes (27 vs. 28, p = 0.337) were comparable between groups. The morbidity within 30 days after surgery was comparable between the groups (12.9 vs. 24.7 %, p = 0.050). The 5-year overall survival rates of the OCME and LCME groups were 77.8 and 90.3 % (p = 0.028), and the 5-year disease-free survival rates were 71.8 and 83.3 % (p = 0.578), respectively.
Conclusions
Herein, we demonstrated the feasibility and safety of LCME for right-sided colon cancer, and in terms of better short-term outcomes, LCME was more advantageous than OCME. Although LCME for right-sided colon cancer was associated with better 5-year overall survival, compared with an open approach, the long-term oncologic outcomes between the groups were comparable.


Similar content being viewed by others
References
Japanese Society for Cancer of the Colon and Rectum, Daicho Gan K. Japanese classification of colorectal carcinoma. 2nd English ed. Tokyo: Kanehara Shuppan; 2009.
Watanabe T, Itabashi M, Shimada Y, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer. Int J Clin Oncol. 2012;17:1–29.
Martling A, Holm T, Rutqvist LE, et al. Impact of a surgical training programme on rectal cancer outcomes in Stockholm. Br J Surg. 2005;92:225–9.
Wibe A, Moller B, Norstein J, et al. A national strategic change in treatment policy for rectal cancer: implementation of total mesorectal excision as routine treatment in Norway—a national audit. Dis Colon Rectum. 2002;45:857–66.
Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S. Standardized surgery for colonic cancer: complete mesocolic excision and central ligation: technical notes and outcome. Colorectal Dis. 2009;11:354–64; discussion 64–5.
Strasberg SM, Linehan DC, Hawkins WG. The Accordion severity grading system of surgical complications. Ann Surg. 2009;250:177–86.
Li JC, Leung KL, Ng SS, Liu SY, Lee JF, Hon SS. Laparoscopic-assisted versus open resection of right-sided colonic cancer: a prospective randomized controlled trial. Int J Colorectal Dis. 2012;27:95–102.
Gouvas N, Pechlivanides G, Zervakis N, Kafousi M, Xynos E. Complete mesocolic excision in colon cancer surgery: a comparison between open and laparoscopic approach. Colorectal Dis. 2012;14:1357–64.
Pramateftakis MG. Optimizing colonic cancer surgery: high ligation and complete mesocolic excision during right hemicolectomy. Tech Coloproctol. 2010;14(Suppl. 1):S49–51.
Han DP, Lu AG, Feng H, et al. Long-term results of laparoscopy-assisted radical right hemicolectomy with D3 lymphadenectomy: clinical analysis with 177 cases. Int J Colorectal Dis. 2012;28:623–9.
Sun YW, Chi P, Lin HM, et al. [Comparison of efficacy between laparoscopic versus open complete mesocolic excision for colon cancer]. Zhonghua Wei Chang Wai Ke Za Zhi. 2012;15:24–7.
Nakamura T, Onozato W, Mitomi H, et al. Retrospective, matched case-control study comparing the oncologic outcomes between laparoscopic surgery and open surgery in patients with right-sided colon cancer. Surg Today. 2009;39:1040–5.
Sun YW, Chi P, Lin HM, et al. [Risk factors of postoperative chyle leak following complete mesocolic excision for colon cancer]. Zhonghua Wei Chang Wai Ke Za Zhi. 2012;15:328–31.
Wang T, Ye YJ, Han YM, et al. [Short-term efficacy comparison of complete mesocolic excision and traditional colon cancer resection]. Zhonghua Wei Chang Wai Ke Za Zhi. 2013;16:264–7.
Chang GJ, Rodriguez-Bigas MA, Skibber JM, Moyer VA. Lymph node evaluation and survival after curative resection of colon cancer: systematic review. J Natl Cancer Inst. 2007;99:433–41.
Le Voyer TE, Sigurdson ER, Hanlon AL, et al. Colon cancer survival is associated with increasing number of lymph nodes analyzed: a secondary survey of Intergroup trial INT-0089. J Clin Oncol. 2003;21:2912–9.
COLOR Study Group. COLOR: a randomized clinical trial comparing laparoscopic and open resection for colon cancer. Dig Surg. 2000;17:617–22.
Fleshman J, Sargent DJ, Green E, et al. Laparoscopic colectomy for cancer is not inferior to open surgery based on 5-year data from the COST Study Group trial. Ann Surg. 2007;246:655–62; discussion 62–4.
Jayne DG, Guillou PJ, Thorpe H, et al. Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3-year results of the UK MRC CLASICC Trial Group. J Clin Oncol. 2007;25:3061–8.
Lacy AM, Garcia-Valdecasas JC, Delgado S, et al. Laparoscopy-assisted colectomy versus open colectomy for treatment of non-metastatic colon cancer: a randomised trial. Lancet. 2002;359:2224–9.
Lehnert T, Abel U, Kienle P, Hinz U. Open versus laparoscopy assisted colectomy. Lancet. 2003;361:74; author reply 5–6.
Ceulemans R, Henri M, Dutson E, Leroy J, Marescaux J. Open versus laparoscopy assisted colectomy. Lancet. 2003;361:73–4; author reply 5–6.
Bilimoria KY, Bentrem D, Nelson H, et al. Use and outcomes of laparoscopic-assisted colectomy for cancer in the United States. Arch Surg. 2008;143:832–9.
Guerrieri M, Campagnacci R, De Sanctis A, et al. Laparoscopic versus open colectomy for TNM stage III colon cancer: results of a prospective multicenter study in Italy. Surg Today. 2012;42:1071–7.
Kim KY, Hwang DW, Park YK, Lee HS. A single surgeon’s experience with 54 consecutive cases of multivisceral resection for locally advanced primary colorectal cancer: can the laparoscopic approach be performed safely? Surg Endosc. 2012;26:493–500.
Kitano S, Kitajima M, Konishi F, et al. A multicenter study on laparoscopic surgery for colorectal cancer in Japan. Surg Endosc. 2006;20:1348–52.
Kirman I, Cekic V, Poltaratskaia N, et al. Plasma from patients undergoing major open surgery stimulates in vitro tumor growth: lower insulin-like growth factor binding protein 3 levels may, in part, account for this change. Surgery. 2002;132:186–92.
Shirabe K, Matsumata T, Shimada M, et al. A comparison of parenteral hyperalimentation and early enteral feeding regarding systemic immunity after major hepatic resection: the results of a randomized prospective study. Hepatogastroenterology. 1997;44:205–9.
Acknowledgment
This work was supported by the Student’s Association of the Graduate School of Yonsei University funded by the Graduate School of Yonsei University.
Disclosures
The authors have nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bae, S.U., Saklani, A.P., Lim, D.R. et al. Laparoscopic-Assisted Versus Open Complete Mesocolic Excision and Central Vascular Ligation for Right-Sided Colon Cancer. Ann Surg Oncol 21, 2288–2294 (2014). https://doi.org/10.1245/s10434-014-3614-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3614-9