Abstract
Background
Intraductal papillary mucinous neoplasms (IPMNs) are the most common cystic precursor lesions of invasive pancreatic cancer. The recent identification of activating GNAS mutations at codon 201 in IPMNs is a promising target for early detection and therapy. The purpose of this study was to explore clinicopathological correlates of GNAS mutational status in resected IPMNs.
Methods
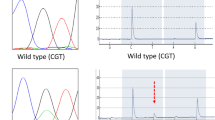
Clinical and pathologic characteristics were retrieved on 54 patients in whom GNAS codon 201 mutational status was previously reported (“historical group”, Wu et al. Sci Transl Med 3:92ra66, 2011). In addition, a separate cohort of 32 patients (validation group) was included. After microdissection and DNA extraction, GNAS status was determined in the validation group by pyrosequencing.
Results
GNAS activating mutations were found in 64 % of the 32 IPMNs included in the validation group, compared with a previously reported prevalence of 57 % in the historical group. Overall, 52 of 86 (61 %) of IPMNs demonstrated GNAS mutations in the two studies combined. Analysis of both groups confirmed that demographic characteristics, tumor location, ductal system involvement, focality, size, grade of dysplasia, presence of an associated cancer, and overall survival were not correlated with GNAS mutational status. Stratified by histological subtype, 100 % of intestinal type IPMNs demonstrated GNAS mutations compared to 51 % of gastric IPMN, 71 % of pancreatobiliary IPMNs, and 0 % of oncocytic IPMNs.
Conclusions
GNAS activating mutations can be reliably detected in IPMNs by pyrosequencing. In terms of clinicopathological parameters, only histological subtype was correlated with mutational frequency, with the intestinal phenotype always associated with GNAS mutations.


Similar content being viewed by others
References
Hruban RH, Takaori K, Klimstra DS, et al. An illustrated consensus on the classification of pancreatic intraepithelial neoplasia and intraductal papillary mucinous neoplasms. Am J Surg Pathol. 2004;28:977–87.
Longnecker DS, Adsay NV, Fernandez-del Castillo C, et al. Histopathological diagnosis of pancreatic intraepithelial neoplasia and intraductal papillary–mucinous neoplasms: interobserver agreement. Pancreas. 2005;31:344–9.
Adsay NV, Merati K, Basturk O, et al. Pathologically and biologically distinct types of epithelium in intraductal papillary mucinous neoplasms: delineation of an “intestinal” pathway of carcinogenesis in the pancreas. Am J Surg Pathol. 2004;28:839–48.
Adsay NV. Cystic neoplasia of the pancreas: pathology and biology. J Gastrointest Surg. 2008;12:401–4.
Maitra A, Fukushima N, Takaori K, Hruban RH. Precursors to invasive pancreatic cancer, Adv Anat Pathol. 2005;12:81–91.
Correa-Gallego C, Ferrone CR, Thayer SP, et al. Incidental pancreatic cysts: do we really know what we are watching? Pancreatology. 2010;10:144–50.
Salvia R, Malleo G, Marchegiani G, et al. Pancreatic resections for cystic neoplasms: from the surgeon’s presumption to the pathologist’s reality. Surgery. 2012;152:S135–42.
Wu J, Matthaei H, Maitra A, et al. Recurrent GNAS mutations define an unexpected pathway for pancreatic cyst development. Sci Transl Med. 2011;3:92ra66.
Wu J, Jiao Y, Dal Molin M, et al. Whole-exome sequencing of neoplastic cysts of the pancreas reveals recurrent mutations in components of ubiquitin-dependent pathways. Proc Natl Acad Sci USA. 2011;108:21188–93.
Matthaei H, Norris AL, Tsiatis AC, et al. Clinicopathological characteristics and molecular analyses of multifocal intraductal papillary mucinous neoplasms of the pancreas. Ann Surg. 2012;255:326–33.
Hruban RH, Klimstra DS, Pitman MB. Tumors of the pancreas. Washington, DC: American Registry of Pathology; 2006.
Furukawa T, Kloppel G, Volkan Adsay N, et al. Classification of types of intraductal papillary–mucinous neoplasm of the pancreas: a consensus study. Virchows Arch. 2005;447:794–9.
R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2012.
Weinstein LS, Liu J, Sakamoto A, Xie T, Chen M. GNAS: normal and abnormal functions (minireview). Endocrinology. 2004;145:5459–64.
Kanda M, Knight S, Topazian M, et al. Mutant GNAS detected in duodenal collections of secretin-stimulated pancreatic juice indicates the presence or emergence of pancreatic cysts. Gut. 2013;62(7):1024–33.
Chen G, Olson MT, O’Neill A, et al. A virtual pyrogram generator to resolve complex pyrosequencing results. J Mol Diagn. 2012;14:149–59.
Furukawa T, Kuboki Y, Tanji E, et al. Whole-exome sequencing uncovers frequent GNAS mutations in intraductal papillary mucinous neoplasms of the pancreas. Sci Rep. 2011;1:161.
Kalfa N, Lumbroso S, Boulle N, et al. Activating mutations of Gsα in kidney cancer. J Urol. 2006;176:891–5.
Collins MT, Sarlis NJ, Merino MJ, et al. Thyroid carcinoma in the McCune-Albright syndrome: contributory role of activating Gs alpha mutations. J Clin Endocrinol Metab. 2003;88:4413–7.
Freda P, Chung W, Matsuoka N, et al. Analysis of GNAS mutations in 60 growth hormone secreting pituitary tumors: correlation with clinical and pathological characteristics and surgical outcome based on highly sensitive GH and IGF-I criteria for remission. Pituitary. 2007;10:275–82.
Nault JC, Fabre M, Couchy G, et al. GNAS-activating mutations define a rare subgroup of inflammatory liver tumors characterized by STAT3 activation. J Hepatol. 2012;56:184–91.
Sjoblom T, Jones S, Wood LD, et al. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314:268–74.
Yamada M, Sekine S, Ogawa R, et al. Frequent activating GNAS mutations in villous adenoma of the colorectum. J Pathol. 2012;228:113–8.
Freda PU, Chung WK, Matsuoka N, et al. Analysis of GNAS mutations in 60 growth hormone secreting pituitary tumors: correlation with clinical and pathological characteristics and surgical outcome based on highly sensitive GH and IGF-I criteria for remission. Pituitary. 2007;10:275–82.
Nishihara E, Amino N, Maekawa K, et al. Prevalence of TSH receptor and Gsalpha mutations in 45 autonomously functioning thyroid nodules in Japan. Endocr J. 2009;56:791–8.
Tominaga E, Tsuda H, Arao T, et al. Amplification of GNAS may be an independent, qualitative, and reproducible biomarker to predict progression-free survival in epithelial ovarian cancer. Gynecol Oncol. 2010;118:160–6.
Acknowledgment
The authors thank Michael Rolfe and the Lustgarten Foundations for their support.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Marco Dal Molin and Hanno Matthaei contributed equally to this article, and both should be considered first author.
Rights and permissions
About this article
Cite this article
Dal Molin, M., Matthaei, H., Wu, J. et al. Clinicopathological Correlates of Activating GNAS Mutations in Intraductal Papillary Mucinous Neoplasm (IPMN) of the Pancreas. Ann Surg Oncol 20, 3802–3808 (2013). https://doi.org/10.1245/s10434-013-3096-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-3096-1