Abstract
Purpose
Ovarian serous carcinoma is an aggressive cancer that often presents with metastatic disease. Although primary tumor and established metastatic foci in the omentum are generally compared to identify proteins involved in drug resistance, we investigated a potential bridge, the malignant cells from ascites, as facilitator of drug resistance and recurrence.
Methods
We evaluated the expression of drug resistance markers P-glycoprotein (P-gp), canalicular multispecific organic anion transporter (MRP2), and lung resistance-related protein (LRP) in malignant cells from ascites and matched omental metastasis from 25 patients with advanced-stage ovarian serous carcinoma who were chemotherapeutic naïve and undergoing initial cytoreductive surgery. Cell viability in vitro, patient response to chemotherapy, and patient survival were correlated with these biomarkers.
Results
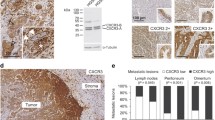
Of the 25 patients evaluated for a correlation of LRP to 1-year recurrence, we correctly predicted the 1-year recurrence of 24 patients based solely on the presence of LRP in ascitic tumor cells (p = 0.01). P-gp and MRP2 were not expressed in malignant cells of ascites or omental metastases. Malignant cells from ascites had higher expression of LRP and were found to be more resistant to carboplatin treatment than cells from omental metastasis (p = 0.00375) by in vitro assay. LRP expression in the malignant cells of ascites correlated with carboplatin resistance (p = 0.001) by in vitro assay and recurrence at 1 year (p = 0.0125).
Conclusions
LRP expression in malignant cells of ascites is a promising marker to predict response to first-line chemotherapy in patients with advanced ovarian serous carcinoma.





Similar content being viewed by others
References
Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Lengyel E. Ovarian cancer development and metastasis. Am J Pathol. 2010;177:1053–64.
Cannistra SA. Cancer of the ovary. N Engl J Med. 2004;351:2519–29.
Goldstein LJ, Pastan I, Gottesman MM. Multidrug resistance in human cancer. Crit Rev Oncol Hematol. 1992;12:243–53.
Bradley G, Ling V. P-glycoprotein, multidrug resistance and tumor progression. Cancer Metastasis Rev. 1994;13:223–33.
Taniguchi K, Wada M, Kohno K, et al. A human canalicular multispecific organic anion transporter (cMOAT) gene is overexpressed in cisplatin-resistant human cancer cell lines with decreased drug accumulation. Cancer Res. 1996;56:4124–9.
Kool M, de Hass M, Scheffer GL, et al. Analysis of expression of cMOAT (MRP2), MRP3, MRP4, and MRP5, homologues of the multidrug resistance-associated protein gene (MRP1), in human cancer cell lines. Cancer Res. 1997;57:3537–47.
Arts HJ, Katsaros D, de Vries EG, et al. Drug resistance-associated markers P-glycoprotein, multidrug resistance-associated protein 1, multidrug resistance-associated protein 2, and lung resistance protein as prognostic factors in ovarian carcinoma. Clin Cancer Res. 1999;5:2798–805.
Izquierdo MA, Scheffer GL, Schroeijers AB, et al. Vault-related resistance to anticancer drugs determined by the expression of the major vault protein LRP. Cytotechnology. 1998;27:137–48.
Kedersha NL, Rome LH, Isolation and characterization of a novel ribonucleoprotein particle: large structures contain a single species of small RNA. J Cell Biol. 1986;103:699–709.
Rome L, Kedersha N, Chugani D, Unlocking vaults: organelles in search of a function. Trends Cell Biol. 1991;1:47–50.
Oda Y, Ohishi Y, Basaki Y, et al. Prognostic implications of the nuclear localization of Y-box-binding protein-1 and CXCR4 expression in ovarian cancer: their correlation with activated Akt, LRP/MVP and P-glycoprotein expression. Cancer Sci. 2007;98:1020–6.
Yakirevich, E, Sabo E, Naroditsky I, et al. Multidrug resistance-related phenotype and apoptosis-related protein expression in ovarian serous carcinomas. Gynecol Oncol. 2006;100:152–9.
Goff BA, Paley PJ, Greer BE, et al. Evaluation of chemoresistance markers in women with epithelial ovarian carcinoma. Gynecol Oncol. 2001;81:18–24.
Goff BA, Ries JA, Els LP, et al. Immunophenotype of ovarian cancer as predictor of clinical outcome: evaluation at primary surgery and second-look procedure. Gynecol Oncol. 1998;70:378–85.
Shield K, Ackland ML, Ahmed N, et al. Multicellular spheroids in ovarian cancer metastases: biology and pathology. Gynecol Oncol. 2009;113:143–8.
Comin CE, Saieva C, Messerini L. h-Caldesmon, calretinin, estrogen receptor, and Ber-EP4: a useful combination of immunohistochemical markers for differentiating epithelioid peritoneal mesothelioma from serous papillary carcinoma of the ovary. Am J Surg Pathol. 2007;31:1139–48.
Ordonez NG. The diagnostic utility of immunohistochemistry and electron microscopy in distinguishing between peritoneal mesotheliomas and serous carcinomas: a comparative study. Mod Pathol. 2006;19:34–48.
Estes JM, Oliver PG, Straughn JM Jr, et al. Efficacy of anti-death receptor 5 (DR5) antibody (TRA-8) against primary human ovarian carcinoma using a novel ex vivo tissue slice model. Gynecol Oncol. 2007;105:291–8.
Frederick PJ, Kendrick JE, Straughn JM Jr, et al. Effect of TRA-8 anti-death receptor 5 antibody in combination with chemotherapy in an ex vivo human ovarian cancer model. Int J Gynecol Cancer. 2009;19:814–9.
Grizzle WE. Tissue resources in the detection and evaluation of markers. In: Srivastava S, editor. Early detection of cancer: molecular markers. Armonk: Futura Publishing Co., 1997:69–76.
Casey RC, Burleson KM, Skubitz KM, et al. Beta 1-integrins regulate the formation and adhesion of ovarian carcinoma multicellular spheroids. Am J Pathol. 2001;159:2071–80.
Burleson KM, Casey RC, Skubitz KM, et al. Ovarian carcinoma ascites spheroids adhere to extracellular matrix components and mesothelial cell monolayers. Gynecol Oncol. 2004;93:170–81.
Burleson KM, Boente MP, Pambuccian SE, et al. Disaggregation and invasion of ovarian carcinoma ascites spheroids. J Transl Med. 2006;4:6.
Inch WR, McCredie JA, Sutherland RM. Growth of nodular carcinomas in rodents compared with multi-cell spheroids in tissue culture. Growth. 1970;34:271–82.
Rustin GJS, Timmers P, Nelstrop A, et al. Comparison of CA-125 and standard definitions of progression of ovarian cancer in the Intergroup trial of cisplatin and paclitaxel versus cisplatin and cyclophosphamide. J Clin Oncol. 2006;24:45–51.
Lee CK, Friedlander M, Brown C, et al. Absence of early decline in CA125 is a poor surrogate for benefit of liposomal doxorubicin and carboplatin treatment in the CALYPSO study. J Natl Cancer Inst. 2011;103:1338–42.
Acknowledgment
This project was supported by grants from the Kaleidoscope of Hope Ovarian Cancer Foundation to LRM and the Lupton Family Fellowship to EHK.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2013_2878_MOESM1_ESM.pdf
Patients clinicopathological features Table 1 Legend: FIGO The International Federation of Gynecology and Obstetrics (PDF 12 kb)
10434_2013_2878_MOESM2_ESM.pdf
Patient’s malignant cells in ascites and omental metastasis were examined for LRP levels using Western blot. Patients 1 and 2 stained positive for LRP by IHC, whereas patient 3 stained negative for LRP by IHC (A = malignant cells in ascites; O = omental metastasis). Therefore, LRP expression by Western blot analysis correlated with LRP expression by IHC. Tumor cells from ascites fluid collected from patients with recurrent cancer also stained positive for LRP (patients A-F) (PDF 41 kb)
Rights and permissions
About this article
Cite this article
Kerr, E.H., Frederick, P.J., Egger, M.E. et al. Lung Resistance-Related Protein (LRP) Expression in Malignant Ascitic Cells as a Prognostic Marker for Advanced Ovarian Serous Carcinoma. Ann Surg Oncol 20, 3059–3065 (2013). https://doi.org/10.1245/s10434-013-2878-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-2878-9