Abstract
Background
The beta2-adrenergic receptor (Beta2-AR) is overexpressed and highly associated with poor prognosis in many malignancies. Nevertheless, the role of Beta2-AR in hepatocellular carcinoma (HCC) has not been thoroughly elucidated. The aim of this study is to investigate the expression of Beta2-AR and its clinicopathological/prognostic value in HCC patients after curative resection.
Materials and Methods
Semiquantitative reverse transcription PCR (RT-PCR) and real-time quantitative PCR (qPCR) were used to measure Beta2-AR RNA expression in 60 pairs of HCC tumors and matched nontumorous tissues. Beta2-AR expression was detected in HCC cell lines by Western blot analysis. Furthermore, we investigated Beta2-AR expression in correlation with the clinicopathological features and analyzed the potential prognostic significance of Beta2-AR in 192 HCC patients by immunohistochemistry (IHC).
Results
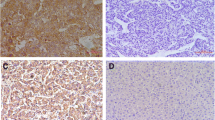
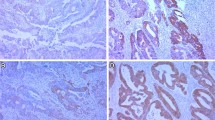
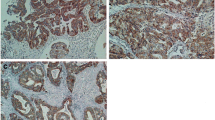
Upregulation of Beta2-AR mRNA was significantly higher in HCC tumor tissues than in their paired nontumorous liver specimens. The expression of Beta2-AR protein was detected in five HCC cell lines. Positive Beta2-AR protein expression was significantly associated with a high α-fetoprotein (AFP) level (P = 0.001), large tumor size (P < 0.001), tumor encapsulation (P = 0.002), vascular invasion (P = 0.004), microsatellite formation (P = 0.002), and poor differentiation (P < 0.001). In univariate and multivariate analyses, Beta2-AR was an excellent predictive factor for both recurrence-free survival and overall survival (OS). Beta2-AR expression status was associated with poor prognosis independent of AFP, tumor-node-metastasis stage and Edmondson stage.
Conclusions
The Beta2-AR is a potential prognostic biomarker for survival and tumor recurrence in HCC patients after curative resection.


Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
McGlynn KA, Tarone RE, El-Serag HB. A comparison of trends in the incidence of hepatocellular carcinoma and intrahepatic cholangiocarcinoma in the United States. Cancer Epidemiol Biomarkers Prev. 2006;15:1198–203.
Gish RG, Porta C, Lazar L, Ruff P, Feld R, Croitoru A, et al. Phase III randomized controlled trial comparing the survival of patients with unresectable hepatocellular carcinoma treated with nolatrexed or doxorubicin. J Clin Oncol. 2007;25:3069–75.
Maki T, Kontula K, Harkonen M. The beta-adrenergic system in man: physiological and pathophysiological response. Regulation of receptor density and functioning. Scand J Clin Lab Invest Suppl. 1990;201:25–43.
Maudsley S, Pierce KL, Zamah AM, Miller WE, Ahn S, Daaka Y, et al. The beta(2)-adrenergic receptor mediates extracellular signal-regulated kinase activation via assembly of a multi-receptor complex with the epidermal growth factor receptor. J Biol Chem. 2000;275:9572–80.
Entschladen F, Drell TL 4th, Lang K, Joseph J, Zaenker KS. Tumour-cell migration, invasion, and metastasis: navigation by neurotransmitters. Lancet Oncol. 2004;5:254–8.
Entschladen F, Drell TLt, Lang K, Joseph J, Zaenker KS. Neurotransmitters and chemokines regulate tumor cell migration: potential for a new pharmacological approach to inhibit invasion and metastasis development. Curr Pharm Des. 2005;11:403–11.
Ramberg H, Eide T, Krobert KA, Levy FO, Dizeyi N, Bjartell AS, et al. Hormonal regulation of beta2-adrenergic receptor level in prostate cancer. Prostate. 2008;68:1133–42.
Yang EV, Kim SJ, Donovan EL, Chen M, Gross AC, Webster Marketon JI, et al. Norepinephrine upregulates VEGF, IL-8, and IL-6 expression in human melanoma tumor cell lines: implications for stress-related enhancement of tumor progression. Brain Behav Immun. 2009;23:267–75.
Shang ZJ, Liu K, Liang de F. Expression of beta2-adrenergic receptor in oral squamous cell carcinoma. J Oral Pathol Med. 2009;38:371–6.
Shi M, Liu D, Duan H, Qian L, Wang L, Niu L, et al. The beta2-adrenergic receptor and Her2 comprise a positive feedback loop in human breast cancer cells. Breast Cancer Res Treat. 2011;125:351–62.
Schuller HM. Beta-adrenergic signaling, a novel target for cancer therapy? Oncotarget. 2010;1:466–9.
Chen L, Yuan YF, Li Y, Chan TH, Zheng BJ, Huang J, et al. Clinical significance of CHD1L in hepatocellular carcinoma and therapeutic potentials of virus-mediated CHD1L depletion. Gut. 2011;60:534–43.
Tung-Ping Poon R, Fan ST, Wong J. Risk factors, prevention, and management of postoperative recurrence after resection of hepatocellular carcinoma. Ann Surg. 2000;232:10–24.
El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557–76.
Dorsam RT, Gutkind JS. G-protein-coupled receptors and cancer. Nat Rev Cancer. 2007;7:79–94.
Takezaki T, Hamajima N, Matsuo K, Tanaka R, Hirai T, Kato T, et al. Association of polymorphisms in the beta-2 and beta-3 adrenoceptor genes with risk of colorectal cancer in Japanese. Int J Clin Oncol. 2001;6:117–22.
Soresi M, Magliarisi C, Campagna P, Leto G, Bonfissuto G, Riili A, et al. Usefulness of alpha-fetoprotein in the diagnosis of hepatocellular carcinoma. Anticancer Res. 2003;23:1747–53.
Johnson PJ. The role of serum alpha-fetoprotein estimation in the diagnosis and management of hepatocellular carcinoma. Clin Liver Dis. 2001;5:145–59.
Lee JS, Chu IS, Heo J, Calvisi DF, Sun Z, Roskams T, et al. Classification and prediction of survival in hepatocellular carcinoma by gene expression profiling. Hepatology. 2004;40:667–76.
Pullar CE, Isseroff RR. The beta 2-adrenergic receptor activates pro-migratory and pro-proliferative pathways in dermal fibroblasts via divergent mechanisms. J Cell Sci. 2006;119:592–602.
Yuan A, Li Z, Li X, Yi S, Wang S, Cai Y, et al. The mitogenic effectors of isoproterenol in human hepatocellular carcinoma cells. Oncol Rep. 2010;23:151–7.
Liu X, Wu WK, Yu L, Li ZJ, Sung JJ, Zhang ST, et al. Epidermal growth factor-induced esophageal cancer cell proliferation requires transactivation of beta-adrenoceptors. J Pharmacol Exp Ther. 2008;326:69–75.
Guo K, Ma Q, Wang L, Hu H, Li J, Zhang D, et al. Norepinephrine-induced invasion by pancreatic cancer cells is inhibited by propranolol. Oncol Rep. 2009;22:825–30.
Stork PJ, Schmitt JM. Crosstalk between cAMP and MAP kinase signaling in the regulation of cell proliferation. Trends Cell Biol. 2002;12:258–66.
Bos JL. Linking rap to cell adhesion. Curr Opin Cell Biol. 2005;17:123–8.
Drell TL 4th, Joseph J, Lang K, Niggemann B, Zaenker KS, Entschladen F. Effects of neurotransmitters on the chemokinesis and chemotaxis of MDA-MB-468 human breast carcinoma cells. Breast Cancer Res Treat. 2003;80:63–70.
Sood AK, Bhatty R, Kamat AA, Landen CN, Han L, Thaker PH, et al. Stress hormone-mediated invasion of ovarian cancer cells. Clin Cancer Res. 2006;12:369–75.
Masur K, Niggemann B, Zanker KS, Entschladen F. Norepinephrine-induced migration of SW 480 colon carcinoma cells is inhibited by beta-blockers. Cancer Res. 2001;61:2866–9.
Lang K, Drell TL 4th, Lindecke A, Niggemann B, Kaltschmidt C, Zaenker KS, et al. Induction of a metastatogenic tumor cell type by neurotransmitters and its pharmacological inhibition by established drugs. Int J Cancer. 2004;112:231–8.
Palm D, Lang K, Niggemann B, Drell TL 4th, Masur K, Zaenker KS, et al. The norepinephrine-driven metastasis development of PC-3 human prostate cancer cells in BALB/c nude mice is inhibited by beta-blockers. Int J Cancer. 2006;118:2744–9.
Powe DG, Voss MJ, Zanker KS, Habashy HO, Green AR, Ellis IO, et al. Beta-blocker drug therapy reduces secondary cancer formation in breast cancer and improves cancer specific survival. Oncotarget. 2010;1:628–38.
Powe DG, Entschladen F. Targeted therapies: using beta-blockers to inhibit breast cancer progression. Nat Rev Clin Oncol. 2011;8:511–2.
Acknowledgment
This work was supported by the National Natural Science Foundation of China (30972849).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dongtai Chen and Wei Xing contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2012_2396_MOESM1_ESM.jpg
Supplementary FIG. 1: Expression of Beta2-AR mRNA in HCC.Overexpression of Beta2-AR was detected in 60 pairs of HCC tumor(T) and matched nontumorous tissues (NT) by q-PCR. (JPG 12.3 mb)
10434_2012_2396_MOESM2_ESM.jpg
Supplementary FIG. 2: Beta2-AR is overexpressed in HCC.Representative images of Beta2-AR expression in HCC tumor (T) and matched nontumorous tissues (NT) detected by immunohistochemistry, Original magnification, ×100. Positive staining (Left) was detected in tumor tissues (T), negative staining (Right) was detected in matched nontumorous tissues (NT), Original magnification, ×200. (JPG 3.44 mb)
Rights and permissions
About this article
Cite this article
Chen, D., Xing, W., Hong, J. et al. The Beta2-Adrenergic Receptor is a Potential Prognostic Biomarker for Human Hepatocellular Carcinoma After Curative Resection. Ann Surg Oncol 19, 3556–3565 (2012). https://doi.org/10.1245/s10434-012-2396-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2396-1