Abstract
Background
The negative impact of anastomotic leakage on cancer-specific survival and recurrence patterns has been recognized in colorectal cancer. In pancreatic cancer, pancreatic fistula (PF) is a serious morbidity, but its negative effect on long-term outcome remains to be elucidated. The aim of this study was to determine the impact of PF on pancreatic cancer recurrence.
Methods
The medical records of 184 patients with curative pancreatectomy for pancreatic cancer were reviewed. PF was scored on the basis of the International Study Group of Pancreatic Fistula classification. Overall and disease-free survivals and recurrence patterns were analyzed. Grade A PF was excluded because the negative effects can be negligible.
Results
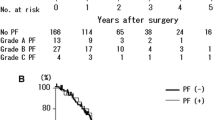
PF occurred in 51 of the 184 patients (27.7%). The mortality related to PF was 0.5% (1 of 184). PF was an independent risk factor for peritoneal recurrence (hazard ratio 3.974; 95% confidence interval 1.345–11.737; P = 0.013). According to the analysis of disease-free survival in patients with peritoneal recurrence, time to recurrence was shorter and the survival rate was worse in patients with PF than in those without PF (5.6 vs. 8.2 months; 6-month survival, 40 vs. 71%; 1-year survival, 7 vs. 19%; P = 0.053). PF was an independent prognostic factor after multivariate analysis (hazard ratio 3.257; 95% confidence interval 1.201–8.828; P = 0.020).
Conclusions
PF was statistically significantly related to peritoneal recurrence, and patients with PF developed peritoneal recurrence earlier than those without PF. With regard to the development of peritoneal recurrence, PF may be considered to be a negative prognostic factor.


Similar content being viewed by others
References
Aranha GV, Aaron JM, Shoup M, Pickleman J. Current management of pancreatic fistula after pancreaticoduodenectomy. Surgery. 2006;140:561–8.
Bassi C, Butturini G, Molinari E, et al. Pancreatic fistula rate after pancreatic resection. The importance of definitions. Dig Surg. 2004;21:54–9.
Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 2005;138:8–13.
Haddad LB, Scatton O, Randone B, et al. Pancreatic fistula after pancreaticoduodenectomy: the conservative treatment of choice. HPB (Oxford). 2009;11:203–9.
Pedrazzoli S, Liessi G, Pasquali C, Ragazzi R, Berselli M, Sperti C. Postoperative pancreatic fistulas: preventing severe complications and reducing reoperation and mortality rate. Ann Surg. 2009;249:97–104.
Vin Y, Sima CS, Getrajdman GI, et al. Management and outcomes of postpancreatectomy fistula, leak, and abscess: results of 908 patients resected at a single institution between 2000 and 2005. J Am Coll Surg. 2008;207:490–8.
Goh BK, Tan YM, Chung YF, et al. Critical appraisal of 232 consecutive distal pancreatectomies with emphasis on risk factors, outcome, and management of the postoperative pancreatic fistula: a 21-year experience at a single institution. Arch Surg. 2008;143:956–65.
Lai EC, Lau SH, Lau WY. Measures to prevent pancreatic fistula after pancreatoduodenectomy: a comprehensive review. Arch Surg. 2009;144:1074–80.
Kanda M, Fujii T, Kodera Y, Nagai S, Takeda S, Nakao A. Nutritional predictors of postoperative outcome in pancreatic cancer. Br J Surg. 2011;98:268–74.
Saito T, Tsuchiya T, Kenjo A, et al. Successful treatment of pseudoaneurysms of celiac and superior mesenteric arteries by combined endovascular and surgical approach. J Hepatobiliary Pancreat Surg. 2008;15:444–8.
Sugimoto H, Kaneko T, Ishiguchi T, et al. Delayed rupture of a pseudoaneurysm following pancreatoduodenectomy: report of a case. Surg Today. 2001;31:932–5.
Yamashita Y, Taketomi A, Fukuzawa K, et al. Risk factors for and management of delayed intraperitoneal hemorrhage after pancreatic and biliary surgery. Am J Surg. 2007;193:454–9.
Yekebas EF, Wolfram L, Cataldegirmen G, et al. Postpancreatectomy hemorrhage: diagnosis and treatment: an analysis in 1669 consecutive pancreatic resections. Ann Surg. 2007;246:269–80.
Conlon KC, Labow D, Leung D, et al. Prospective randomized clinical trial of the value of intraperitoneal drainage after pancreatic resection. Ann Surg. 2001;234:487–93.
Marra F, Steffen T, Kalak N, et al. Anastomotic leakage as a risk factor for the long-term outcome after curative resection of colon cancer. Eur J Surg Oncol. 2009;35:1060–4.
Law WL, Choi HK, Lee YM, Ho JW, Seto CL. Anastomotic leakage is associated with poor long-term outcome in patients after curative colorectal resection for malignancy. J Gastrointest Surg. 2007;11:8–15.
McArdle CS, McMillan DC, Hole DJ. Impact of anastomotic leakage on long-term survival of patients undergoing curative resection for colorectal cancer. Br J Surg. 2005;92:1150–4.
Walker KG, Bell SW, Rickard MJ, et al. Anastomotic leakage is predictive of diminished survival after potentially curative resection for colorectal cancer. Ann Surg. 2004;240:255–9.
den Dulk M, Marijnen CA, Collette L, et al. Multicentre analysis of oncological and survival outcomes following anastomotic leakage after rectal cancer surgery. Br J Surg. 2009;96:1066–75.
Bell SW, Walker KG, Rickard MJ, et al. Anastomotic leakage after curative anterior resection results in a higher prevalence of local recurrence. Br J Surg. 2003;90:1261–6.
McMillan DC, Canna K, McArdle CS. Systemic inflammatory response predicts survival following curative resection of colorectal cancer. Br J Surg. 2003;90:215–19.
Ausania F, Cook N, Jamieson N, Huguet E, Jah A, Praseedom R. Impact of pancreatic leaks on survival following pancreaticoduodenectomy. JOP. 2010;11:226–9.
Sobin L, Gospodarowicz M, Wittekind C. TNM classification of malignant tumors, 7th ed. New York: Wiley; 2009.
Nakao A, Takeda S, Inoue S, et al. Indications and techniques of extended resection for pancreatic cancer. World J Surg. 2006;30:976–82.
Ishikawa O, Wada H, Ohigashi H, et al. Postoperative cytology for drained fluid from the pancreatic bed after “curative” resection of pancreatic cancers: does it predict both the patient’s prognosis and the site of cancer recurrence? Ann Surg. 2003;238:103–10.
Gertsch P, Baer HU, Kraft R, Maddern GJ, Altermatt HJ. Malignant cells are collected on circular staplers. Dis Colon Rectum. 1992;35:238–41.
Keynes WM. Implantation from the bowel lumen in cancer of the large intestine. Ann Surg. 1961;153:357–64.
DerHagopian RP. Inflammatory oncotaxis. JAMA. 1978;240:374–5.
Hirai T, Matsumoto H, Yamashita K, et al. Surgical oncotaxis—excessive surgical stress and postoperative complications contribute to enhancing tumor metastasis, resulting in a poor prognosis for cancer patients. Ann Thorac Cardiovasc Surg. 2005;11:4–6.
Shine T, Wallack MK. Inflammatory oncotaxis after testing the skin of the cancer patient. Cancer. 1981;47:1325–8.
Valentini V, Calvo F, Reni M, et al. Intra-operative radiotherapy (IORT) in pancreatic cancer: joint analysis of the ISIORT–Europe experience. Radiother Oncol. 2009;91:54–9.
Halloran CM, Ghaneh P, Bosonnet L, Hartley MN, Sutton R, Neoptolemos JP. Complications of pancreatic cancer resection. Dig Surg. 2002;19:138–46.
Sato N, Yamaguchi K, Shimizu S, et al. Coil embolization of bleeding visceral pseudoaneurysms following pancreatectomy: the importance of early angiography. Arch Surg. 1998;133:1099–102.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagai, S., Fujii, T., Kodera, Y. et al. Recurrence Pattern and Prognosis of Pancreatic Cancer After Pancreatic Fistula. Ann Surg Oncol 18, 2329–2337 (2011). https://doi.org/10.1245/s10434-011-1604-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-011-1604-8