Abstract
Background
Operative management of pancreatic ductal adenocarcinoma (PDAC) is complicated by several key decisions during the procedure. Identification of metastatic disease at the outset and, when none is found, complete (R0) resection of primary tumor are key to optimizing clinical outcomes. The use of tumor-targeted molecular imaging, based on photoacoustic and fluorescence optical imaging, can provide crucial information to the surgeon. The first-in-human use of multimodality molecular imaging for intraoperative detection of pancreatic cancer is reported using cetuximab-IRDye800, a near-infrared fluorescent agent that binds to epidermal growth factor receptor.
Methods
A dose-escalation study was performed to assess safety and feasibility of targeting and identifying PDAC in a tumor-specific manner using cetuximab-IRDye800 in patients undergoing surgical resection for pancreatic cancer. Patients received a loading dose of 100 mg of unlabeled cetuximab before infusion of cetuximab-IRDye800 (50 mg or 100 mg). Multi-instrument fluorescence imaging was performed throughout the surgery in addition to fluorescence and photoacoustic imaging ex vivo.
Results
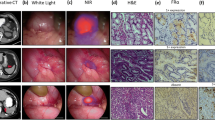
Seven patients with resectable pancreatic masses suspected to be PDAC were enrolled in this study. Fluorescence imaging successfully identified tumor with a significantly higher mean fluorescence intensity in the tumor (0.09 ± 0.06) versus surrounding normal pancreatic tissue (0.02 ± 0.01), and pancreatitis (0.04 ± 0.01; p < 0.001), with a sensitivity of 96.1% and specificity of 67.0%. The mean photoacoustic signal in the tumor site was 3.7-fold higher than surrounding tissue.
Conclusions
The safety and feasibilty of intraoperative, tumor-specific detection of PDAC using cetuximab-IRDye800 with multimodal molecular imaging of the primary tumor and metastases was demonstrated.




Similar content being viewed by others
References
Christians KK, et al. Survival of patients with resectable pancreatic cancer who received neoadjuvant therapy. Surgery. 2016;159(3):893–900.
Roeder F. Neoadjuvant radiotherapeutic strategies in pancreatic cancer. World J Gastrointest Oncol. 2016;8(2):186–97.
Bassi C, et al. Value of regional lymphadenectomy in pancreatic cancer. HPB (Oxford). 2005;7(2):87–92.
Bachmann J, et al. Pancreatic resection for pancreatic cancer. HPB (Oxford). 2006;8(5):346–51.
Tummala P, Howard T, Agarwal B. Dramatic survival benefit related to R0 resection of pancreatic adenocarcinoma in patients with tumor ≤ 25 mm in size and ≤ 1 involved lymph nodes. Clin Transl Gastroenterol. 2013;4:e33.
Verbeke CS, Gladhaug IP. Resection margin involvement and tumour origin in pancreatic head cancer. Br J Surg. 2012;99(8):1036–49.
Zhang JF, et al. Influence of perineural invasion on survival and recurrence in patients with resected pancreatic cancer. Asian Pac J Cancer Prev. 2013;14(9):5133–9.
Zackrisson S, van de Ven SM, Gambhir SS. Light in and sound out: emerging translational strategies for photoacoustic imaging. Cancer Res. 2014;74(4):979–1004.
Vahrmeijer AL, et al. Image-guided cancer surgery using near-infrared fluorescence. Nat Rev Clin Oncol. 2013;10(9):507–18.
Hutteman M, et al. Clinical translation of ex vivo sentinel lymph node mapping for colorectal cancer using invisible near-infrared fluorescence light. Ann Surg Oncol. 2011;18(4):1006–14.
Rosenthal EL, et al. Safety and tumor-specificity of cetuximab-IRDye800 for surgical navigation in head and neck cancer. Clin Cancer Res. 2015;21(16):3658–66.
Bloomston, M, et al. Epidermal growth factor receptor expression in pancreatic carcinoma using tissue microarray technique. Dig Surg. 2006;23(1-2):74–9.
Uegaki K, et al. Clinicopathological significance of epidermal growth factor and its receptor in human pancreatic cancer. Anticancer Res. 1997;17(5B):3841–7.
de Geus SW, et al. Selecting tumor-specific molecular targets in pancreatic adenocarcinoma: paving the way for image-guided pancreatic surgery. Mol Imaging Biol. 2016;18(6):807–19.
Tummers WS, Boonstra MC, Prevoo HA, et al. Selection of optimal molecular targets for tumor-specific imaging in pancreatic ductal adenocarcinoma. Oncotarget. 2017;8(34):56816–28.
Moore LS, et al. Effects of an unlabeled loading dose on tumor-specific uptake of a fluorescently labeled antibody for optical surgical navigation. Mol Imaging Biol. 2016;19(4):610–16.
Zinn KR, et al. IND-directed safety and biodistribution study of intravenously injected cetuximab-IRDye800 in cynomolgus macaques. Mol Imaging Biol. 2015;17(1):49–57.
Kothapalli SR, et al. Deep tissue photoacoustic imaging using a miniaturized 2-D capacitive micromachined ultrasonic transducer array. IEEE Trans Biomed Eng. 2012;59(5):1199–204.
Deeken JF, et al. Evaluation of the relationship between cetuximab therapy and corrected QT interval changes in patients with advanced malignancies from solid tumors. Cancer Chemother Pharmacol. 2013;71(6):1473–83.
Administration, U.S.F.a.D. ERBITUX® (cetuximab) injection, for intravenous infusion—label. 2004 2012 [cited 2017 July 27].
Kothapalli SR. Human prostate imaging using a novel integrated transrectal ultrasound and photoacoustic instrument. Nature Commun. 2017.
Rosenthal E, et al. Sensitivity and specificity of Cetuximab-IRDye800CW to identify regional metastatic disease in head and neck cancer. Clin Cancer Res. 2017;23(16):4744–52.
Buchler P, Müller M, AlKhatib J, Buchler MW, Survival benefit of extended resection in pancreatic cancer. Am J Surg. 2007;194:S120–6.
Gerritsen A, et al. Preoperative characteristics of patients with presumed pancreatic cancer but ultimately benign disease: a multicenter series of 344 pancreatoduodenectomies. Ann Surg Oncol. 2014;21(12):3999–4006.
Cohen R, et al. Inert coupling of IRDye800CW to monoclonal antibodies for clinical optical imaging of tumor targets. EJNMMI Res. 2011;1(1):31.
Philip PA. Targeted therapies for pancreatic cancer. Gastrointest Cancer Res. 2008;2(4 Suppl):S16–9.
Heinemann V, et al. Meta-analysis of randomized trials: evaluation of benefit from gemcitabine-based combination chemotherapy applied in advanced pancreatic cancer. BMC Cancer. 2008;8:82.
Von Hoff DD, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369(18):1691–703.
Moore MJ, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007;25(15):1960–6.
Mitry E, et al. Safety and activity of masitinib in combination with gemcitabine in patients with advanced pancreatic cancer. Cancer Chemother Pharmacol. 2010;66(2):395–403.
Adiseshaiah PP, et al. Nanomedicine strategies to overcome the pathophysiological barriers of pancreatic cancer. Nat Rev Clin Oncol. 2016;13(12):750–65.
Acknowledgment
The authors acknowledge support from Stanford Cancer Institute Translational Research Grant, and Intuitive Surgical Clinical Robotics Research Grant. Tummers WS contribution to this work was supported in part by Michaël-van Vloten Fonds, Lisa Waller Hayes Foundation, Jo Kolk Studiefonds, McKinsey Grant, and Ketel1 Studiefonds. Huland DM contribution to this work was supported in part by NCI training grant: T32 CA118681.
Author Contributions
ELR, GAP, SSG, BAB, ALV, RJS and WST designed the research; WST, SEM, NT, AG, IS, DMH, SH, SRK, AH, RE, TAL, GAF, and GAP performed the research; and WST, SEM, and ELR analyzed data and wrote the paper.
Disclosure
The authors declare no potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tummers, W.S., Miller, S.E., Teraphongphom, N.T. et al. Intraoperative Pancreatic Cancer Detection using Tumor-Specific Multimodality Molecular Imaging. Ann Surg Oncol 25, 1880–1888 (2018). https://doi.org/10.1245/s10434-018-6453-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6453-2