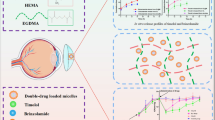
Abstract
Bimatoprost is widely used for the management of glaucoma. Currently, it is delivered via eye drop solution, which is highly inefficient due to low bioavailability. To control the release of ocular drugs, contact lenses are used by scientists. However, the conventional soaking method showed high burst release due to absence of any efficient controlling membrane. The objective of the paper was to apply molecular imprinting technology to improve the loading of bimatoprost from the soaking solution and to sustain the release of drug from the contact lens. The bimatoprost was loaded by conventional soaking method (BT-SM) and compared with the molecular imprinted contact lenses (BT-MP). The loading of bimatoprost by molecular imprinting technology affect the swelling of the contact lens; however, the batch BT-MP-10 did not showed significant alterations. The uptake study showed improvement in the bimatoprost loading by molecular imprinting technology in comparison to the conventional soaking technology. The in vitro bimatoprost release data showed improvement in the bimatoprost release rate profiles with BT-MP contact lenses (up to 36–60 h) lenses in comparison to BT-SM contact lenses (up to 24–36 h). The in vivo rabbit tear fluid data with BT-MP batch showed improvement in the bimatoprost retention time in comparison to BT-SM contact lens and eye drop solution. The rabbit model failed to respond bimatoprost; thus, the efficacy studies need to be conducted on canines or human primates. The paper revealed the potential of using molecular imprinting technology to improve the uptake of bimatoprost and to achieve sustain release kinetics without altering the swelling, transmittance and folding endurance properties of the contact lens.



Similar content being viewed by others
Change history
18 August 2022
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1208/s12249-022-02353-2
References
Mainardes RM, Urban MC, Cinto PO, Khalil NM, Chaud MV, Evangelista RC, et al. Colloidal carriers for ophthalmic drug delivery. Curr Drug Targets. 2005;6(3):363–71.
Davies NM. Biopharmaceutical considerations in topical ocular drug delivery. Clin Exp Pharmacol Physiol. 2000;27(7):558–62.
Ahuja M, Dhake AS, Sharma SK, Majumdar DK. Topical ocular delivery of NSAIDs. AAPS J. 2008;10(2):229–35.
Agrahari V, Mandal A, Agrahari V, Trinh HM, Joseph M, Ray A, et al. A comprehensive insight on ocular pharmacokinetics. Drug Deliv Transl Res. 2016;6(6):735–54.
Rawas-Qalaji M, Williams C-A. Advances in ocular drug delivery. Curr Eye Res. 2012;37(5):345–56.
Gaudana R, Ananthula HK, Parenky A, Mitra AK. Ocular drug delivery. AAPS J. 2010;12(3):348–60.
Kumar A, Malviya R, Sharma PK. Recent trends in ocular drug delivery: a short review. Eur J Appl Sci. 2011;3(3):86–92.
Gaudana R, Jwala J, Boddu SH, Mitra AK. Recent perspectives in ocular drug delivery. Pharm Res. 2009;26(5):1197–205.
Ranch KM, Maulvi FA, Naik MJ, Koli AR, Parikh RK, Shah DO. Optimization of a novel in situ gel for sustained ocular drug delivery using Box-Behnken design: in vitro, ex vivo, in vivo and human studies. Int J Pharm. 2019;554:264–75.
Maulvi FA, Thakkar VT, Soni TG, Gandhi TR. Optimization of aceclofenac solid dispersion using Box-Behnken design: in-vitro and in-vivo evaluation. Curr Drug Deliv. 2014;11(3):380–91.
Maulvi FA, Patil RJ, Desai AR, Shukla MR, Vaidya RJ, Ranch KM, et al. Effect of gold nanoparticles on timolol uptake and its release kinetics from contact lenses: in vitro and in vivo evaluation. Acta Biomater. 2019;86:350–62.
Guzman-Aranguez A, Colligris B, Pintor J. Contact lenses: promising devices for ocular drug delivery. J Ocul Pharmacol Ther. 2013;29(2):189–99.
Xu J, Xue Y, Hu G, Lin T, Gou J, Yin T, et al. A comprehensive review on contact lens for ophthalmic drug delivery. J Control Release. 2018;281:97–118.
Tieppo A, White C, Paine A, Voyles M, McBride M, Byrne M. Sustained in vivo release from imprinted therapeutic contact lenses. J Control Release. 2012;157(3):391–7.
Karlgard C, Wong N, Jones L, Moresoli C. In vitro uptake and release studies of ocular pharmaceutical agents by silicon-containing and p-HEMA hydrogel contact lens materials. Int J Pharm. 2003;257(1–2):141–51.
Soluri A, Hui A, Jones L. Delivery of ketotifen fumarate by commercial contact lens materials. Optom Vis Sci. 2012;89(8):1140–9.
Zhang W, Zu D, Chen J, Peng J, Liu Y, Zhang H, et al. Bovine serum albumin–meloxicam nanoaggregates laden contact lenses for ophthalmic drug delivery in treatment of postcataract endophthalmitis. Int J Pharm. 2014;475(1–2):25–34.
Chandasana H, Prasad YD, Chhonker YS, Chaitanya TK, Mishra NN, Mitra K, et al. Corneal targeted nanoparticles for sustained natamycin delivery and their PK/PD indices: an approach to reduce dose and dosing frequency. Int J Pharm. 2014;477(1–2):317–25.
Kim H-J, Zhang K, Moore L, Ho D. Diamond nanogel-embedded contact lenses mediate lysozyme-dependent therapeutic release. ACS Nano. 2014;8(3):2998–3005.
Venkatesh S, Sizemore SP, Byrne ME. Biomimetic hydrogels for enhanced loading and extended release of ocular therapeutics. Biomaterials. 2007;28(4):717–24.
Hiratani H, Mizutani Y, Alvarez-Lorenzo C. Controlling drug release from imprinted hydrogels by modifying the characteristics of the imprinted cavities. Macromol Biosci. 2005;5(8):728–33.
White CJ, McBride MK, Pate KM, Tieppo A, Byrne ME. Extended release of high molecular weight hydroxypropyl methylcellulose from molecularly imprinted, extended wear silicone hydrogel contact lenses. Biomaterials. 2011;32(24):5698–705.
Ciolino JB, Hudson SP, Mobbs AN, Hoare TR, Iwata NG, Fink GR, et al. A prototype antifungal contact lens. Invest Ophthalmol Vis Sci. 2011;52(9):6286–91.
Ciolino JB, Stefanescu CF, Ross AE, Salvador-Culla B, Cortez P, Ford EM, et al. In vivo performance of a drug-eluting contact lens to treat glaucoma for a month. Biomaterials. 2014;35(1):432–9.
Desai AR, Maulvi FA, Pandya MM, Ranch KM, Vyas BA, Shah SA, et al. Co-delivery of timolol and hyaluronic acid from semi-circular ring-implanted contact lenses for the treatment of glaucoma: in vitro and in vivo evaluation. Biomater Sci. 2018;6(6):1580–91.
Maulvi FA, Singhania SS, Desai AR, Shukla MR, Tannk AS, Ranch KM, et al. Contact lenses with dual drug delivery for the treatment of bacterial conjunctivitis. Int J Pharm. 2018;548(1):139–50.
Costa VP, Braga ME, Duarte CM, Alvarez-Lorenzo C, Concheiro A, Gil MH, et al. Anti-glaucoma drug-loaded contact lenses prepared using supercritical solvent impregnation. J Supercrit Fluids. 2010;53(1–3):165–73.
Costa VP, Braga ME, Guerra JP, Duarte AR, Duarte CM, Leite EO, et al. Development of therapeutic contact lenses using a supercritical solvent impregnation method. J Supercrit Fluids. 2010;52(3):306–16.
Duarte ARC, Simplicio AL, Vega-González A, Subra-Paternault P, Coimbra P, Gil M, et al. Supercritical fluid impregnation of a biocompatible polymer for ophthalmic drug delivery. J Supercrit Fluids. 2007;42(3):373–7.
Li C-C, Abrahamson M, Kapoor Y, Chauhan A. Timolol transport from microemulsions trapped in HEMA gels. J Colloid Interface Sci. 2007;315(1):297–306.
Jones L, Powell CH. Uptake and release phenomena in contact lens care by silicone hydrogel lenses. Eye & Contact Lens. 2013;39(1):29–36.
Li C-C, Chauhan A. Modeling ophthalmic drug delivery by soaked contact lenses. Ind Eng Chem Res. 2006;45(10):3718–34.
Andrade Vivero P, Fernandez Gabriel E, Alvarez Lorenzo C, Concheiro A. Improving the loading and release of NSAIDs from pHEMA hydrogels by copolymerization with functionalized monomers. J Pharm Sci. 2007;96(4):802–13.
White CJ, Byrne ME. Molecularly imprinted therapeutic contact lenses. Expert Opin Drug Deliv. 2010;7(6):765–80.
Alvarez Lorenzo C, Hiratani H, Gómez Amoza JL, Martínez Pacheco R, Souto C, Concheiro A. Soft contact lenses capable of sustained delivery of timolol. J Pharm Sci. 2002;91(10):2182–92.
Hiratani H, Mizutani Y, Alvarez Lorenzo C. Controlling drug release from imprinted hydrogels by modifying the characteristics of the imprinted cavities. Macromol Biosci. 2005;5(8):728–33.
Alvarez-Lorenzo C, Yanez F, Barreiro-Iglesias R, Concheiro A. Imprinted soft contact lenses as norfloxacin delivery systems. J Control Release. 2006;113(3):236–44.
Tieppo A, Pate KM, Byrne ME. In vitro controlled release of an anti-inflammatory from daily disposable therapeutic contact lenses under physiological ocular tear flow. Eur J Pharm Biopharm. 2012;81(1):170–7.
Tang L, Zhao C-Y, Wang X-H, Li R-S, Yang J-R, Huang Y-P, et al. Macromolecular crowding of molecular imprinting: a facile pathway to produce drug delivery devices for zero-order sustained release. Int J Pharm. 2015;496(2):822–33.
Malakooti N, Alexander C, Alvarez-Lorenzo C. Imprinted contact lenses for sustained release of polymyxin B and related antimicrobial peptides. J Pharm Sci. 2015;104(10):3386–94.
Maulvi FA, Parmar RJ, Shukla MR, Desai AR, Desai DT, Ranch KM, et al. Plackett-Burman design for screening of critical variables and their effects on the optical transparency and swelling of gatifloxacin-Pluronic-loaded contact lens. Int J Pharm. 2019;566:513–9.
Alvarez-Lorenzo C, Hiratani H, Gomez-Amoza JL, Martínez-Pacheco R, Souto C, Concheiro A. Soft contact lenses capable of sustained delivery of timolol. J Pharm Sci. 2002;91(10):2182–92.
Hiratani H, Alvarez-Lorenzo C. The nature of backbone monomers determines the performance of imprinted soft contact lenses as timolol drug delivery systems. Biomaterials. 2004;25(6):1105–13.
Kim J, Chauhan A. Dexamethasone transport and ocular delivery from poly (hydroxyethyl methacrylate) gels. Int J Pharm. 2008;353(1–2):205–22.
Quesnel N-M, Simonet P. Spectral transmittance of UV-absorbing soft and rigid gas permeable contact lenses. Optometry vision science: official publication of the American Academy of Optometry. 1995;72(1):2–10.
Laube T, Apel H, Koch H-R. Ultraviolet radiation absorption of intraocular lenses. Ophthalmology. 2004;111(5):880–5.
Quesnel N-M, Fares F, Verret E, Giasson C. Evaluation of the spectral transmittance of UV-absorbing disposable contact lenses. The CLAO journal: official publication of the Contact Lens Association of Ophthalmologists, Inc. 2001;27(1):23–9.
Frohn A, Dick HB, Augustin AJ, Grus FH. Late opacification of the foldable hydrophilic acrylic lens SC60B-OUV. Ophthalmology. 2001;108(11):1999–2004.
Boateng J, Popescu A. Composite bi-layered erodible films for potential ocular drug delivery. Colloids Surf B: Biointerfaces. 2016;145:353–61.
Rao PCM, Nappinnai M, Raju S, Rao UMV, Reddy VB. Fluconazole ocular inserts: formulation and in-vitro evaluation. J Pharm Sci Res. 2010;2(6):344–50.
Maxey KM, Johnson JL, LaBrecque J. The hydrolysis of bimatoprost in corneal tissue generates a potent prostanoid FP receptor agonist. Surv Ophthalmol. 2002;47:S34–40.
Kumar SS, Natraj K, Khan A, Kumar BK, Rao J. Development and validation of RP-HPLC method for estimation of bimatoprost in pharmaceutical dosage forms. J Pharm Res. 2011;4(10):3733–4.
Ciolino JB, Hoare TR, Iwata NG, Behlau I, Dohlman CH, Langer R, et al. A drug-eluting contact lens. Invest Ophthalmol Vis Sci. 2009;50(7):3346–52.
Lee SE, Kim SR, Park M. Oxygen permeability of soft contact lenses in different pH, osmolality and buffering solution. Int J Ophthalmol. 2015;8(5):1037–40.
Pozuelo J, Compañ V, González-Méijome JM, González M, Mollá S. Oxygen and ionic transport in hydrogel and silicone-hydrogel contact lens materials: an experimental and theoretical study. J Membr Sci. 2014;452:62–72.
Peng C-C, Chauhan A. Ion transport in silicone hydrogel contact lenses. J Membr Sci. 2012;399:95–105.
W.C. Hoffman, Light filtering contact lens, Google Patents, 1997.
Nichols P, Mortensen N, Tingy D, Vasquez A, Bates J. Pharmacokinetics of molecular imprinted hydrogels as drug delivery vehicles. Biochem Pharmacol. 2018;7(238):2167–0501.1000238.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1208/s12249-022-02353-2"
Electronic supplementary material
ESM 1
(DOCX 192 kb)
About this article
Cite this article
Yan, F., Liu, Y., Han, S. et al. RETRACTED ARTICLE: Bimatoprost Imprinted Silicone Contact Lens to Treat Glaucoma. AAPS PharmSciTech 21, 63 (2020). https://doi.org/10.1208/s12249-020-1622-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-020-1622-6