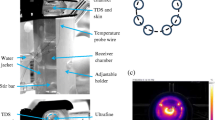
Abstract
During skin sonoporation and sonophoresis, time-consuming duty cycles or fluid replacement is often required to mitigate coupling fluid temperature increases. This study demonstrates an alternative method for temperature regulation: a circulating, thermoelectric system. Porcine skin samples were sonoporated continuously for 10 min at one of three intensities (23.8, 34.2, 39.4 W/m2). A caffeine solution was then applied to the skin and left to diffuse for 20 h. During sonoporation, the system was able to maintain the temperature between 10 and 16°C regardless of the intensity. No increase in transdermal transport was achieved with an intensity of 23.8 W/m2. Intensities of 34.2 and 39.4 W/m2 resulted in 3.5-fold (p < 0.05) and 3.7-fold (p < 0.05) increases in mean transport, relative to a control case with no ultrasound. From these results, it is concluded that a significant transport increase can be achieved with a system that circulates and cools the coupling fluid during ultrasound application. Relative to the previous methods of temperature control (duty cycles and fluid replacement), use of this circulation system will lead to significant time savings in future experimental studies.













Similar content being viewed by others
References
Prausnitz MR, Langer R. Transdermal drug delivery. Nat Biotechnol. 2008;26(11):1261–8.
Boucaud A, Montharu J, Machet L, Arbeille B, Machet MC, Patat F, et al. Clinical, histologic, and electron microscopy study of skin exposed to low-frequency ultrasound. Anat Rec. 2001;264(1):114–9.
Ogura M, Paliwal S, Mitragotri S. Low-frequency sonophoresis: current status and future prospects. Adv Drug Deliv Rev. 2008;60(10):1218–23.
Lee SE, Choi KJ, Menon GK, Kim HJ, Choi EH, Ahn SK, et al. Penetration pathways induced by low-frequency sonophoresis with physical and chemical enhancers: iron oxide nanoparticles versus lanthanum nitrates. J Invest Dermatol. 2010;130(4):1063–72.
Merino G, Kalia YN, Delgado-Charro MB, Potts RO, Guy RH. Frequency and thermal effects on the enhancement of transdermal transport by sonophoresis. J Control Release. 2003;88(1):85–94.
Polat BE, Deen WM, Langer R, Blankschtein D. A physical mechanism to explain the delivery of chemical penetration enhancers into skin during transdermal sonophoresis—insight into the observed synergism. J Control Release. 2012;158(2):250–60.
Mitragotri S, Blankschtein D, Langer R. Transdermal drug delivery using low-frequency sonophoresis. Pharm Res. 1996;13(3):411–20.
Morimoto Y, Mutoh M, Ueda H, Fang L, Hirayama K, Atobe M, et al. Elucidation of the transport pathway in hairless rat skin enhanced by low-frequency sonophoresis based on the solute–water transport relationship and confocal microscopy. J Control Release. 2005;103(3):587–97.
Mitragotri S, Kost J. Low-frequency sonophoresis: a review. Adv Drug Deliv Rev. 2004;56(5):589–601.
Zorec B, Jelenc J, Miklavčič D, Pavšelj N. Ultrasound and electric pulses for transdermal drug delivery enhancement: ex vivo assessment of methods with in vivo oriented experimental protocols. Int J Pharm. 2015;490(1–2):65–73.
Boucaud A, Garrigue MA, Machet L, Lc V, Patat F. Effect of sonication parameters on transdermal delivery of insulin to hairless rats. J Control Release. 2002;81(1):113–9.
Kushner J, Blankschtein D, Langer R. Experimental demonstration of the existence of highly permeable localized transport regions in low-frequency sonophoresis. J Pharm Sci. 2004;93(11):2733–45.
Ueda H, Mutoh M, Seki T, Kobayashi D, Morimoto Y. Acoustic cavitation as an enhancing mechanism of low-frequency sonophoresis for transdermal drug delivery. Biol Pharm Bull. 2009;32(5):916–20.
Rangsimawong W, Obata Y, Opanasopit P, Ngawhirunpat T, Takayama K. Enhancement of galantamine HBr skin permeation using sonophoresis and limonene-containing PEGylated liposomes. AAPS PharmSciTech. 2018;19(3):1093–104.
Terahara T, Mitragotri S, Kost J, Langer R. Dependence of low-frequency sonophoresis on ultrasound parameters; distance of the horn and intensity. Int J Pharm. 2002a;235(1–2):35–42.
Herwadkar A, Sachdeva V, Taylor LF, Silver H, Banga AK. Low frequency sonophoresis mediated transdermal and intradermal delivery of ketoprofen. Int J Pharm. 2012;423(2):289–96.
Terahara T, Mitragotri S, Langer R. Porous resins as a cavitation enhancer for low-frequency sonophoresis. J Pharm Sci. 2002b;91(3):753–9.
Lindeque BGPMD, Shuler FDMDP, Bates CMMD. Skin temperatures generated following plaster splint application. Orthopedics (Online). 2013;36(5):364–7.
Hao J, Ghosh P, Li SK, Newman B, Kasting GB, Raney SG. Heat effects on drug delivery across human skin. Expert Opin Drug Deliv. 2016;13(5):755–68.
Moritz AR, Henriques FC. Studies of thermal injury: II. The relative importance of time and surface temperature in the causation of cutaneous burns. Am J Pathol. 1947;23(5):695–720.
Williamson C, Scholtz JR. Time-temperature relationships in thermal blister formation*. J Investig Dermatol. 1949;12(1):41–7.
Tang H, Wang CCJ, Blankschtein D, Langer R. An investigation of the role of cavitation in low-frequency ultrasound-mediated transdermal drug transport. Pharm Res. 2002a;19(8):1160–9.
Lavon I, Grossman N, Kost J. The nature of ultrasound–SLS synergism during enhanced transdermal transport. J Control Release. 2005;107(3):484–94.
Paliwal S, Menon GK, Mitragotri S. Low-frequency sonophoresis: ultrastructural basis for stratum corneum permeability assessed using quantum dots. J Invest Dermatol. 2006;126(5):1095–101.
Polat BE, Figueroa PL, Blankschtein D, Langer R. Transport pathways and enhancement mechanisms within localized and non-localized transport regions in skin treated with low-frequency sonophoresis and sodium lauryl sulfate. J Pharm Sci. 2011a;100(2):512–29.
Tezel A, Sens A, Tuchscherer J, Mitragotri S. Frequency dependence of sonophoresis. Pharm Res. 2001;18(12):1694–700.
Tezel A, Dokka S, Kelly S, Hardee GE, Mitragotri S. Topical delivery of anti-sense oligonucleotides using low-frequency sonophoresis. Pharm Res. 2004;21(12):2219–25.
Sarheed O, Abdul Rasool BK. Development of an optimised application protocol for sonophoretic transdermal delivery of a model hydrophilic drug. Open Biomed Eng J. 2011;5:14–24.
Rich KT, Hoerig CL, Rao MB, Mast TD. Relations between acoustic cavitation and skin resistance during intermediate- and high-frequency sonophoresis. J Control Release. 2014;194:266–77.
Han T, Das DB. Permeability enhancement for transdermal delivery of large molecule using low-frequency sonophoresis combined with microneedles. J Pharm Sci. 2013;102(10):3614–22.
Mitragotri S, Farrell J, Tang H, Terahara T, Kost J, Langer R. Determination of threshold energy dose for ultrasound-induced transdermal drug transport. J Control Release. 2000a;63(1–2):41–52.
Mitragotri S, Ray D, Farrell J, Tang H, Yu B, Kost J, et al. Synergistic effect of low-frequency ultrasound and sodium lauryl sulfate on transdermal transport. J Pharm Sci. 2000b;89(7):892–900.
Mutoh M, Ueda H, Nakamura Y, Hirayama K, Atobe M, Kobayashi D, et al. Characterization of transdermal solute transport induced by low-frequency ultrasound in the hairless rat skin. J Control Release. 2003;92(1–2):137–46.
Robertson J, Becker S. Influence of acoustic reflection on the inertial cavitation dose in a Franz diffusion cell. Ultrasound Med Biol. 2018;44(5):1100–9.
Dunne A, Crampton D, Egaña M. Effect of post-exercise hydrotherapy water temperature on subsequent exhaustive running performance in normothermic conditions. J Sci Med Sport. 2013;16(5):466–71.
Vichare V, Mujgond P, Tambe V, Dhole S. Simultaneous spectrophotometric determination of paracetamol and caffeine in tablet formulation. Int J PharmTech Res. 2010;2(4):2512–6.
Franeta J, Agbaba D, Eric S, Pavkov S, Aleksic M, Vladimirov S. HPLC assay of acetylsalicylic acid, paracetamol, caffeine and phenobarbital in tablets. Il Farmaco. 2002;57(9):709–13.
Rodrigues CI, Marta L, Maia R, Miranda M, Ribeirinho M, Máguas C. Application of solid-phase extraction to brewed coffee caffeine and organic acid determination by UV/HPLC. J Food Compos Anal. 2007;20(5):440–8.
Fernandez P, Martin M, Gonzalez A, Pablos F. HPLC determination of catechins and caffeine in tea. Differentiation of green, black and instant teas. Analyst. 2000;125(3):421–5.
Hubert M, Vandervieren E. An adjusted boxplot for skewed distributions. Comput Stat Data Anal. 2008;52(12):5186–201.
Krzywinski M, Altman N. Points of significance: visualizing samples with box plots: Nature Publishing Group; 2014.
Spitzer M, Wildenhain J, Rappsilber J, Tyers M. BoxPlotR: a web tool for generation of box plots. Nat Methods. 2014;11(2):121–2.
Whyte WA, Orlando DA, Hnisz D, Abraham BJ, Lin CY, Kagey MH, et al. Master transcription factors and mediator establish super-enhancers at key cell identity genes. Cell. 2013;153(2):307–19.
Tezel A, Sens A, Mitragotri S. Investigations of the role of cavitation in low-frequency sonophoresis using acoustic spectroscopy. J Pharm Sci. 2002;91(2):444–53.
Bader KB, Raymond JL, Mobley J, Church CC, Felipe Gaitan D. The effect of static pressure on the inertial cavitation threshold. J Acoust Soc Am. 2012;132(2):728–37.
Brabec K, Mornstein V. Detection of ultrasonic cavitation based on low-frequency analysis of acoustic signal. Cent Eur J Biol. 2007;2(2):213–21.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Robertson, J., Squire, M. & Becker, S. A Thermoelectric Device for Coupling Fluid Temperature Regulation During Continuous Skin Sonoporation or Sonophoresis. AAPS PharmSciTech 20, 147 (2019). https://doi.org/10.1208/s12249-019-1357-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-019-1357-4