Abstract
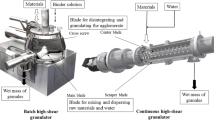
The objective of this study is to use near-infrared spectroscopy (NIRS) coupled with multivariate chemometric models to monitor granule and tablet quality attributes in the formulation development and manufacturing of ciprofloxacin hydrochloride (CIP) immediate release tablets. Critical roller compaction process parameters, compression force (CFt), and formulation variables identified from our earlier studies were evaluated in more detail. Multivariate principal component analysis (PCA) and partial least square (PLS) models were developed during the development stage and used as a control tool to predict the quality of granules and tablets. Validated models were used to monitor and control batches manufactured at different sites to assess their robustness to change. The results showed that roll pressure (RP) and CFt played a critical role in the quality of the granules and the finished product within the range tested. Replacing binder source did not statistically influence the quality attributes of the granules and tablets. However, lubricant type has significantly impacted the granule size. Blend uniformity, crushing force, disintegration time during the manufacturing was predicted using validated PLS regression models with acceptable standard error of prediction (SEP) values, whereas the models resulted in higher SEP for batches obtained from different manufacturing site. From this study, we were able to identify critical factors which could impact the quality attributes of the CIP IR tablets. In summary, we demonstrated the ability of near-infrared spectroscopy coupled with chemometrics as a powerful tool to monitor critical quality attributes (CQA) identified during formulation development.













Similar content being viewed by others
References
Teng Y, Qiu Z, Wen H. Systematical approach of formulation and process development using roller compaction. Eur J Pharm Biopharm. 2009;73(2):219–29.
Malkowska S, Khan KA. Effect of re-conpression on the properties of tablets prepared by dry granulation. Drug Dev Ind Pharm. 1983;9(3):331–47.
Sun C, Himmelspach M. Reduced tabletability of roller compacted granules as a result of granule size enlargement. J Pharm Sci. 2006;95(1):200–6.
Roberts RJ, Rowe RC. The effect of punch velocity on the compaction of a variety of materials. J Pharm Pharmacol. 1985;37(6):377–84.
Ruegger CE, Celik M. The effect of compression and decompression speed on the mechanical strength of compacts. Pharm Dev Technol. 2000;5(4):485–94.
Bacher C, Olsen PM, Bertelsen P, Kristensen J, Sonnergaard JM. Improving the compaction properties of roller compacted calcium carbonate. Int J Pharm. 2007;342(1–2):115–23.
Inghelbrecht S, Remon JP. Roller compaction and tableting of microcrystalline cellulose/drug mixtures. Int J Pharm. 1998;161(2):215–24.
Daugherity PD, Chu JH. Investigation of serrated roll surface differences on ribbon thickness during roller compaction. Pharm Dev Technol. 2007;12(6):603–8.
Tabasi S, Fahmy R, Bensley D, O’Brien C, Hoag S. Quality by design, part I: application of NIR spectroscopy to monitor tablet manufacturing process. J Pharm Sci. 2008;97(9):4040–51.
ICH. Q8(R2): Pharmaceutical development. Part I: pharmaceutical development, and part II: annex to pharmaceutical development. http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q8_R1/Step4/Q8_R2_Guideline.pdf (2009).
Cantor S, Hoag S, Ellison C, Khan M, Lyon R. NIR spectroscopy applications in the development of a compacted multiparticulate system for modified release. AAPS PharmSciTech. 2011;12(1):262–78.
Tabasi S, Fahmy R, Bensley D, O’Brien C, Hoag S. Quality by design, part II: application of NIR spectroscopy to monitor the coating process for a pharmaceutical sustained release product. J Pharm Sci. 2008;97(9):4052–66.
Tatavarti A, Fahmy R, Wu H, Hussain A, Marnane W, Bensley D, et al. Assessment of NIR spectroscopy for nondestructive analysis of physical and chemical attributes of sulfamethazine bolus dosage forms. AAPS PharmSciTech. 2005;6(1):E91–9.
Gupta A, Peck G, Miller R, Morris K. Influence of ambient moisture on the compaction behavior of microcrystalline cellulose powder undergoing uni-axial compression and roller-compaction: a comparative study using near-infrared spectroscopy. J Pharm Sci. 2005;94(10):2301–13.
Gupta A, Peck G, Miller R, Morris K. Real-time near-infrared monitoring of content uniformity, moisture content, compact density, tensile strength, and Young’s modulus of roller compacted powder blends. J Pharm Sci. 2005;94(7):1589–97.
Soh J, Boersen N, Carvajal T, Morris K, Peck G, Pinal R. Importance of raw material attributes for modeling ribbon and granule properties in roller compaction: multivariate analysis on roll gap and NIR spectral slope as process critical control parameters. J Pharm Innov. 2007;2:106–24.
ICH: Q9 Quality Risk Management. http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q9/Step4/Q9_Guideline.pdf (2005).
ICH: Q10 Pharmaceutical Quality System. http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q10/Step4/Q10_Guideline.pdf (2008).
Fahmy R, Kona R, Dandu R, Xie W, Claycamp G, Hoag S. Quality by design I: application of failure mode effect analysis (FMEA) and Plackett-Burman design of experiments in the identification of “main factors” in the formulation and process design space for roller-compacted ciprofloxacin hydrochloride immediate-release tablets. AAPS PharmSciTech. 2012;13(4):1243–54.
Claycamp G, Kona R, Fahmy R, Polli J, Martinez M, Hoag S. Quality-by-design II: application of quantitative risk analysis to the formulation of ciprofloxacin tablets. in preparation for PharmSciTech. 2012.
Desai D, Rinaldi F, Kothari S, Paruchuri S, Li D, Lai M, et al. Effect of hydroxypropyl cellulose (HPC) on dissolution rate of hydrochlorothiazide tablets. Int J Pharm. 2006;308(1–2):40–5.
Olivera ME, Manzo RH, Junginger HE, Midha KK, Shah VP, Stavchansky S, et al. Biowaiver monographs for immediate release solid oral dosage forms: Ciprofloxacin hydrochloride. J Pharm Sci. 2011;100(1):22–33.
Lim H, Dave V, Kidder L, Neil Lewis E, Fahmy R, Hoag S. Assessment of the critical factors affecting the porosity of roller compacted ribbons and the feasibility of using NIR chemical imaging to evaluate the porosity distribution. Int J Pharm. 2011;410(1–2):1–8.
Okoye P. Lubrication of direct-compressible blends. Pharm Technol. 2007;31(9):116.
Dave V, Fahmy R, Bensley D, Hoag S. Eudragit(®) RS PO/RL PO as rate-controlling matrix-formers via roller compaction: influence of formulation and process variables on functional attributes of granules and tablets. Drug Dev Ind Pharm. 2012.
Dave V, Fahmy R, Hoag SW. Roller Compaction of Eudragit polymers Part 2. Drug Dev Ind Pharm. 2011; Submitted to Drug Dev Ind Pharm.
Acknowledgments
The authors would like to acknowledge the financial assistance from the FDA and CIPET. Drs. Bancha Chuasuwan, Ramesh Dandu, and Walter Xie for their technical assistance with the CIP assay. Thanks to Ryan McCann for his assistance with roller compaction at Purdue University, School of Pharmacy.
Disclaimer
The views expressed in this manuscript are the personal opinion of the authors and do not necessarily reflects the views or policies of the FDA
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Robin Bogner, James Drennen, Mansoor Khan, Cynthia Oksanen, and Gintaras Reklaitis
Glossary
- ANOVA
-
Analysis of variance
- CFt
-
Compression force
- D or D(3,4)
-
Volume mean diameter
- DT
-
Disintegration time
- Dt
-
Tapped density
- Db
-
Bulk density
- NIRS
-
Near-infrared spectrometry
- PAT
-
Process analytical technology
- PCA
-
Principal component analysis
- PCR
-
Principal component regression
- PLS
-
Partial least squares
- PLSR
-
Partial least square regression
- PRESS
-
Prediction residual error-sum squares
- SEC
-
Standard error of calibration
- SECV
-
Standard error of cross-validation
- SEP
-
Standard error of prediction
- SG
-
Savitzky–Golay
- 2nd D
-
Second derivative
Rights and permissions
About this article
Cite this article
Kona, R., Fahmy, R.M., Claycamp, G. et al. Quality-by-Design III: Application of Near-Infrared Spectroscopy to Monitor Roller Compaction In-process and Product Quality Attributes of Immediate Release Tablets. AAPS PharmSciTech 16, 202–216 (2015). https://doi.org/10.1208/s12249-014-0180-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-014-0180-1