Abstract
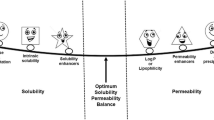
Rifaximin is a BCS class IV (low-solubility, low-permeability) drug and also a P-gp substrate. The aims of this work were to assess the efficiency of different rifaximin amorphous solid dispersion (ASDs) formulations in achieving and maintaining supersaturation and to investigate the consequent solubility-permeability interplay. Spray-dried rifaximin ASDs were prepared with different hydrophilic polymers and their ability to achieve and maintain supersaturation was assessed. Then, rifaximin’s apparent intestinal permeability was investigated as a function of increasing supersaturation both in vitro using the parallel artificial membrane permeability assay (PAMPA) and in vivo using the single-pass rat intestinal perfusion (SPIP) model. The efficiency of the different ASDs to achieve and maintain supersaturation of rifaximin was found to be highly polymer dependent, and the copovidone/HPC-SL formulation was found to be superior to the other two, allowing supersaturation of 200× that of the crystalline solubility for 20 h. In vitro, rifaximin flux was increased and the apparent permeability was constant as a function of increasing supersaturation level. In vivo, on the other hand, absorption rate coefficient (k a) was first constant as a function of increasing supersaturation, but at 250×, the crystalline solubility k a was doubled, similar to the k a in the presence of the strong P-gp inhibitor GF120918. In conclusion, a new and favorable nature of solubility-permeability interplay was revealed in this work: delivering high supersaturation level of the BCS class IV drug rifaximin via ASD, thereby saturating the drugs’ P-gp-mediated efflux transport, led to the favorable unique win-win situation, where both the solubility and the permeability increased simultaneously.





Similar content being viewed by others
References
Dahan A, Wolk O, Kim YH, Ramachandran C, Crippen GM, Takagi T, et al. Purely in silico BCS classification: science based quality standards for the world’s drugs. Mol Pharm. 2013;10:4378–90.
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev. 2001;46:3–26.
Dahan A, Beig A, Lindley D, Miller JM. The solubility–permeability interplay and oral drug formulation design: two heads are better than one. Adv Drug Deliv Rev. 2016;101:99–107.
Dahan A, Miller J. The solubility–permeability interplay and its implications in formulation design and development for poorly soluble drugs. AAPS J. 2012;14:244–51.
Amidon GL, Lennernas H, Shah VP, Crison JR. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res. 1995;12:413.
Dahan A, Miller J, Amidon G. Prediction of solubility and permeability class membership: provisional BCS classification of the world’s top oral drugs. AAPS J. 2009;11:740–6.
Lobenberg R, Amidon GL. Modern bioavailability, bioequivalence and biopharmaceutics classification system. New scientific approaches to international regulatory standards. Eur J Pharm Biopharm. 2000;50:3–12.
Beig A, Agbaria R, Dahan A. The use of captisol (SBE7-β-CD) in oral solubility-enabling formulations: comparison to HPβCD and the solubility–permeability interplay. Eur J Pharm Sci. 2015;77:73–8.
Dahan A, Miller JM, Hoffman A, Amidon GE, Amidon GL. The solubility-permeability interplay in using cyclodextrins as pharmaceutical solubilizers: mechanistic modeling and application to progesterone. J Pharm Sci. 2010;99:2739–49.
Miller JM, Dahan A. Predicting the solubility–permeability interplay when using cyclodextrins in solubility-enabling formulations: model validation. Int J Pharm. 2012;430:388–91.
Miller JM, Beig A, Krieg BJ, Carr RA, Borchardt TB, Amidon GE, et al. The solubility-permeability interplay: mechanistic modeling and predictive application of the impact of micellar solubilization on intestinal permeation. Mol Pharm. 2011;8:1848–56.
Beig A, Miller JM, Dahan A. Accounting for the solubility–permeability interplay in oral formulation development for poor water solubility drugs: the effect of PEG-400 on carbamazepine absorption. Eur J Pharm Biopharm. 2012;81:386–91.
Beig A, Miller JM, Lindley D, Dahan A. Striking the optimal solubility-permeability balance in oral formulation development for lipophilic drugs: maximizing carbamazepine blood levels. Mol Pharm. 2017;14:319–27.
Miller JM, Beig A, Carr RA, Webster GK, Dahan A. The solubility–permeability interplay when using cosolvents for solubilization: revising the way we use solubility-enabling formulations. Mol Pharm. 2012;9:581–90.
Beig A, Lindley D, Miller JM, Agbaria R, Dahan A. Hydrotropic solubilization of lipophilic drugs for oral delivery: the effects of urea and nicotinamide on carbamazepine solubility–permeability interplay. Front Pharmacol. 2016;7:379.
Brouwers J, Brewster ME, Augustijns P. Supersaturating drug delivery systems: the answer to solubility-limited oral bioavailability? J Pharm Sci. 2009;98:2549–72.
Kawabata Y, Wada K, Nakatani M, Yamada S, Onoue S. Formulation design for poorly water-soluble drugs based on biopharmaceutics classification system: basic approaches and practical applications. Int J Pharm. 2011;420:1–10.
van Hoogevest P, Liu X, Fahr A. Drug delivery strategies for poorly water-soluble drugs: the industrial perspective. Expert Opin Drug Deliv. 2011;8:1481–500.
Bhugra C, Pikal MJ. Role of thermodynamic, molecular, and kinetic factors in crystallization from the amorphous state. J Pharm Sci. 2008;97:1329–49.
Murdande SB, Pikal MJ, Shanker RM, Bogner RH. Solubility advantage of amorphous pharmaceuticals: I. A thermodynamic analysis. J Pharm Sci. 2010;99:1254–64.
Vasconcelos T, Sarmento B, Costa P. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov Today. 2007;12:1068–75.
Beig A, Miller JM, Lindley D, Carr RA, Zocharski P, Agbaria R, et al. Head-to-head comparison of different solubility-enabling formulations of etoposide and their consequent solubility-permeability interplay. J Pharm Sci. 2015;104:2941–7.
Dahan A, Beig A, Ioffe-Dahan V, Agbaria R, Miller J. The twofold advantage of the amorphous form as an oral drug delivery practice for lipophilic compounds: increased apparent solubility and drug flux through the intestinal membrane. AAPS J. 2013;15:347–53.
Miller JM, Beig A, Carr RA, Spence JK, Dahan A. A win–win solution in oral delivery of lipophilic drugs: supersaturation via amorphous solid dispersions increases apparent solubility without sacrifice of intestinal membrane permeability. Mol Pharm. 2012;9:2009–16.
Adachi JA, DuPont HL. Rifaximin: a novel nonabsorbed rifamycin for gastrointestinal disorders. Clin Infect Dis. 2006;42:541–7.
Cottreau J, Baker SF, DuPont HL, Garey KW. Rifaximin: a nonsystemic rifamycin antibiotic for gastrointestinal infections. Expert Rev Anti Infect Ther. 2010;8:747–60.
Benet LZ, Broccatelli F, Oprea TI. BDDCS applied to over 900 drugs. AAPS J. 2011;13:519–47.
Pham-The H, Garrigues T, Bermejo M, González-Álvarez I, Monteagudo MC, Cabrera-Pérez MÁ. Provisional classification and in silico study of biopharmaceutical system based on Caco-2 cell permeability and dose number. Mol Pharm. 2013;10:2445–61.
Wolk O, Agbaria R, Dahan A. Provisional in-silico biopharmaceutics classification (BCS) to guide oral drug product development. Drug Des Devel Ther. 2014;8:1563–75.
Fairstein M, Swissa R, Dahan A. Regional-dependent intestinal permeability and BCS classification: elucidation of pH-related complexity in rats using pseudoephedrine. AAPS J. 2013;15:589–97.
Beig A, Agbaria R, Dahan A. Oral delivery of lipophilic drugs: the tradeoff between solubility increase and permeability decrease when using cyclodextrin-based formulations. PLoS One. 2013;8:e68237.
Zur M, Hanson AS, Dahan A. The complexity of intestinal permeability: assigning the correct BCS classification through careful data interpretation. Eur J Pharm Sci. 2014;61:11–7.
Zur M, Cohen N, Agbaria R, Dahan A. The biopharmaceutics of successful controlled release drug product: segmental-dependent permeability of glipizide vs. metoprolol throughout the intestinal tract. Int J Pharm. 2015;489:304–10.
Zur M, Gasparini M, Wolk O, Amidon GL, Dahan A. The low/high BCS permeability class boundary: physicochemical comparison of metoprolol and labetalol. Mol Pharm. 2014;11:1707–14.
Lozoya-Agullo I, Zur M, Beig A, Fine N, Cohen Y, González-Álvarez M, et al. Segmental-dependent permeability throughout the small intestine following oral drug administration: single-pass vs. Doluisio approach to in-situ rat perfusion. Int J Pharm. 2016;515:201–8.
Lozoya-Agullo I, Zur M, Wolk O, Beig A, González-Álvarez I, González-Álvarez M, et al. In-situ intestinal rat perfusions for human Fabs prediction and BCS permeability class determination: investigation of the single-pass vs. the Doluisio experimental approaches. Int J Pharm. 2015;480:1–7.
Beig A, Dahan A. Quantification of carbamazepine and its 10,11-epoxide metabolite in rat plasma by UPLC-UV and application to pharmacokinetic study. Biomed Chromatogr. 2014;28:934–8.
Dahan A, Amidon GL. Segmental dependent transport of low permeability compounds along the small intestine due to P-gp: the role of efflux transport in the oral absorption of BCS class III drugs. Mol Pharm. 2009;6:19–28.
Dahan A, Amidon GL. Grapefruit juice and its constituents augment colchicine intestinal absorption: potential hazardous interaction and the role of p-glycoprotein. Pharm Res. 2009;26:883–92.
Dahan A, Sabit H, Amidon GL. Multiple efflux pumps are involved in the transepithelial transport of colchicine: combined effect of P-gp and MRP2 leads to decreased intestinal absorption throughout the entire small intestine. Drug Metab Dispos. 2009;37:2028–36.
Dahan A, Sabit H, Amidon GL. The H2 receptor antagonist nizatidine is a P-glycoprotein substrate: characterization of its intestinal epithelial cell efflux transport. AAPS J. 2009;11:205–13.
Amidon GE, Higuchi WI, Ho NFH. Theoretical and experimental studies of transport of micelle-solubilized solutes. J Pharm Sci. 1982;71:77–84.
Beig A, Miller JM, Dahan A. The interaction of nifedipine with selected cyclodextrins and the subsequent solubility–permeability trade-off. Eur J Pharm Biopharm. 2013;85:1293–9.
Bermejo MV, Pérez-Varona AT, Segura-Bono MJ, Martín-Villodre A, Plá-Delfina JM, Garrigues TM. Compared effects of synthetic and natural bile acid surfactants on xenobiotic absorption I. Studies with polysorbate and taurocholate in rat colon. Int J Pharm. 1991;69:221–31.
Frank KJ, Westedt U, Rosenblatt KM, Hölig P, Rosenberg J, Mägerlein M, et al. Impact of FaSSIF on the solubility and dissolution-/permeation rate of a poorly water-soluble compound. Eur J Pharm Sci. 2012;47:16–20.
Feeney OM, Crum MF, McEvoy CL, Trevaskis NL, Williams HD, Pouton CW, et al. 50 years of oral lipid-based formulations: provenance, progress and future perspectives. Adv Drug Deliv Rev. 2016;101:167–94.
Frank KJ, Westedt U, Rosenblatt KM, Hölig P, Rosenberg J, Mägerlein M, et al. What is the mechanism behind increased permeation rate of a poorly soluble drug from aqueous dispersions of an amorphous solid dispersion? J Pharm Sci. 2014;103:1779–86.
Grant D, Higuchi T. Activities of solutes, selection of standard state, and Henry’s law constants. In: Weissberger A, editor. Solubility behavior of organic compounds. New York: Wiley; 1990. p. 89–133.
Grant D, Higuchi T. Solubility in and partitioning into water. In: Weissberger A, editor. Solubility behavior of organic compounds. New York: Wiley; 1990. p. 355–98.
Bajaj JS, Riggio O. Drug therapy: rifaximin. Hepatology. 2010;52:1484–8.
Huang DB, DuPont HL. Rifaximin—a novel antimicrobial for enteric infections. J Infect. 2005;50:97–106.
Blandizzi C, Viscomi G, Scarpignato C. Impact of crystal polymorphism on the systemic bioavailability of rifaximin, an antibiotic acting locally in the gastrointestinal tract, in healthy volunteers. Drug Des Devel Ther. 2014;9:1–11.
Acknowledgments
This work was supported by a research grant from AbbVie Inc. This work is part of Avital Beig’s Ph.D. dissertation. The authors thank Dr Philip Zocharski and Dr Xiaochun Lou for their assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beig, A., Fine-Shamir, N., Lindley, D. et al. Advantageous Solubility-Permeability Interplay When Using Amorphous Solid Dispersion (ASD) Formulation for the BCS Class IV P-gp Substrate Rifaximin: Simultaneous Increase of Both the Solubility and the Permeability. AAPS J 19, 806–813 (2017). https://doi.org/10.1208/s12248-017-0052-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-017-0052-1