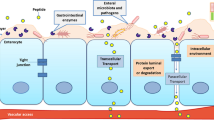
Abstract
Absorption enhancers are functional excipients included in formulations to improve the absorption of a pharmacologically active drug. The term absorption enhancer usually refers to an agent whose function is to increase absorption by enhancing membrane permeation, rather than increasing solubility, so such agents are sometimes more specifically termed permeation enhancers. Absorption enhancers have been investigated for at least two decades, particularly in efforts to develop non-injection formulations for peptides, proteins, and other pharmacologically active compounds that have poor membrane permeability. While at least one product utilizing an absorption enhancer for transdermal use has reached the market, quite a few more appear to be at the threshold of becoming products, and these include oral and transmucosal applications. This paper will review some of the most advanced absorption enhancers currently in development and the formulation technologies employed that have led to their success. In addition, a more basic review of the barriers to absorption and the mechanisms by which those barriers can be surmounted is presented. Factors influencing the success of absorption-enhancing formulations are discussed. If ultimately successful, the products now in development should offer non-injection alternatives for several peptide or protein drugs currently only administered by injection. The introduction of new absorption enhancers as accepted pharmaceutical excipients, and the development of formulation technologies that afford the greatest benefit/risk ratio for their use, may create opportunities to apply these enabling technologies more broadly to existing drugs with non-optimal delivery properties.
Similar content being viewed by others
REFERENCES
Aungst BJ. Intestinal permeation enhancers. J Pharm Sci. 2000;89:429–42.
Swenson ES, Curatolo WJ. Intestinal permeability enhancement for proteins, peptides and other polar drugs: mechanisms and potential toxicity. Adv Drug Del Rev. 1992;8:39–92.
Hochman J, Artursson P. Mechanisms of absorption enhancement and tight junction regulation. J Contr Rel. 1994;29:253–67.
Fix JA. Strategies for delivery of peptides using absorption-enhancing agents. J Pharm Sci. 1996;85:1282–5.
Arbit E, Kidron M. Oral insulin: The rationale for this approach and current developments. J Diabet Sci Technol. 2009;3:562–7.
Burcham DL, Aungst BJ, Hussain M, Gorko MA, Quon CY, Huang S-M. The effect of absorption enhancers on the oral absorption of the GPIIb/IIIa receptor antagonist, DMP 728, in rats and dogs. Pharm Res. 1995;12:2065–70.
Aungst BJ, Saitoh H, Burcham DL, Huang S-M, Mousa SA, Hussain MA. Enhancement of the intestinal absorption of peptides and non-peptides. J Cont Rel. 1996;41:19–31.
Zhang H, Yao M, Morrison RA, Chong S. Commonly used surfactant, Tween 80, improves absorption of P-glycoprotein substrate, digoxin, in rats. Arch Pharm Res. 2003;26:768–72.
Guo A, Hu P, Balimane PV, Leibach FH, Sinko PJ. Interactions of a nonpeptidic drug, valacyclovir, with the human intestinal peptide transporter (hPEPT1) expressed in a mammalian cell line. J Pharmacol Exp Ther. 1999;289:448–54.
Kondoh M, Yagi K. Progress in absorption enhancers based on tight junction. Expert Opin Drug Deliv. 2007;4:275–86.
Johnson PH, Frank D, Costantino HR. Discovery of tight junction modulators: significance for drug development and delivery. Drug Disc Today. 2008;13:261–7.
Salama NN, Eddington ND, Fasano A. Tight junction modulation and its relationship to drug delivery. Adv Drug Del Rev. 2006;58:15–28.
Okuda T, Kadotsuji K, Takayama C, Hanada K, Mukaizawa F, Ogawara K-I, et al. Involvement of intracellular Ca2+ dynamics in cytoprotective action by amino acids and cytotoxicity by sodium laurate, an absorption enhancer. J Pharm Sci. 2006;95:2256–65.
Miller JM, Dahan A, Gupta D, Varghese S, Amidon GL. Enabling the intestinal absorption of highly polar antiviral agents: ion-pair facilitated membrane permeation of zanamivir heptyl ester and guanidine oseltamivir. Mol Pharm. 2010;7:1223–34.
Williams AC, Barry BW. Skin absorption enhancers. Crit Rev Ther Drug Carr Sys. 1992;9:305–53.
Francoeur ML, Golden GM, Potts RO. Oleic acid: its effects on stratum corneum in relation to (trans)dermal drug delivery. Pharm Res. 1990;7:621–7.
Aungst BJ, Blake JA, Hussain MA. Contributions of drug solubilization, partitioning, barrier disruption, and solvent permeation to the enhancement of skin permeation of various compounds with fatty acids and amines. Pharm Res. 1990;7:712–8.
Nicolazzo JA, Reed BL, Finnin BC. Buccal penetration enhancers—How do they really work? J Contr Rel. 2005;105:1–15.
Leary AC, Dowling M, Cussen K, O’Brein J, Stote RM. Pharmacokinetics and pharmacodynamics of intranasal insulin spray (Nasulin) administered to healthy male volunteers: Influence of the nasal cycle. J Diabet Sci Tech. 2008;2:1054–60.
Ding X, Rath P, Angelo R, Stringfellow T, Flanders E, Dinh S, et al. Oral absorption enhancement of cromolyn through noncovalent complexation. Pharm Res. 2004;21:2196–206.
Alani AWG, Robinson JR. Mechanistic understanding of oral drug absorption enhancement of cromolyn sodium by an amino acid derivative. Pharm Res. 2008;25:48–54.
Riley MGI, Castelli MC, Paehler EA. Subchronic oral toxicity of salcaprozate sodium (SNAC) in Sprague–Dawley and Wistar rats. Int J Toxicol. 2009;28:278–93.
Karsdal MA, Byrjalsen I, Riis BJ, Christiansen C. Optimizing bioavailability of oral administration of small peptides through pharmacokinetic and pharmacodynamic parameters: The effect of water and timing of meal intake on oral delivery of salmon calcitonin. BMC Clin Pharmacol. 2008;8. doi:10.1186/1472-6904-8-5.
Karsdal MA, Byrjalsen I, Henriksen K, Riis BJ, Lau EM, Arnold M, et al. The effect of oral salmon calcitonin delivered with 5-CNAC on bone and cartilage degradation in osteoarthritic parients: a 14 day randomized study. Osteoarthr Cartil. 2010;18:150–9.
Maher S, Leonard TW, Jacobsen J, Brayden DJ. Safety and efficacy of sodium caprate in promoting oral drug absorption: from in vitro to the clinic. Adv Drug Del Rev. 2009;61(15):1427–49.
Raoof AA, Chiu P, Ramtoola Z, Cumming IK, Teng C, Weinbach SP, et al. Oral bioavailability and multiple dose tolerability of an antisense oligonucleotide tablet formulated with sodium caprate. J Pharm Sci. 2004;93:1431–9.
Tillman LG, Geary RS, Hardee GE. Oral delivery of antisense oligonucleotides in man. J Pharm Sci. 2008;97:225–36.
Salama P, Mamluk R, Marom K, Weinstein I, Tzabari M. Pharmaceutical compositions and related methods of delivery. US Patent Appl. 2010/0105627 A1.
Eldor R, Kidron M, Arbit E. Open-label study to assess the safety and pharmacodynamics of five oral insulin formulations in healthy subjects. Diabet Obes Metab. 2010;12:219–23.
Kidron M. Methods and compositions for oral administration of proteins. US Patent Appl. 2011/0014247 A1.
Luzio SD, Dunseath G, Lockett A, Broke-Smith TP, New RR, Owens DR. The glucose lowering effect of an oral insulin (Capsulin) during an isoglycaemic clamp study in persons with type 2 diabetes. Diabet Obes Metab. 2010;12:82–7.
Bernstein G. Delivery of insulin to the buccal mucosa utilizing the RapidMist system. Expert Opin Drug Deliv. 2008;5:1047–55.
Modi P. Methods of administering and enhancing absorption of pharmaceutical agents. U.S. Patent No. 7,087,215 B2; 2008.
Crotts G, Ghebre-Sellassie I, Sheth A. Oral peptide pharmaceutical dosage form and method of production. US Patent 7,316,819; 2008.
Arnold JJ, Ahsan F, Meezah E, Pillion DJ. Correlation of tetradecylmaltoside induced increases in nasal peptide drug delivery with morphological changes in nasal epithelial cells. J Pharm Sci. 2004;93:2205–13.
Arnold JJ, Fyrberg MD, Meezan E, Pillion DJ. Reestablishment of the nasal permeability barrier to several peptides following exposure to the absorption enhancer tetradecyl-β-D-maltoside. J Pharm Sci. 2010;99:1912–20.
Maggio ET, Meezan E, Ghambeer DKS, Pillion DJ. Highly bioavailable nasal calcitonin—potential for expanded use in analgesia. Drug Del Tech. 2010;10:58–63.
Thanou M, Verhoef JC, Marbach P, Junginger HE. Intestinal absorption of octreotide: N-trimethyl chitosan chloride (TMC) ameliorates the permeability and absorption properties of the somatostatin analogue in vitro and in vivo. J Pharm Sci. 2000;89:951–7.
Vetter A, Martien R, Bernkop-Schnurch A. Thiolated polycarbophil as an adjuvant for permeation enhancement in nasal delivery of antisense oligonucleotides. J Pharm Sci. 2010;99:1427–39.
Wong O, Huntington J, Nishihata T, Rytting JH. New alkyl N, N-dialkyl-substituted amino acetates as transdermal penetration enhancers. Pharm Res. 1989;6:286–95.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Bruce Aungst and Craig K. Svensson
Rights and permissions
About this article
Cite this article
Aungst, B.J. Absorption Enhancers: Applications and Advances. AAPS J 14, 10–18 (2012). https://doi.org/10.1208/s12248-011-9307-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-011-9307-4