1bstract
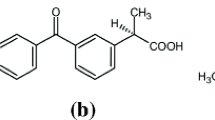
Purpose: The purpose of this study was to confirm the hypothesis that a site-II-to-site-I displacement takes place when some nonsteroidal anti-inflammatory drugs are displaced by another drug from their high-affinity binding site to a site of lower affinity on human serum albumin (HSA).Methods: Diclofenac, sodium salt, was used as a representative example because of its prominent reversal of the Cotton effect. Effects of site-specific drugs on the free fraction of diclofenac were determined by equilibrium dialysis, and effects on induced circular dichroism (CD) of diclifenac bound to HSA were studied by CD and CD simulation techniques.Results: Ibuprofen, a site-II-specific drug, altered the CD spectrum of the diclofenac-HSA complex at a molar ratio of 0.5∶1 to that obtained at a higher ratio (5∶1) without ibuprofen. The induced CD spectrum obtained in the presence of ibuprofen was very similar to one that assumed that all diclofenac displaced from its high-affinity binding site (site II) became rebound to a lower-affinity site (site I). The rebinding could be influenced by a free energy linkage between the two sites which would make site I (or parts thereof) more suitable for diclofenac binding.Conclusion: We have confirmed the existence of a site II-to-site displacement, which is very striking and pharmacologically important, because the concentration of unbound drug being displaced is much lower than expected for a competitive mechanism.
Similar content being viewed by others
Abbreviations
- HSA:
-
human serum albumin
- NSAID:
-
ironsteroidal anti-inflammatory drug
- 3D:
-
3ircular dichroism
- ED:
-
equilibrium dialysis
- 4NSA:
-
4ansyl-L-asparagine
- 4NSS:
-
4ansylsarcosine
References
Kober A, Sjöholm I. The binding sites on human serum albumin for some nonsteroidal antiinflammatory drugs. Mol Pharmacol. 1980: 18: 421–426.
Kragh-Hansen U. Molecular aspects of ligand binding to serum albumin. Pharmacol Rev. 1981: 33: 17–53.
Peters T. Jr. All about albumin. Biochemistry, genetics, and medical applications. Academic Press, San Diego. 1996.
Fehske KJ, Muller WE, Wollert U. The location of drug binding sites in human serum albumin. Biochem Pharmacol. 1981: 30: 687–692.
Rahman MH, Maruyama T, Okada T, Imai T, Otagiri M. Study of interaction of carprofen and its enantiomers with human serum albumin-II. Stereoselective site-to-site displacement of carprofen by ibuprofen. Biochem Pharmacol. 1993: 46: 1733–1740.
Chamouard J-M, Barre J, Urien S, Houin G, Tillement JP. Diclofenac binding to albumin and lipoproteins in human serum. Biochem Pharmacol. 1985; 34: 1695–1700.
Chen RF. Removal of fatty acids from serum albumin by charcoal treatment. J Biol Chem. 1967; 242: 173–183.
Rahman MH, Maruyama T, Okada T, Yamasaki K, Otagiri M. Study of interaction of carprofen and its enantiomers with human serum albumin-I Mechanism of binding studied by dialysis and spectroscopic methods. Biochem Pharmacol. 1993; 46: 1721–1731.
Yamaoka K, Tanigawara Y, Nakagawa T, Uno T. A pharmacokinetic analysis program (MULTI) for microcomputer. J Pharmacobio-Dyn. 1981; 4: 879–885.
Rahman MH, Yamasaki K, Shin Y-H, Lin CC, Otagiri M. Characterization of high affinity binding sites of nonsteroidal anti-inflammatory drugs with respect to site-specific probes on human serum albumin. Biol Pharm Bull. 1993; 16: 1169–1174
Tonn GR, Kerr CR, Axelson JE. In vitro protein binding of propafenone and 5-hydroxypropafenone in serum, in solutions of isolated serum proteins and to red blood cells. J Pharm Sci. 1992; 81: 1098–1103.
Otagiri M, Otagiri Y, Perrin JH. Some fluorescent investigation of the interaction between the enantioners of warfarin and human serum albumin. Int J Pharm. 1979; 2: 283–294.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamasaki, K., Rahman, M.H., Tsutsumi, Y. et al. 3ircular dichroism simulation shows a site-II-to-site-I displacement of human serum albumin-bound diclofenac by ibuprofen. AAPS PharmSciTech 1, 12 (2000). https://doi.org/10.1208/pt010212
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/pt010212