Abstract
Background
Multiple patient-related variables have been associated with reduced rates of survival to hospital discharge (SHD) after in-hospital cardiac arrest (IHCA). As opposed to most of these, anemia may be reversible. This retrospective single-center study aims to examine the relationship between prearrest hemoglobin levels, comorbidities, and survival after cardiopulmonary resuscitation (CPR) among patients with non-traumatic IHCA. Patients were classified as anemic (hemoglobin < 10 g/dL) or non-anemic (hemoglobin ≥ 10 g/dL) based on their lowest hemoglobin measurement in the 48 h preceding the arrest. The primary outcome was SHD. The secondary outcome was return of spontaneous circulation (ROSC).
Results
Of 1515 CPR reports screened, 773 patients were included. Half of the patients (50.5%, 390) were classified as anemic. Anemic patients had higher Charlson Comorbidity Indices (CCIs), less cardiac causes, and more metabolic causes for the arrest. An inverse association was found between CCI and lowest hemoglobin. Overall, 9.1% (70 patients) achieved SHD and 49.5% (383) achieved ROSC. Similar rates of SHD (7.3 vs. 10.7%, p = 0.118) and ROSC (49.5 vs. 51.0%, p = 0.688) were observed in anemic and non-anemic patients. These findings remained consistent after adjustment for comorbidities, in sensitivity analyses on the independent variable (i.e., hemoglobin) and on potential confounders and in subgroups based on sex or blood transfusion in the 72 h preceding the arrest.
Conclusions
Prearrest hemoglobin levels lower than 10 g/dL were not associated with lower rates of SHD or ROSC in IHCA patients after controlling for comorbidities. Further studies are required to confirm our findings and to establish whether post-arrest hemoglobin levels reflect the severity of the inflammatory post-resuscitation processes.
Similar content being viewed by others
Background
In-hospital cardiac arrest (IHCA) occurs in approximately 1.6:1000 hospital admissions [1]. The rates of survival from cardiac arrest are low (13.4% at 1 year) [2] and survivors have multiple complications [3, 4]. Multiple patient-related variables have been associated with reduced rates of survival to hospital discharge (SHD) after CPR (cardio-pulmonary resuscitation), including female sex [5], older age, comorbidities, and more [6]. As opposed to most of these patient-related variables, anemia may be reversible. However, it would require the use of blood products which may result in complications [7]. As a prospective study on this topic may be costly in terms of funding and potential complications, more data is required to determine whether such a study is justifiable in patients with cardiac arrest and if so, what cutoff values should be studied.
Few studies regarding the relationship between anemia and survival after CPR exist. These studies mostly associated lower hemoglobin levels with a lower likelihood of SHD [8, 9] and neurologically intact survival after CPR [8,9,10,11,12,13,14] (supplement 1). Correspondingly, anemia seems to be associated with poorer outcomes in various diseases states (e.g., heart failure [15], diabetes [16], malignancy [17]). Meanwhile, patients with an overall higher burden of disease, i.e., higher Charlson comorbidity score (CCI) [18, 19] and specific chronic diseases, e.g., heart failure, renal disease [20], and malignancy [19] have poorer survival outcomes after CPR. A conundrum therefore remains, whether the presence of anemia itself or the presence of comorbidities associated with anemia explains the association with poorer outcomes in anemic patients after CPR.
This study aimed to examine the relationship between prearrest hemoglobin levels and survival after CPR.
Methods
Study design
Ethical approval was waived for this single center study within the framework of a resuscitation quality assurance project (waiver P4.15, 18-Jan-2015). The findings of this cohort study are reported in accordance with STROBE (Strengthening the Reporting of Observational Studies) requirements [21].
Setting
We retrospectively analyzed observational data collected in real time over a period of 4 years (2016–2019) in a tertiary teaching hospital (the Shaare Zedek Medical Center, SZMC). The SZMC staff undergo routine periodic accreditation in CPR as required by the Joint Commission Resources (JCR). CPR is conducted in accordance with International Liaison Committee of Resuscitation (ILCOR) recommendations. Resuscitation events are mandatorily reported on a standardized electronic sheet embedded within the patient electronic medical records (EMRs) (supplement 2) which conforms to Utstein reporting recommendations.
Participants (eligibility criteria)
All IHCA reports for patients aged 18 years or over (Jan 2016–Dec 2019) were screened for inclusion. Reports of traumatic cardiac arrest, cardiac arrest during pregnancy or the peripartum period, and reports for patients without a single hemoglobin measurement in the 48 h before cardiac arrest were excluded. Also excluded were reports with hemoglobin measurements that did not pass logical testing (below 4 g/dL or above 20 g/dL) and reports of repeat resuscitations (supplement 3). Intraoperative arrests were a priori not included in this study.
Based on prior literature (section “Management of quantitative variables”) the patients were divided into two groups: anemic (hemoglobin < 10 g/dL) and non-anemic (hemoglobin ≥ 10 g/dL).
Study outcomes
The primary outcome was the association between prearrest anemia (< 10 g/dL) in the 48 h preceding the event and SHD before and after adjusting for background diseases. The secondary outcome was the association between prearrest anemia and ROSC before and after adjustment. We hypothesized that prearrest hemoglobin levels would be statistically related to unadjusted survival rates and that this association would disappear after adjustment for comorbidities.
Variables
Among other variables we collected, patient demographics (e.g., age, sex), burden of comorbidity (specific diseases, CCI), hemoglobin levels, and data on prearrest blood transfusion (supplement 4).
Because missing data are major concern in retrospective studies, we collected two values of hemoglobin, the last value before the arrest and the lowest value in the 48 h preceding the arrest, and used the value with the least amount of missing data is for the main analysis.
We analyzed hemoglobin in three manners: dichotomized to anemic and non-anemic patients (primary analysis), as a continuous variable and divided to tertiles (secondary analyses).
Method and sources of data collection
All data were extracted from the hospital CPR database and patient EMRs. The database algorithm collects data on patient comorbidities using the ICD-9 codes listed in the patients EMRs (supplement 5). For liver disease, renal failure, malignancy, and cerebrovascular disease, we determined disease severity—which is required for calculating the CCI [18]—case-by-case through manual evaluation of the full patient file. The data validation process is described in supplement 6.
Address of bias
Reporting bias is always a possibility; however, our overall rate of SHD is similar to that described elsewhere [2, 22]. Selection bias stemming from a greater likelihood of measuring hemoglobin in patients who were in a worse clinical condition was dealt with by studying hemoglobin levels both 24 and 48 h before the event. Patients with no relevant measurement comprised less than 10% of eligible cases even before reduction of repeat resuscitations (supplement 7). Hemoglobin is measured using three devices in our hospital; therefore, a systematic measurement bias may be possible. However, our devices undergo testing for miss-calibration in two manners; (1) monthly external quality assurance by the UK National External Quality Assessment Service and (2) daily internal quality assurance and harmonization between all three devices by the laboratory staff.
Sample size
We calculated the study power for the primary analysis—difference in survival among anemic versus non-anemic (< 10 g/dL vs ≥ 10 g/dL) patients. Based on preliminary evaluation of the existing database we assumed at least 350 patients in each group. We assumed a survival rate of 25% among non-anemic patients versus 10% among anemic patients based on previously reported survival rates [22] and odds ratios for survival [10]. With a two-sided significance level of 5%, the study power would be > 99%. We also calculated that the study power would be 96% with half the sample size and 83% with a lower difference in survival rates (8 vs 15%).
Management of quantitative variables
For the definition of anemia, we selected a-priori a cutoff of 10 g/dL based on previous studies that found an association between hemoglobin and CPR outcomes [8, 10, 11] and on the accepted maximal transfusion threshold for patients with cerebral and/or cardiac ischemia [23, 24]. The World Health Organization criteria for anemia differ for men and women [25]. We nonetheless chose a uniform threshold since no such differentiation is usually made for critically ill patients [8,9,10,11,12,13,14, 26].
We used a modified version CCI as follows: points for peptic ulcer disease were added for a proven diagnosis rather than based on receipt of therapy due to increased use of preventive treatment since the CCI was published [27]. In addition, points were not added for age. We examined age as a standalone variable since it is strongly related to both comorbidities and survival [28]. Furthermore, points for age were only added to the later iteration of the CCI [29]. Other than these two caveats, the score added per comorbidity adheres to the standard [18].
Statistical methods
We used descriptive statistics to assess demographics, comorbidities, and other patients’ characteristics. All proportions were calculated from the total number of patients (including those with missing data). We report all summary measures regardless of variable distribution.
Categorical variables were compared using the chi-square or Fisher’s exact tests, and continuous variables were compared using the T test or one-way analysis of variance, as required. Correlations were studied using the Pearson correlation coefficient. All p values were two-sided (when applicable), and confidence intervals were computed at the 95% confidence level.
Receiver operating characteristic (ROC) analysis was used to test the a priori chosen cutoff value used to define anemia (10 g/dL) against other possible cutoff values found using Liu’s method [30].
Multivariable logistic regression analysis (enter method) was used in order to simultaneously assess the effect of several independent factors on the dichotomous dependent outcomes (ROSC or SHD). The threshold for inclusion in the multivariable model was a p value ≤ 0.1. Multicollinearities were sought using clinical logic and confirmed using the chi-square test. In case of strong multicollinearity, we examined the effect of excluding each variable on the models and chose the models with the best goodness-of-fit. An initial shockable rhythm was highly correlated with shock delivery and coronary angiography was highly correlated with coronary reperfusion attempt. We therefore excluded shock delivery and coronary reperfusion attempt from the models.
In order to study model stability, we performed sensitivity analysis on the definition of the independent variable, on potential confounders and on post-CPR coronary angiography (in order to examine the effect of the treatment while retaining in the model patients that did not achieve ROSC).
We also conducted post hoc analyses to test for association between anemia and survival outcomes (SHD and ROSC) for three subgroups: males, females, and patients who had not received a blood transfusion within the 72 h before CPR.
All analyses were performed using SPSS version 26.0 (IBM corp., Armonk, NY, USA). [31].
Results
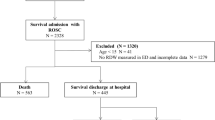
Overall, 1515 reports were screened. At the end of the inclusion–exclusion process 773 patients were included (Fig. 1). We hereby describe mostly results based on the lowest hemoglobin measurements (primary analysis). More results based on the last hemoglobin measurement are available in supplement 8.
Study inclusion exclusion flow chart. RRT rapid response team. OHCA out-of-hospital cardiac arrest. ROSC return of spontaneous circulation. 1Ascertained either by manual evaluation or by a statement in the electronic form that cardiac arrest had not occurred and no evidence of chest compressions was found. 2No information entered to the electronic form by the on-site medical team and therefore could not be verified. 3Ascertained by excluding events reported from the delivery room, maternity wards, or department of gynecology. 4Ascertained by manual review of cases reported from the ED, general surgery, operating theatre, postoperative care unit, or any location in the imaging. 5In case of repeat resuscitations of a single patient, only the first arrest was included. 6Hemoglobin measurements lower than 4 g/dL or higher than 20 g/dL
Cohort characteristics
The mean age was 76.58 ± 14.21 years and 55.8% were male. The mean CCI was 2.07 ± 1.91. The mean lowest hemoglobin level was 10.27 ± 2.31 g/dL (median 9.9 g/dL, IQR 8.5–11.8 g/dL, range 4.6–19.2 g/dL). Lowest hemoglobin tertiles cutoffs were 1st: ≤ 8.9 g/dL, 2nd: > 9.0 g/dL to ≤ 11.1 g/dL, and 3rd: > 11.1 g/dL. For hemoglobin distribution, see supplement 9. 49.5% (383) achieved ROSC and 9.1% (70) survived to hospital discharge (Tables 1 and 2).
Based on the lowest hemoglobin measurement, half of the patients (50.5%, n = 390) were classified as anemic. Anemic patients had a higher prevalence of diabetes, renal failure, solid tumors and hematologic malignancies, and a lower prevalence of dementia and acute CVA. Anemic patients had a higher overall burden of disease (i.e., higher CCIs) and different causes for arrest. More anemic patients received a blood transfusion in the 72 h preceding the arrest. Less anemic patients had an initial shockable rhythm and anemic patients received less adrenaline (epinephrine). Less anemic patients underwent coronary angiography and a coronary reperfusion attempt after CPR (Tables 1 and 2).
Lowest hemoglobin and Charlson comorbidity index
An inverse association was found between CCI and lowest hemoglobin; heat maps of the relationship between CCI and hemoglobin show that as CCIs rise, hemoglobin measurements are lower (Fig. 2).
We studied this association in three manners: First by comparing mean CCI in anemic and non-anemic patients (2.28 ± 1.96 vs 1.86 ± 1.84, p = 0.002), then by comparing the mean CCI in three groups of hemoglobin tertiles (1st: 2.32 ± 1.96, 2nd: 2.02 ± 1.86, and 3rd: 1.87 ± 1.89, p = 0.022) and finally, using CCI and hemoglobin as continuous variables (p value 0.005, Pearson’s r − 0.100) (supplement 10a).
Survival to hospital discharge
ROC analysis
ROC analysis (Fig. 3) highlighted a cutoff of 10.35 g/dL with sensitivity and specificity of 0.557 and 0.570 compared to 0.571 and 0.526 for 10 g/dL. Due to the comparability between the cut-offs, we proceeded with the a priori chosen cutoff (10 g/dL).
Univariable analysis
No unadjusted association was found between the lowest hemoglobin and SHD. We studied this association in three manners: First by comparing survival rates in anemic vs. non-anemic patients (7.7 vs 10.4%, p = 0.183) (primary outcome), then by comparing survival rates in the three hemoglobin tertiles (1st: 6.5%, 2nd: 9.7%, and 3rd: 11.1%, p = 0.171) and by comparing hemoglobin levels in survivors and non-survivors (10.8 ± 2.4 g/dL vs 10.2 ± 2.3 g/dL, p = 0.055) (supplement 11).
We did find an unadjusted association between last hemoglobin and SHD (11.2 ± 2.5 g/dL in survivors vs 10.6 ± 2.3 g/dL in non-survivors, p = 0.042) (supplement 8).
Other factors found associated with decreased SHD in univariable analysis were older age, solid tumors, higher CCI, initial non-shockable rhythm, no shock administration (not included in the models), higher adrenaline dose, and no post-CPR coronary angiography or reperfusion attempt (also not included) (supplements 11,12).
Multivariable analysis
The primary multivariable analysis included 348 patients. CCI, an initial shockable rhythm, lower adrenaline dose and coronary angiography remained associated with SHD in the adjusted analysis. Neither age nor anemia based on the lowest hemoglobin (adjusted OR: 0.701, CI: 0.363–1.352, p = 0.289) were associated with SHD in the adjusted analysis (Table 3, model 1).
Anemia based on the last hemoglobin (adjusted OR 0.605, CI 0.294–1.245, p = 0.172) was also not associated with SHD in the adjusted analysis (supplement 8).
Sensitivity analyses
Sensitivity analysis on the independent variable (hemoglobin) as hemoglobin levels of survivors and non-survivors and hemoglobin tertiles showed no adjusted association with SHD.
Sensitivity analysis on the potential confounders (comorbidities) was performed by entering individual comorbidities that showed an unadjusted association with SHD into the multivariable analysis. Hence, solid tumors were entered. No adjusted association was seen between anemia and SHD.
Sensitivity analysis including patients that did not achieve ROSC showed no adjusted association between anemia and SHD (Table 3, model 2).
These results were consistent in a secondary analysis, based on last hemoglobin measurements (supplement 13).
Subgroup analyses
We found no unadjusted association between anemia and SHD in males, females, or patients that did not receive blood transfusion in the 72 h preceding their arrest (supplement 14).
ROSC
No unadjusted association was found between the lowest hemoglobin and ROSC when comparing ROSC rates in anemic and non-anemic patients (50.0 vs 49.1%, p = 0.799) (Table 1).
Anemia based on the lowest hemoglobin (adjusted OR 1.040, CI 0.746–1.450, p = 0.816) and CCI were not associated with ROSC after adjusting for age, blood transfusion, witnessed event, initial rhythm, and adrenaline dose (supplement 15).
Discussion
This study showed that hemoglobin levels below 10 g/dL prior to cardiac arrest were not associated with lower rates of survival to hospital discharge or ROSC in IHCA patients. These results were consistent after controlling for comorbidities and in subgroup analyses done by sex and the subgroup of patients that had no blood transfusion prior to the arrest. Higher hemoglobin levels were found in survivors to hospital discharge; however, this association disappeared after controlling for comorbidities (i.e., CCI), demographics (i.e., age), event-related factors (i.e., initial shockable rhythm and adrenaline dose), and post-CPR treatments (i.e., coronary angiography after CPR). The consistency of the results, in addition to power of the study as calculated by the sample size calculation and previously reported proportions [10], should establish that such association is at the very least diminished in the setting of IHCA.
This study is unique due to its focus on IHCA patients and the measurement of pre-arrest hemoglobin levels. Moreover, the array of confounders studied and our approach to data analysis are also unique when comparing to the current literature on the topic.
The proportion of males, mean age, rates of initial shockable rhythm, and ROSC rates in our study are comparable to those in other studies on IHCA patients [1]. We also found similar comorbidity rates (e.g., diabetes mellitus, myocardial infarction, heart failure, acute CVA) to those described in other studies of IHCA patients [9, 10]. Although we studied pre-arrest hemoglobin levels, we found hemoglobin levels similar to those described in studies of post-ROSC hemoglobin in IHCA [9, 10]. The survival rates in our study are within the range described in other studies on IHCA patients [2, 22].
Our findings differ from those of prior studies on this topic in several aspects. Previous studies showed an association between hemoglobin and cardiac arrest outcomes even after controlling for comorbidities [8,9,10, 13, 14]. Our focus on IHCA patients may be one reason for this difference. Several previous studies controlled for individual comorbidities [8,9,10, 13, 14] and others did not control for any [11, 12]. We controlled for CCI which better represents the burden of comorbidity than individual comorbidities and also studied individual comorbidities separately. We included in the univariable analysis, patients that underwent CPR regardless of their ROSC status and performed a sensitivity analysis of the multivariable models by alternately excluding and including patients that did not achieve ROSC. Previous studies only excluded patients that did not achieve ROSC [8,9,10,11,12,13,14] and post-ROSC hemoglobin may be related to the severity of post cardiac arrest syndrome. Few patients received TTM in our study, most likely due to its controversial use in IHCA [32].
The results of our study support the findings of a recent study that suggested that in this context hemoglobin may act as a marker of other systemic processes; this study showed an association between increasing hemoglobin levels after cardiac arrest and worse outcomes [33]. The authors theorized that hemoglobin concentration may serve as a marker for the vascular permeability caused by post cardiac-arrest inflammation. If so, changes in hemoglobin levels may indicate the severity of the ongoing acute processes and a single value may be unrelated to survival despite its role in oxygen delivering capacity, as proposed in previous studies [14].
Our study has several limitations. The study was performed in a single center and may therefore not be generalizable. However, we show a similar case mix in terms of age [1] and background diseases [9, 10] as described in other studies on IHCA. Our data is highly dependent on the coding of ICD-9 diagnoses in the electronic medical charts. However, part of the work done for the purpose of this study was a quality assurance process to ensure code correctness. We present no data on neurological or functional outcomes yet these are considered the most meaningful outcomes after CPR. This decision stemmed from the small number of neurologically intact survivors expected in this single center and some lack of data.
In conclusion, pre-arrest hemoglobin levels lower than 10 g/dL were not associated with lower rates of survival to hospital discharge or ROSC in IHCA patients both before and after controlling for comorbidities. In this study, this finding is supported in both the dichotomous analysis and the continuous analysis (i.e., the ROC curve), suggesting that setting a simple numeric threshold for treatment of hemoglobin levels may not be relevant in this patient population. Our findings need corroboration in additional cohorts of IHCA patients. Studies seeking an association between anemia and neurological outcome should also be conducted in these patients. Finally, both laboratory and human studies are required to establish whether post resuscitation hemoglobin concentration is indeed a marker for the severity of vascular permeability caused by the severity of post cardiac-arrest inflammation.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CCI:
-
Charlson comorbidity indices
- CPR:
-
Cardio pulmonary resuscitation
- ICD-9:
-
International Classification of Disease ninth revision
- IHCA:
-
Intra hospital cardiac arrest
- ILCOR:
-
International Liaison Committee of Resuscitation
- JCR:
-
Joint commission resources
- ROSC:
-
Return of spontaneous circulation
- SHD:
-
Survival to hospital discharge
- STROBE:
-
Strengthening the Reporting of Observational Studies
- SZMC:
-
Shaare Zedek Medical Center
- TTM:
-
Targeted temperature management
References
Nolan JP, Soar J, Smith GB, Gwinnutt C, Parrott F, Power S et al (2014) Incidence and outcome of in-hospital cardiac arrest in the United Kingdom National Cardiac Arrest Audit. Resuscitation 85(8):987–992. https://doi.org/10.1016/j.resuscitation.2014.04.002
Schluep M, Gravesteijn BY, Stolker RJ, Endeman H, Hoeks SE (2018) One-year survival after in-hospital cardiac arrest: a systematic review and meta-analysis. Resuscitation 132:90–100. https://doi.org/10.1016/j.resuscitation.2018.09.001
Neumar RW, Nolan JP, Adrie C, Aibiki M, Berg RA, Bottiger BW et al (2008) Post-cardiac arrest syndrome: epidemiology, pathophysiology, treatment, and prognostication. A consensus statement from the International Liaison Committee on Resuscitation (American Heart Association, Australian and New Zealand Council on Resuscitation, European Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Council of Asia, and the Resuscitation Council of Southern Africa); the American Heart Association Emergency Cardiovascular Care Committee; the Council on Cardiovascular Surgery and Anesthesia; the Council on Cardiopulmonary, Perioperative, and Critical Care; the Council on Clinical Cardiology; and the Stroke Council. Circulation 118(23):2452–2483. https://doi.org/10.1161/CIRCULATIONAHA.108.190652
Mongardon N, Dumas F, Ricome S, Grimaldi D, Hissem T, Pene F et al (2011) Postcardiac arrest syndrome: from immediate resuscitation to long-term outcome. Ann Intensive Care 1(1):45. https://doi.org/10.1186/2110-5820-1-45
Helviz Y, Ong M, Einav S (2019) Cardiac arrest, gender and resuscitation outcomes. Intensive Care Med 45(2):278–281. https://doi.org/10.1007/s00134-018-5487-z
Fernando SM, Tran A, Cheng W, Rochwerg B, Taljaard M, Vaillancourt C et al (2019) Pre-arrest and intra-arrest prognostic factors associated with survival after in-hospital cardiac arrest: systematic review and meta-analysis. BMJ 367:l6373. https://doi.org/10.1136/bmj.l6373
Janssen MP, van Tilborgh AJW, de Vooght KMK, Bokhorst AG, Wiersum-Osselton JC (2018) Direct costs of transfusion reactions - an expert judgement approach. Vox Sang 113(2):143–151. https://doi.org/10.1111/vox.12614
Kim D, Kim SH, Park KN, Oh SH, Kim YM, Youn CS (2018) Hemoglobin concentration is associated with neurologic outcome after cardiac arrest in patients treated with targeted temperature management. Clin Exp Emerg Med 5(3):150–155. https://doi.org/10.15441/ceem.17.250
Wormsbecker A, Sekhon MS, Griesdale DE, Wiskar K, Rush B (2017) The association between anemia and neurological outcome in hypoxic ischemic brain injury after cardiac arrest. Resuscitation 112:11–16. https://doi.org/10.1016/j.resuscitation.2016.12.010
Johnson NJ, Rosselot B, Perman SM, Dodampahala K, Goyal M, Gaieski DF et al (2016) The association between hemoglobin concentration and neurologic outcome after cardiac arrest. J Crit Care 36:218–222. https://doi.org/10.1016/j.jcrc.2016.07.012
Albaeni A, Eid SM, Akinyele B, Kurup LN, Vaidya D, Chandra-Strobos N (2016) The association between post resuscitation hemoglobin level and survival with good neurological outcome following Out Of Hospital cardiac arrest. Resuscitation 99:7–12. https://doi.org/10.1016/j.resuscitation.2015.11.015
Hayashida K, Nishiyama K, Suzuki M, Abe T, Orita T, Ito N et al (2014) Estimated cerebral oxyhemoglobin as a useful indicator of neuroprotection in patients with post-cardiac arrest syndrome: a prospective, multicenter observational study. Crit Care 18(4):500. https://doi.org/10.1186/s13054-014-0500-6
group, S.-K. s. (2012) Relationship between the hemoglobin level at hospital arrival and post-cardiac arrest neurologic outcome. Am J Emerg Med 30(5):770–774. https://doi.org/10.1016/j.ajem.2011.03.031
Wang CH, Huang CH, Chang WT, Tsai MS, Yu PH, Wang AY et al (2016) Association between hemoglobin levels and clinical outcomes in adult patients after in-hospital cardiac arrest: a retrospective cohort study. Intern Emerg Med 11(5):727–736. https://doi.org/10.1007/s11739-015-1386-2
Groenveld HF, Januzzi JL, Damman K, van Wijngaarden J, Hillege HL, van Veldhuisen DJ et al (2008) Anemia and mortality in heart failure patients a systematic review and meta-analysis. J Am Coll Cardiol 52(10):818–827. https://doi.org/10.1016/j.jacc.2008.04.061
Gauci R, Hunter M, Bruce DG, Davis WA, Davis TME (2017) Anemia complicating type 2 diabetes: prevalence, risk factors and prognosis. J Diabetes Complications 31(7):1169–1174. https://doi.org/10.1016/j.jdiacomp.2017.04.002
Caro JJ, Salas M, Ward A, Goss G (2001) Anemia as an independent prognostic factor for survival in patients with cancer: a systemic, quantitative review. Cancer 91(12):2214–2221
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383. https://doi.org/10.1016/0021-9681(87)90171-8
Piscator E, Hedberg P, Goransson K, Djarv T (2016) Survival after in-hospital cardiac arrest is highly associated with the age-combined Charlson Co-morbidity Index in a cohort study from a two-site Swedish University hospital. Resuscitation 99:79–83. https://doi.org/10.1016/j.resuscitation.2015.11.023
Andrew E, Nehme Z, Bernard S, Smith K (2017) The influence of comorbidity on survival and long-term outcomes after out-of-hospital cardiac arrest. Resuscitation 110:42–47. https://doi.org/10.1016/j.resuscitation.2016.10.018
Erik von E, strobe@ispm.unibe.ch, Institute of S, Preventive Medicine, U. o. B. B. S, Douglas GA, Centre for Statistics in Medicine, U. o. O. O. U. K., et al. (2007). The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. The Lancet 370(9596):1453-1457 https://doi.org/10.1016/S0140-6736(07)61602-X
Zhu A, Zhang J (2016) Meta-analysis of outcomes of the 2005 and 2010 cardiopulmonary resuscitation guidelines for adults with in-hospital cardiac arrest. Am J Emerg Med 34(6):1133–1139. https://doi.org/10.1016/j.ajem.2016.03.008
Vlaar AP, Oczkowski S, de Bruin S, Wijnberge M, Antonelli M, Aubron C et al (2020) Transfusion strategies in non-bleeding critically ill adults: a clinical practice guideline from the European Society of Intensive Care Medicine. Intensive Care Med 46(4):673–696. https://doi.org/10.1007/s00134-019-05884-8
Retter A, Wyncoll D, Pearse R, Carson D, McKechnie S, Stanworth S et al (2013) Guidelines on the management of anaemia and red cell transfusion in adult critically ill patients. Br J Haematol 160(4):445–464. https://doi.org/10.1111/bjh.12143
Organisation WH (2011) Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity. https://apps.who.int/iris/rest/bitstreams/1161313/retrieve. Accessed 07/10/2022 2022
Holst LB, Petersen MW, Haase N, Perner A, Wetterslev J (2015) Restrictive versus liberal transfusion strategy for red blood cell transfusion: systematic review of randomised trials with meta-analysis and trial sequential analysis. BMJ 350:h1354. https://doi.org/10.1136/bmj.h1354
Mishuk AU, Chen L, Gaillard P, Westrick S, Hansen RA, Qian J (2020) National trends in prescription proton pump inhibitor use and expenditure in the United States in 2002–2017. J Am Pharm Assoc 61(2003):87. https://doi.org/10.1016/j.japh.2020.09.015
Einav S, Cortegiani A, Marcus EL (2021) Cardiac arrest in older adult patients. Curr Opin Anaesthesiol 34(1):40–47. https://doi.org/10.1097/ACO.0000000000000942
Charlson M, Szatrowski TP, Peterson J, Gold J (1994) Validation of a combined comorbidity index. J Clin Epidemiol 47(11):1245–1251. https://doi.org/10.1016/0895-4356(94)90129-5
Liu X (2012) Classification accuracy and cut point selection. Stat Med 31(23):2676–2686. https://doi.org/10.1002/sim.4509
IBM (2019) IBM SPSS statistics for Windows
Chan PS, Berg RA, Tang Y, Curtis LH, Spertus JA, With AHAG, the Guidelines-Resuscitation, I. (2016) Association between therapeutic hypothermia and survival after in-hospital cardiac arrest. JAMA 316(13):1375–1382. https://doi.org/10.1001/jama.2016.14380
Schriefl C, Schoergenhofer C, Ettl F, Poppe M, Clodi C, Mueller M, Grafeneder J, Jilma B, Magnet IAM, Buchtele N, Boegl MS, Holzer M, Sterz F and Schwameis M (2021) Change of hemoglobin levels in the early post-cardiac arrest phase is associated with outcome. Front Med (Lausanne) 8:639803. https://doi.org/10.3389/fmed.2021.639803
Acknowledgements
We would like to express our sincere gratitude to Tali Bdolah-Abram, MSc, for statistical support and to our research nurse Avital Lifschitz, BSN, for her assistance with IRB submission.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Funding
No funding was received for conducting this research.
Author information
Authors and Affiliations
Contributions
LS: methodology, software, validation, formal analysis, investigation, data curation, writing–original draft, visualization, and project administration.
YH: manuscript preparation and critical review.
SE: conceptualization, methodology, software, validation, resources, data curation, writing- review and editing, supervision, and project administration.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Ethical approval was waived for this single-center study within the framework of a resuscitation quality assurance project (waiver SZMC P4.15, 18-Jan-2015).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shor, L., Helviz, Y. & Einav, S. Anemia before in-hospital cardiac arrest and survival from cardio-pulmonary resuscitation—a retrospective cohort study. J Anesth Analg Crit Care 2, 51 (2022). https://doi.org/10.1186/s44158-022-00080-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s44158-022-00080-5