Abstract
Background
Hospital-acquired pneumonia (HAP) and ventilator-associated pneumonia (VAP) represent a major health problem among hospitalized patients leading to consequent morbidity and mortality specially after Covid-19 era and spread of multidrug-resistance organisms)MDRO) in hospitals.
Aim
This study aimed to analyze the commonest microorganisms responsible for HAP and VAP at Tanta University Chest Hospital.
Methods
This prospective observational study was done at Chest Department, Faculty of Medicine, started from June 2022 to February 2023. Fifty HAP patients’ sputum samples and 50 VAP patients (25 endotracheal aspirates and 25 bronchoalveolar lavages) were included. All collected samples were submitted to standard microbiological tests at Microbiology Department, Tanta Faculty of Medicine.
Results
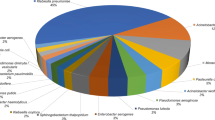
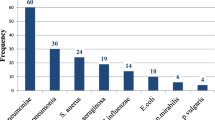
A total number of 50 HAP and 50 VAP cases were included. Microbial isolates were relatively the same in both groups, where Klebsiella pneumoniae was the predominant isolates (56) followed by Staphylococcus aureus (25), Escherichia coli (14), Pseudomonas aeruginosa (13), Acinetobacter baumannii (5), Streptococcus pneumoniae (4), Enterococci (3), Stenotrophomonas maltophilia (2), Citrobacter freundii (2), Streptococcus pyogenes (2), Providencia stuartii (1), and 7 isolates of Candida. Antimicrobial susceptibility showed predominance of drug-resistance organisms in VAP (78%) versus HAP (28%), P-value: < 0.001. Klebsiella pneumonia showed higher rates of resistance (37 out of 56 isolates, P-value = 0.020). Four out of 5 Acinetobacter baumannii isolates were multidrug resistant, and 2 isolates of Stenotrophomonas maltophilia were polydrug resistant. Multivariate logistic regression analysis showed that intravenous antibiotic administration within last 3 months was associated with risk for MDR-HAP, while univariate regression analysis showed that intravenous antibiotic administration within the last 3 months was the strongest predictor for MDR-VAP. A higher mortality rate was recorded in VAP group (66%), versus (14%) in HAP group (P-value: < 0.001).
Conclusion
Microbial isolates were relatively the same in both HAP and VAP, but VAP showed significant predominance of drug-resistant organisms. The most significant predictor for risk of MDRO infection in HAP and VAP was intravenous antibiotic administration within last 3 months; so, raising attention towards antibiotic choice may improve the outcome. Finally, VAP was associated with higher mortality compared to HAP.
Similar content being viewed by others
Introduction
Nosocomial pneumonia is a major health concern that represents about 22% of hospital infections worldwide [1]. Nosocomial pneumonia is a term that describes hospital-acquired pneumonia (HAP) and ventilator-acquired pneumonia (VAP). HAP is defined as pneumonia which is developed at least 48 h after hospital admission either in intensive care unit or in hospital wards, while VAP describes pneumonia which is developed after 48 h on mechanical ventilation [2]. Diagnosis of nosocomial pneumonia includes different approaches including clinical examination, laboratory investigations, and radiological assessment. Furthermore, microbiological assessment of nosocomial pneumonia is essential for proper management. It is known that early treatment of nosocomial pneumonia utilizing empirical antibiotics is advised to improve the clinical outcome until cultures of the causative organisms become available [3]. Hence, microbiological analysis in different healthcare facilities is mandatory for recognition of the commonest pathogens and targeting those pathogens with empirical antibiotic regimen according to the collected data [4].
Patients and methods
Patient selection
Study protocol was approved from Ethical Committee of Faculty of Medicine, Tanta University, with approval code (22/5/35500), and informed consents for procedure were obtained from the patients or their relatives. This prospective observational study was carried out at Chest Department, Tanta Faculty of Medicine, within 9 months: from June 2022 to February 2023. This study included 50 patients diagnosed as hospital-acquired pneumonia defined as follows: pneumonia developing 48 h after hospital admission and not incubating at admission time, classified as early-onset HAP which developed 2–4 days after admission, and late-onset HAP which developed ≥ 5 days after hospitalization. Also, this study included 50 cases diagnosed as ventilator-associated pneumonia, defined as pneumonia developing in a patient who has received mechanical ventilation for ≥ 48 h, classified as early-onset VAP which developed 2–4 days after intubation, and late-onset VAP which developed ≥ 5 days after intubation. Criteria for diagnosis of both HAP and VAP depend on the presence of new pulmonary infiltrates acquired in hospital with two or more of the following: temperature > 38.3 °C < or 36 °C, leukocytic count > 10 × 109 /L or < 4 × 109 /L, and the presence of purulent respiratory tract secretions [3]. Patients with community-acquired pneumonia (CAP) and severe immunocompromised patients including chemotherapy, HIV, and organ transplant were excluded as well as patients who refused to participate in the study. All included patients were subjected to history taking, physical examination, laboratory investigations including complete blood count (CBC), C-reactive protein (CRP), D-dimer, liver function tests, blood urea and serum creatinine, INR as well as radiological investigations, and chest X-ray (CXR) done for all cases, while computerized tomography (CT) chest scan was requested for some HAP patients when CXR was apparently normal in the presence of suggestive clinical and laboratory findings of nosocomial pneumonia. Regarding 50 included HAP patients, sputum samples were taken, while 50 included VAP cases; 25 patients who were unfit for bronchoscopy were sampled by endotracheal aspirate (ETA), and another 25 patients who were fit for bronchoscopy were sampled by bronchoalveolar lavage (BAL). All collected samples (sputum, BAL, ETA) were sent to Medical Microbiology and Immunology Department, for subsequent standard microbiological cultures and examination.
Technique of the procedure
Collection of sputum samples
The patients were provided with sterile specimen container with instructing the patient not to touch the inside of the container; they were asked to take a slow deep breath and to cough after a full inspiration and then expectorate sputum directly into the sterile container. This maneuver was repeated until a sufficient amount at least 5 mL of sputum was collected. The top of the specimen container was secured tightly and lastly; any sputum present on the outside of the container was removed with a disinfectant wipe. Saliva samples were excluded [5].
Collection ETA samples
ETA was performed for mechanically ventilated patients who were unfit for bronchoscopy by intensive care unit (ICU) residents with assistance of nursing staff. First, the patient was hyperoxygenated with 100% FIO2 (fraction of inspired oxygen) for 1 min, then a 50-cm suction catheter (16 French) was inserted through the endotracheal tube (ETT) or tracheostomy tube without suctioning until resistance was met and then was withdrawn, and suction was done in an interrupted manner with 1-min interval between performed 5 insertions according to the recommendations of the latest endotracheal suctioning directives of the American Association for Respiratory Care (AARC) [6]. Finally, aspirates were collected in a sterile container to be sent for microbiological analysis as quickly as possible.
Collection of BAL samples
For mechanically ventilated patients who were fit for bronchoscopy, BAL was performed by pulmonology consultant with assistance of nursing staff. First, the patient was hyperoxygenated with FIO2 100% for 1 min, and then a fiber-optic bronchoscope (KARL STORZ, Germany; model 11004BC1, SN 2252850) was introduced through ETT (8 mm) or tracheostomy tube (8 mm) and then wedged into the targeted segmental bronchi. Sterile saline (0.9% NaCl at room temperature) was instilled in four aliquots of 50 mL and then rapidly aspirated, and recovered BAL fluid samples were collected and transported as quickly as possible for microbiological studies [7].
Handling of the specimens
The collected samples were transferred within 1 to 2 h to Medical Microbiology and Immunology Department, Faculty of Medicine, Tanta University. A full microbiological examination was done to identify the isolated pathogens from different sample types and then stored at 4 °C. Sputum was considered microbiologically adequate only if it contained > 10 white blood cells for each epithelial cell at a magnification of × 400 [8]. The purulent ETA secretions were defined as lower respiratory tract samples that contained ≥ 25 neutrophils and ≤ 10 squamous epithelial cells per lower power field 100 × . The threshold values of 105 colony-forming units (CFU/mL) for ETA and 106 for sputum cultures were taken as positive cultures, and values below the threshold were considered as colonization [9, 10]. For BAL fluid, if BAL recovered volume was less than 20 mL, total cell count was less than 60,000 cells/mL; if prepared samples showed excessive amounts of intracellular debris or damaged nucleated cells or more than 1% squamous epithelial cell, BAL was rejected [7]. The threshold values of 104 CFU/mL for BAL were taken as a positive BAL culture [11].
Samples processing and culture
Samples were vortexed for 1 min and cultured on MacConkey’s blood agar, chocolate agar, and Sabaraud dextrose agar. Blood and chocolate agars were incubated in candle jar overnight at 37 °C incubator. Identification of isolated colonies was done by gram staining and different biochemical tests such as catalase test, coagulase test, oxidase test, urease test, triple sugar iron agar, citrate, and indole tests [12].
Antimicrobial susceptibility tests were done on Muller-Hinton agar plates by Kirby-Bauer disc diffusion plate method. The antibiotic disc used was amikacin (30 μg), amoxicillin/clavulanic acid (30 μg), aztreonam (30 μg), ceftriaxone (30 μg), ceftazidime (30 μg), cefoxitin (30 μg), cefepime (30 μg), cefoperazone (75 µg), clindamycin (2 μg), erythromycin (15 μg), imipenem (10 μg), meropenem (10 µg), gentamicin (10 μg), nitrofurantoin (300 µg), colistin (10 μg), tetracycline (30 μg), tigecycline (15 μg), teicoplanin (30 μg), levofloxacin (5 μg), ciprofloxacin (5 µg), linezolid (30 μg), penicillin G (10 units), piperacillin/tazobactam (110 μg), trimethoprim/sulfamethoxazole (1.25/23.75 μg), and vancomycin (30 μg) (Oxoid, UK). Extended-spectrum beta-lactamase (ESβL) production was tested by phenotypic confirmatory tests, and the interpretation of the results was done according to the CLSI (Clinical and Laboratory Standards Institute, 2022) [13]. The pattern of antibiotic resistance of organisms was defined according to the guidelines suggested by the European Center for Disease Control and Prevention (ECDC). Multidrug resistant (MDR) was defined as acquired nonsusceptibility to at least one agent in three or more antimicrobial categories. Extensively drug resistant (XDR) was defined as nonsusceptibility to at least one agent in all but two or fewer antimicrobial categories (i.e., bacterial isolates remain susceptible to only one or two antimicrobial categories). Pan-drug resistant (PDR) was defined as nonsusceptibility to all agents in all antimicrobial categories [14].
Statistical analysis
Statistical analysis
Statistical analysis was conducted using (SPSS version 20). Descriptive statistics for quantitative data were done using mean, standard deviation, and minimum and maximum of the range, while qualitative data were represented as number and percentage. Continuous variables were compared using Student’s t-test, and categorical variables were compared using Pearson’s chi-square. P-value < 0.05 is considered significant. Simple logistic regression analysis was performed to determine the predictors for risk of multidrug-resistant organisms in nosocomial pneumonia. These were reported as the P-value (level of significance, P-value ≤ 0.05) and odds ratio (OR).
Results
Demographic data of the patients were listed in Table 1 where mean ± SD age in HAP and VAP groups was 54.520 ± 13.762 with median 55 years and 58.940 ± 18.008 with median 64 years respectively, number of males in HAP was 27 versus 26 in VAP, while females were 23 versus 24 patients in VAP group with no statistically significant difference between both groups regarding age and gender.
Among laboratory investigations, CRP and D-dimer showed higher values in VAP than HAP group with significant difference between both groups (P-value: < 0.001 for both). Regarding the cause of hospital admission, variable causes showed a significant difference between both groups (P-value: 0.009, listed in Table 2). Among those acute exacerbation of chronic obstructive pulmonary diseases (AE. COPD) was the predominant cause in both groups followed by AE. asthma and HP in HAP group versus acute exacerbation of idiopathic pulmonary fibrosis (AE. IPF) and bronchiectasis and obesity hypoventilation syndrome in VAP group. Furthermore, radiological findings showed significant difference between both groups (P-value: 0.037) with predominance of lobar pneumonia in HAP (40%), while bronchopneumonia was the predominant (52%) in VAP group, while onset of pneumonia did not show any significant difference between both groups (Table 2). All VAP patients were managed in ICU, while only 15 patients of HAP group were managed in ICU, and the remaining 35 patients were managed in wards.
Regarding microbiological results, among HAP group, thirty-three patients (66%) had one organism in their cultures versus thirty-five patients (70%) in VAP group, and 17 patients (34%) had two organisms in their cultures in HAP versus 15 patients (30%) in VAP group with no significant difference between both groups. Klebsiella pneumoniae was the most predominant organism recorded in both groups: 20 isolates (40%) in HAP group versus 36 isolates (72%) in VAP group with a significant difference between both groups (P-value: 0.001). In HAP group, Staph. aureus was 16 isolates (32%) and E. coli 8 isolates (16%); meanwhile, in VAP group, Staph. aureus was 9 isolates (18%) and pseudomonas aeruginosa 7 isolates (14%). Acinetobacter baumannii was recorded only in VAP group: 5 patients (10%) with a significant difference between both groups (P-value: 0.022). On the other hand, Candida was recorded only in HAP group: 7 patients (14%) (P-value: 0.006) (Table 3).
Antimicrobial sensitivity tests showed a significant difference between both groups (P-value: < 0.001) with predominance of susceptible organisms in HAP group (72%), while resistance was evident in VAP group (78%). Furthermore, the distribution of antimicrobial susceptibility showed a significant difference between both groups (P-value: < 0.001) (Table 3).
The distribution of organisms according to antimicrobial susceptibility test was listed in Table 4. In gram-negative organisms, among 56 isolated Klebsiella pneumoniae, 19 isolates were sensitive to standard antibiotics regimens, while 26 isolates were MDR, 8 were XDR, and 3 were PDR isolates with a predominance of MDR Klebsiella among hospitalized patients (P-value: 0.020). Among 29 carbapenems-resistant Klebsiella, there were 9 isolates resistant to glycylcycline (tigecycline), and one isolate was resistant to polymyxin (colistin), and 3 were resistant to both colistin and tigecycline.
Two isolates of Stenotrophomonas maltophilia were recorded, and both were PDR (P value: < 0.001). Among 14 isolates of E. coli, 8 were sensitive, while 4 isolates were MDR and 2 XDR. Among 4 carbapenems-resistant E. coli, 2 isolates were resistant to tigecycline, while no resistance was recorded to colistin. Pseudomonas aeruginosa sensitive isolates were 6, while 5 isolates were MDR, one was XDR, and another isolate was PDR. Among 6 carbapenems-resistant Pseudomonas aeruginosa, one isolate was resistant to tigecycline, and one isolate was resistant to both tigecycline and colistin. Regarding Acinetobacter baumannii, one isolate was sensitive to standard antibiotics, and 4 isolates were MDR which all were susceptible to tigecycline and colistin.
Among gram-positive organisms, Staph. aureus was the predominant organism, 16 isolates were methicillin-sensitive Staphylococcus aureus (MSSA), and 9 methicillin-resistant Staphylococcus aureus (MRSA) isolates were recorded; 2 isolates were resistant to linezolid and vancomycin; 2 isolates were resistant to teicoplanin; 2 isolates were resistant to linezolid, vancomycin, and teicoplanin; and one isolate was PDR including tigecycline and sulfamethoxazole-trimethoprim. Moreover, Candida albicans was recorded in this study; only in HAP group, 7 cases (14%) and all isolates were susceptible to fluconazole (P-value: 0.037).
Among various risk factors for infection with antibiotic-resistant organisms in HAP group, hospitalization of > 2 days in the last month and intravenous antibiotic use within the last 3 months showed statistical significance (P-value 0.025 and 0.001, respectively), as shown in Table 5. Logistic regression analysis was done for detection of the strongest predictors for infection by MDR organisms HAP; in univariate regression analysis, hospitalization of > 2 days in last month (OR: 6.800, 95.0% confidence interval (CI): 1.082–42.730, P-value 0.041) and intravenous antibiotic use within last 3 months (OR: 8.750, 95.0% confidence interval (CI): 2.156–35.507, P-value 0.002) were the strongest predictors associated with risk for MDR-HAP. In multivariate analysis, intravenous antibiotic use within last 3 months (OR 7000, 95.0% confidence interval (CI): 1.498–32.720, P-value 0.013) was the strongest predictor associated with risk for MDR-HAP (Table 6).
Among various risk factors for infection with antibiotic-resistant organisms in VAP group, intravenous antibiotic use within last 3 months showed statistical significance with P-value, 0.001 (Table 7). Logistic regression analysis was done for detection of the strongest predictors of infection by MDR organisms-VAP; in univariate regression analysis, intravenous antibiotic use within last 3 months (OR 10.333, 95.0% confidence interval (CI): 2.220–48.092, P-value 0.003) was the strongest predictor associated with risk for MDR-VAP (Table 8).
Regarding duration of mechanical ventilation for VAP group, it ranged between 7 and 48 days with mean ± SD 15.160 ± 9.153, while total duration of hospital admission ranged between 10 and 34 days with mean ± SD 21.340 ± 6.026 in HAP group versus range of 7 to 50 days with mean ± SD 19.960 ± 10.654 in VAP group with no significant difference between both groups (Table 9) . On the other hand, outcomes of the patients showed significant difference between both groups (P-value: < 0.001) where mortality was the predominant outcome in VAP group, 33 patients (66%) versus 7 patients (14%) in HAP group in which most of the patients were cured, and discharged 43 patients (86%) versus 17 patients (34%) in VAP group (Table 9).
Discussion
In comparison to our data, Farhadi et al. (2021) reported in their study which aimed to screen frequency of MDR. In Klebsiella pneumoniae, mean age of studied patients was 51.7 years (ranged from 15 to 91 years), where 58% of the patients were females and 42% were males [15]. Also, Maurici et al. (2022) informed that among 329 patients with hospital-acquired respiratory infection, age ranged from 18 to 93 years, 96 (29.2%) were females, while 233 (70.8%) were male individuals; there were no significant differences in the average age compared to gender despite males were more than females. One-hundred forty (42.6%) patients were suffering from chronic diseases, lung disease (n = 46, 14.0%), heart failure (n = 45, 13.7%), kidney failure (51, 15.5%), and onco-hematological (24, 7.3%) diseases [16]. Kumar et al. (2018) showed that among 318 included patients for criteria of HCAP and HAP, 90 cases were diagnosed as HAP and 63 were diagnosed as VAP, whereas a total category (153 patients) age ranged from 16 to 85 years, mean ± SD 45.15 ± 20.6 and median 45 years, and 92 were males, regarding associated diseases, hypertension (71; 46.4), diabetes (11; 7.2%), coronary artery disease (13; 8.5%), chronic kidney diseases (5; 3.3%), neurological (34; 22.2%), and malignancy (13; 8.5%) [17]. In this study, diagnosis of suspected HAP and VAP was depending on clinical findings, regarding fever, deterioration of respiratory manifestation in the context of impaired oxygenation, purulent respiratory secretions, elevated inflammatory markers, and TLC, CRP, and D-dimer beside progressive or new radiological findings. In agreement with this study, Fagon et al. (1993) reported that standard diagnostic clinical criteria for HAP and VAP, beside abnormal chest radiographic findings, are composed of at least two of the three findings: fever, leukocytosis, and purulent tracheal secretions. When those occur, the possibility of VAP is high [18]. Also, the presence of radiographic infiltrates in a patient with fever, leukocytosis, or purulent tracheobronchial secretions has a high diagnostic sensitivity but low specificity. When all four criteria are present, specificity improves, but sensitivity drops to below 50% [19].
C-reactive protein is rapidly upregulated in the liver in response to cytokines originating at pathology site (particularly IL-6, which induces CRP mRNA) [20] and its synthesis rate depending on response to the inflammation intensity. Therefore, CRP is a superior biomarker for complex acute-phase characteristics, e.g., leukocytosis and fever. CRP secretion begins in 4–6 h and peaks at 36–50 h, potentially limiting its efficacy in predicting early treatment failure [21]. A single elevated plasma CRP concentration is not highly informative; so, CRP is not specific enough for diagnosis of nosocomial pneumonia [22], but continuous monitoring of CRP levels appears to be useful in the early prediction of VAP and the response to antibiotics [23]. Another challenge seems to be the differentiation of inflammation and infection using CRP [24]. This study agrees with the previous reports at the point of continuous monitoring, and not only CRP was already repeated for all patients suspecting nosocomial pneumonia but also total leucocytic count was repeated. We use D-dimer as another biomarker for inflammation and diagnosis of nosocomial pneumonia. D-dimer has a significant role not only in evaluation of venous thromboembolism but also in inflammatory diseases. With emergence of Covid-19 and associated hypercoagulable state, significance of D-dimer has increased not only as a predictor for venous thromboembolism but also as an inflammatory marker correlated with other inflammatory markers such as CRP and ferritin for predicted morbidity and mortality. Rueda-Camino et al. (2022) also documented through their study that estimating a corrected value of plasma D-dimer as a linear function of ferritin, CRP, and fibrinogen, establishing a cutoff point of high probability of VTE that high CRP and elevated levels of D-dimer levels among COVID-19 patients predict higher odds of mortality, and by using this predictive model based on levels, CRP and D-dimer can help the clinicians to make timely clinical decisions, improve patients’ treatment, and optimize use of limited clinical resources, but also, they recommended large-scale and longer-term studies to validate their results [25].
Borowiec et al. (2020) concluded that elevated levels of D-dimer in patients with granulomatosis with polyangiitis are associated with disease activity and inflammation rather than with the risk of venous thromboembolism, and value of d-dimer as a biomarker of venous thromboembolism in patients with small vessel vasculitis is low [26]. Also, Bao et al. (2017) documented in their study that correlated D-dimer level with the inflammatory conditions in patients with gastrointestinal diseases, and overall analysis showed positive correlation of D-dimer with white blood cell, percentage of neutrophils, neutrophil count, CRP, high sensitive CRP, procalcitonin, and blood culture results but negative correlation with lymphocyte percentage and count [27]. Higher values of CRP and D-dimer in VAP versus HAP may be explained with amplified inflammatory response and cytokine release in VAP patients [28].
Regarding admission cause, as this study was conducted at chest hospital, all patients were complaining of respiratory illness whatever it is acute or chronic, so most predominant causes were exacerbation of chronic respiratory diseases. Also, in Kumar et al. (2018) who conducted their study in a tertiary care hospital, among 153 recruited patients diagnosed with HAP and VAP, chronic lung disease was diagnosed in 15 patients, COPD was diagnosed in 14, and one cases for both asthma and bronchiectasis [17]. Maurici et al. (2022) conducted a study on 329 patients admitted at Italian teaching hospital diagnosed with hospital-acquired respiratory infections, and lung diseases were recorded in 46 (14%) patients [16]. Also, Chung et al. (2011) reported that among 2554 cases (1577 HAP and 977 VAP), chronic lung disease was determined in 472 (18.5%) patients, 288 in HAP group, and 184 patients in VAP group; also, there were 67 lung cancer cases in HAP versus 14 cases in VAP group [29].
Regarding radiological findings, CXR was done for all patients, and in some cases of HAP group, CT chest was requested when CXR was normal in the presence of clinical findings suggesting nosocomial pneumonia. Wunderink et al. (1992) and Winer-Muram et al. (1993) informed that diagnosis of HAP or VAP based on the presence of alveolar infiltrates on chest radiography has a sensitivity of 58 to 83% when using air bronchogram signs and is 50 to 78% for new or worsening infiltrates when compared with invasive techniques or histological studies [30, 31]. Also, Pugin et al. (1991) combined body temperature, white blood cell count, volume and appearance of tracheobronchial secretions, oxygenation, chest radiographic findings, and tracheal aspirate cultures into a Clinical Pulmonary Infection Score (CPIS) for VAP with a total score greater than six out of a maximum of 12 correlated with high bacterial counts isolated from the lower respiratory tract and a sensitivity and specificity of 93% and 100%, respectively [32]. More recently, Fartoukh et al. (2003) found that clinical prediction alone was inaccurate, but a modified CPIS score, incorporating a gram stain of respiratory tract secretions, improved diagnostic accuracy [33]. In this study, 3 sampling types (sputum, BAL, and ETA) yielded positive culture results. Also, Corrêa Rde, Luna, Anjos, Barbosa, Rezende, Rezende, Pereira, and Rocha (2014) concluded that management of VAP patients, based on the results of quantitative endotracheal aspirate cultures and BAL fluid cultures, had similar clinical outcomes [34].
Regarding microbiological findings in our study, gram-negative organisms were more predominant than gram-positive ones. K. pneumoniae, Staph. aureus, E. coli, and Pseudomonas were the predominant organisms with uniqueness of A. baumannii in VAP versus Candida albicans in HAP. Feng et al. (2019), agreed with our results in that, a predominance of gram -negative than gram- positive bacteria with significant predominance of A. baumannii in VAP, also, in our results K. pneumoniae showed significant predominance in VAP group and Candida albicans showed significant predominance in HAP group [35]. Also, Maurici et al. (2022) reported that respiratory samples including bronchoalveolar lavage fluid, bronchial aspirate, sputum specimen, and tracheal swab were examined, and microbiological analysis revealed that 332 (57.1%) organisms were gram-negative bacteria, 140 (24.1%) gram-positive bacteria, 2 (0.3%) mycobacterium tuberculosis, and 107 (18.4%) were fungal isolates; regarding gram-positive isolates, S. aureus (69.8%) and Enterococcus spp. (19.4%) were the prevalent bacteria, followed by Streptococcus pneumoniae (5.8%), Streptococcus pyogenes (4.3%), and Rothia (0.7%). In the gram-negative group, Klebsiella spp. (23.1%), Pseudomonas spp. (21.6%), and A. baumannii (18.6%) were the most prevalent microorganisms, followed by Enterobacter spp. (9.3%), E coli (9.0%), Stenotrophomonas maltophilia (4.5%), Proteus spp. (3.9%), and others. Candida spp. represented the majority of fungal isolates (87.0%), followed by Aspergillus spp. (12.1%) [16].
On the other hand, Chung et al. (2011) reported in their study that P. aeruginosa (15.6%) and S. aureus (15.5%) were the most frequent isolates in HAP followed by Acinetobacter spp. (13.6%) and K. pneumoniae (12%), while in VAP, Acinetobacter spp. was the most frequently isolated (36.5%) followed by P. aeruginosa (25.9%), K. pneumoniae (16.8%), and S. aureus (12.2%) [29], and Kumar et al. (2018) reported that Acinetobacter baumannii (62; 41%), Pseudomonas aeruginosa (21; 14%), and E. coli (17; 11%) were the most common organisms among HAP and VAP patients [17]. In this study, Providencia stuartii was recorded only in one female HAP case. In concordance with our findings, Abdallah et al. (2018) reported their first case of HAP caused by carbapenem-resistant Providencia stuartii in a 31-year-old man, and the patient responded well to the regimen, targeting carbapenem-resistant P. stuartii which was extended infusion of double-dose meropenem [36].
Although Providencia species have been isolated in humans from urine (most common), stool, blood, and from sputum, skin, and wound cultures, P. stuartii septicemia is primarily of urinary origin. One case study has informed P. stuartii as the etiology of infective endocarditis [37]. Also, an Italian study found that the prevalence of extended spectrum β-lactamases (ESBL)-producing P. stuartii in the general patient population increased from 31% in 1999 to 62% in 2002. Over a 4-year span, P. stuartii was isolated in 0.08% of patients. A total of 87% of isolates were found in urine, 10% in blood, and 3% in respiratory tract secretions [38].
Rahav et al. (1994) reported frequent isolation of P. stuartii from patients with indwelling urinary catheters and more persistence of organism in females. They explained that persistence may be due to different receptor characteristics in male and female urinary tracts and a bacterial predilection for Foley catheters over condom catheters, which are used more commonly in males [39].
Regarding organisms’ distribution according to antimicrobial susceptibility test, in agreement with our results, Ferreira et al. (2019) reported in their study high prevalence of MDR K. pneumoniae (21/25; 84%) with high resistance rate to the common antibiotics used either alone or in association with one another, such as β-lactams (including carbapenems), aminoglycosides, quinolones, glycylcycline, and polymyxin E [40]. Also, Chung et al. (2011) found in their study in Asian countries on HAP and VAP cases common bacterial isolates which were Acinetobacter, P. aeruginosa, S. aureus, and K. pneumoniae; all those isolates were highly resistant to major antimicrobial agents. Eighty-two percent of S. aureus isolates were MRSA, and most of those were MDR. ESBL production rate was high in K. pneumoniae. A significant proportion of P. aeruginosa were MDR and resistant to imipenem as well. Pseudomonas isolates from China were especially highly resistant to imipenem, and three strains showed PDR. Acinetobacter spp. showed high resistance rates to imipenem in Malaysia, Thailand, India, and China, and XDR rate was remarkably high [29]. Also, Maurici et al. (2022) recorded 82 out of the 581 isolated microorganisms were antibiotic resistant. A. baumannii, K. pneumoniae, and S. aureus represented the majority (62.2%) of antibiotic-resistant organisms: 22 MDR and 2 PDR A. baumannii, 9 PDR and 5 MDR K. pneumoniae, and 13 MDR S. aureus. No Pseudomonas spp. isolates exhibited antibiotic resistance. Six Aspergillus spp. isolates were found to be azole resistant, whereas all Candida spp. isolates were non-resistant [16]. Kumar et al. (2018) also reported among 153 cases of HAP and VAP, microbial isolates were 33 MDR, 110 XDR, and 3 PDR with 53 isolates were sensitive only to colistin. In Acinetobacter species, 7 MDR, 60 XDR, zero PDR isolates, and 33 isolates were sensitive only to colistin. In E. coli, 7 MDR, 7 XDR, zero PDR, and 3 isolates were sensitive only to colistin. In Pseudomonas, 1 MDR, 20 XDR, 1 PDR, and 13 were sensitive only to colistin. In K. pneumoniae, 2 MDR, 13 XDR, zero PDR, and 7 isolates were sensitive only to colistin. There were 5 isolates of MRSA, 4 MDR, and 1 XDR [17].
In our study, uniqueness of Candida albicans in HAP (5 ICU cases and 2 ward cases) is explained with several factors, among 7 cases; 4 patients were diabetic (poorly controlled) while other 3 cases; one case had ischemic heart disease and malnourished with late HAP, remaining 2 cases had late HAP, chronic inhaled corticosteroid use with past history of intravenous antibiotic use in last 3 months, and all 7 cases received broad-spectrum antibiotic at early management which predisposed to fungal infection as well as abundance of organism with the absence of another pathogenic organisms at microbiological examination and response to antifungal therapy which encouraged diagnosis of Candida pneumonia. On the other hand, Schnabel et al. (2014) reported that among included 701 BAL specimens, only 5 patients (0.7%) diagnosed with Candida pneumonia (malnutrition, malignancy, steroid therapy, and aspiration were risk factors for Candida pneumonia and informed that most Candida pneumonia reports are based on isolation of Candida from sputum aspirates or BAL in the absence of other causative pathogens with conclusion of Candida pneumonia as a rare clinical issue [41].
Regarding risk factors for infection by MDRO, in agreement with this study, Feng et al. (2019) reported antibiotic therapy in the preceding 90 days was an important risk factor for MDRO-HAP in their study [35]. Also, this finding is similar to that of the previous studies of Nseir et al. (2008) and Raman et al. (2015) that suggested the impact of virulence and antibiotic tolerance [42, 43]. Regarding VAP, Thom et al. (2017) suggested that patients who required mechanical ventilation or have undergone tracheotomy can easily be colonized by MDRO [44]. ICU admission or broad-spectrum antibiotic use also contributes to MDRO infection [45].
On the other hand, Kalil et al. (2016) and Ekren et al. (2018) reported risk factor for MDRO with HAP or VAP was ≥ 5 days of hospitalization [3, 46]. Kumar et al. (2018) documented in their study that among risk factors for antibiotic-resistant organisms, length of hospital stay ≥ 5 days was seen in 68 cases of HCAP and 81 cases of HAP (P-value = 0.036), and by using multivariate regression analysis, chronic lung diseases were the strongest predictor of MDR bacteria (OR 2.73, 95% CI 1.26–5.915, P-value = 0.011). Detection of A. baumannii (OR 35.503, 95% CI 14.688–85.817, P = 0.000), Pseudomonas spp. (OR 8.913, 95% CI 2.762–28.76, P-value = 0.000), and bacteria belonging to family of Enterobacteriaceae (OR 9.533, 95% CI 2.62–34.688, P-value = 0.001) as well as immunocompromised condition (OR 4.050, 95% CI 1.39–11.796, P-value = 0.010) were the strongest predictors for XDR organisms [17].
Regarding outcomes of the patients, in agreement with these results, Feng et al. (2019) reported the 30-day mortality rates in their study of HAP and VAP were 18.5% and 42.5%, respectively, [35], in consistence with previous reports of Ewan et al. (2015) and Ding et al. (2017) [47, 48]. Also, Da Silveira et al. (2019) and Siniscalchi et al. (2016) referred the higher mortality rates in VAP group to higher incidence of severe infection or comorbid conditions in those category [49, 50]. On the other hand, Chung et al. (2011) reported mortality rates of HAP and VAP in their study were 34.4% and 45.7%, respectively [29], and also, Kumar et al. (2018) reported death rate in HAP patients was 35.6% (32/90) while in VAP patients was 28/63, 44% [17].
There were some limitations in this study; this study was a single-center one conducted at University Chest Hospital and included only medical patients with certain indications for admission for respiratory diseases and mechanical ventilation. Small sample size leads to sparse data bias. No scoring system was used to evaluate severity of pneumonia and associated complications, lack of testing for viruses and atypical organisms and lack of invasive sampling procedure, and bronchoalveolar lavage (BAL) and endotracheal aspirate (ETA) for HAP group.
Conclusion
In this study, the etiological organisms for HAP and VAP were relatively the same with predominance of antimicrobial-resistant organisms in VAP group, especially Klebsiella pneumoniae, Acinetobacter baumannii, and Stenotrophomonas maltophilia. Intravenous antibiotic use within the last 3 months was the strongest predictor for risk of MDRO infection in both HAP and VAP group. VAP was associated with higher death rates compared with HAP. Our results should be considered as an alarm for raising attention towards prevention and control of the MDR organisms specially K. pneumoniae in hospitals which showed resistance to drugs commonly used for these organisms as well as antimicrobial agents such as polymyxin E and glycylcycline which are lastly resorted for life-threatening infections in ICU patients.
Availability of data and materials
All are available from the corresponding author on request.
Abbreviations
- A. baumannii :
-
Acinetobacter baumannii
- BAL:
-
Bronchoalveolar lavage
- Cm:
-
Centimeter
- CXR:
-
Chest X-ray
- COPD:
-
Chronic obstructive pulmonary disease
- CPIS:
-
Clinical Pulmonary Infection Score
- CFU:
-
Colony-forming units
- CI:
-
Confidence interval
- Covid-19:
-
Coronavirus disease 2019
- CRP:
-
C-reactive protein
- CT:
-
Computerized tomography
- ETA:
-
Endotracheal aspirate
- ETT:
-
Endotracheal tube
- E. coli :
-
Escherichia coli
- ESBL:
-
Extended spectrum β-lactamases
- FIO2:
-
Fraction of inspired oxygen
- HCAP:
-
Healthcare-associated pneumonia
- HAP:
-
Hospital-acquired pneumonia
- HIV:
-
Human immunodeficiency virus
- ICU:
-
Intensive care unit
- INR:
-
International normalized ratio
- K. pneumoniae :
-
Klebsiella pneumoniae
- L:
-
Liter
- MRSA:
-
Methicillin-resistant Staphylococcus aureus
- mL:
-
Milliliter
- mm:
-
Millimeter
- MDR:
-
Multidrug resistant
- MDRO:
-
Multidrug-resistance organisms
- mRNA:
-
Messenger ribonucleic acid
- OR:
-
Odds ratio
- PDR:
-
Pan-drug resistant
- P. stuartii:
-
Providencia stuartii
- P. aeruginosa:
-
Pseudomonas aeruginosa
- SN:
-
Serial number
- Spp.:
-
Species
- S. aureus:
-
Staph. aureus
- SPSS:
-
Statistical Package for the Social Sciences
- µg:
-
Microgram
- VTE:
-
Venous thromboembolism
- VAP:
-
Ventilator-associated pneumonia
- XDR:
-
Extensively drug-resistant XDR
References
Magill SS, Edwards JR, Fridkin SK (2014) Emerging infections program healthcareassociated infections and antimicrobial use prevalence survey team. Survey of health care-associated infections. N Engl J Med 370(26):2542–3
Martin-Loeches I, Povoa P, Nseir S (2020) A way towards ventilator-associated lower respiratory tract infection research. Intensive Care Med 46:1504–1505
Kalil AC, Metersky ML, Klompas M, Muscedere J, Sweeney DA, Palmer LB, Napolitano LM, O’Grady NP, Bartlett JG, Carratala J et al (2016) Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the infectious diseases society of America and the American thoracic society. Clin Infect Dis 63(5):e61–e111
Hunter JD (2006) Ventilator associated pneumonia. Postgrad Med J 82(965):172–178
Pagana KD, Pagana TJ, Pagana TN (2022) Chapter 7: Microscopic studies and associated testing. In Mosby’s manual of diagnostic and laboratory tests, 7th edn. St. Louis: Elsevier, pp 661–746
American Association for Respiratory Care - AARC (2010) AARC Clinical Practice Guidelines (2010) Endotracheal suctioning of mechanically ventilated patients with artificial airways. Respir Care 55(6):758–764
Linssen CFM, Bekers O, Drent M, Jacobs JA (2008) C-reactive protein and procalcitonin concentrations in bronchoalveolar lavage fluid as a predictor of ventilator-associated pneumonia. Ann Clin Biochem 45(3):293–298. https://doi.org/10.1258/acb.2007.007133
Musher DM, Montoya R, Wanahita A (2004) Diagnostic value of microscopic examination of gram-stained sputum and sputum cultures in patients with bacteremic pneumococcal pneumonia. Clin Infect Dis 39(2):165–169
Magill SS, Klompas M, Balk R, Burns SM, Deutschman CS, Diekema D, Fridkin S, Greene L, Guh A, Gutterman D, Hammer B, Henderson D, Hess D, Hill NS, Horan T, Kollef M, Levy M, Septimus E, Vanantwerpen C, Wright D, Lipsett P (2013) Developing a new national approach to surveillance for ventilator associated events: executive summary. Clin Infect Dis 57(12):1742–1746
Joseph NM, Sistla S, Dutta TK, Badhe AS, Parija SC (2010) Ventilator-associated pneumonia: role of colonizers and value of routine endotracheal aspirate cultures. Int J Infect Dis 14:723–729
Nair S, Sen N, Peter JV et al (2008) Role of quantitative endotracheal aspirate and cultures as a surveillance and diagnostic tool for ventilator-associated pneumonia: a pilot study. Indian J Med Sci 62(8):304–313
Mahon CR, Lehman DC (2022) Textbook of diagnostic microbiology-E-Book. Elsevier Health Science
CLSI (2023) Performance Standars for Antimicrobial Susceptibility Testing. 33rd ed. CLSI supplement M100. Clinical and Laboratory Standars Institute
Basak S, Singh P, Rajurkar M (2016) Multidrug resistant and extensively drug resistant bacteria: a study. J Pathog 2016: Article ID 4065603, https://doi.org/10.1155/2016/4065603
Farhadi M, Ahanjan M, Goli HR, Haghshenas MR, Gholami M (2021) High frequency of multidrug-resistant (MDR) Klebsiella pneumoniae harboring several β-lactamase and integron genes collected from several hospitals in the north of Iran. Ann Clin Microbiol Antimicrob 20:70. https://doi.org/10.1186/s12941-021-00476-1
Maurici M, D’Alò GL, Fontana C, Santoro V, Gaziano R, Ciotti M, Cicciarella Modica D, De Filippis P, Sarmati L, De Carolis G, Pica F (2022) Microbiology and clinical outcome of hospital-acquired respiratory infections in an Italian teaching hospital: a retrospective study. Healthcare 10:2271. https://doi.org/10.3390/healthcare10112271
Kumar S, Jan RA, Fomda BA, Rasool R, Koul P, Shah S, Khan UH, Qadri SM, Masoodi SR, Mantoo S, Muzamil M (2018) Healthcare-associated pneumonia and hospital-acquired pneumonia: bacterial aetiology, antibiotic resistance and treatment outcomes: a study from North India. Lung 196:469–479
Fagon JY, Chastre J, Hance AJ, Domart Y, Trouillet JL, Gibert C (1993) Evaluation of clinical judgment in the identification and treatment of nosocomial pneumonia in ventilated patients. Chest 103:547–553
Sutherland KR, Steinberg KP, Maunder RJ, Milberg JA, Allen DL, Hudson LD (1995) Pulmonary infection during the acute respiratory distress syndrome. Am J Respir Crit Care Med 152:550–556
Salluh JIF, Souza-Dantas VC, Povoa P (2017) The current status of biomarkers for the diagnosis of nosocomial pneumonias. Curr Opin Crit Care 23:391–397
McLean AS, Tang B, Huang SJ (2015) Investigating sepsis with biomarkers. BMJ 350:h254
Lin Q, Fu F, Shen L, Zhu B (2013) Pentraxin 3 in the assessment of ventilator-associated pneumonia: an early marker of severity. Heart Lung 42:139–145
Povoa P, Coelho L, Almeida E, Fernandes A, Mealha R, Moreira P, Sabino H (2005) C-reactive protein as a marker of ventilator-associated pneumonia resolution: a pilot study. Eur Respir J 25:804–812. https://doi.org/10.1183/09031936.05.00071704
Samraj RS, Zingarelli B, Wong HR (2013) Role of biomarkers in sepsis care. Shock 40:358–365. https://doi.org/10.1097/SHK.0b013e3182a66bd6
Rueda-Camino JA, Sendín-Martín V, Joya-Seijo MD, Angelina-García M, Zamarro-García C, Gimena-Rodríguez FJ, Barba-Martín R (2022) Plasma D-dimer value corrected by inflammatory markers in patients with SARS-CoV-2 infection: its prognostic value in the diagnosis of venous thromboembolism. Med Clin (Barc) 158(6):265–269. https://doi.org/10.1016/j.medcli.2021.03.034
Borowiec A, Dąbrowski R, Kowalik I, Rusinowicz T, Hadzik-Błaszczyk M, Krupa R, Życińska K (2020) Elevated levels of d-dimer are associated with inflammation and disease activity rather than risk of venous thromboembolism in patients with granulomatosis with polyangiitis in long term observation. Adv Med Sci 65(1):97–101. https://doi.org/10.1016/j.advms.2019.12.007
Bao W, Qi X, Li H, Hou F, Zhang X, Wang R, Guo X (2017) Correlation of D-dimer level with the inflammatory conditions: a retrospective study. AME Med J 2(3):27. https://doi.org/10.21037/amj.2017.02.07
Povoa P (2002) C-reactive protein: a valuable marker of sepsis. Intensive Care Med 28:235–243. https://doi.org/10.1007/s00134-002-1209-6
Chung DR, Song JH, Kim SH, Thamlikitkul V, Huang SG, Wang H, So TM, Yasin RM, Hsueh PR, Carlos CC, Hsu LY, Buntaran L, Lalitha MK, Kim MJ, Choi JY, Kim SI, Ko KS, Kang CI, Peck KR, Asian Network for Surveillance of Resistant Pathogens Study Group (2011) High prevalence of multidrug-resistant nonfermenters in hospital-acquired pneumonia in Asia. Am J Respir Crit Care Med 184(12):1409–17
Wunderink RG, Woldenberg LS, Zeiss J, Day CM, Ciemins J, Lacher DA (1992) The radiologic diagnosis of autopsy-proven ventilator-associated pneumonia. Chest 101:458–463
Winer-Muram HT, Rubin SA, Ellis JV et al (1993) Pneumonia and ARDS in patients receiving mechanical ventilation: diagnostic accuracy of chest radiography. Radiology 188:479–485
Pugin J, Auckenthaler R, Mili N, Janssens JP, Lew PD, Suter PM (1991) Diagnosis of ventilator-associated pneumonia by bacteriologic analysis of bronchoscopic and nonbronchoscopic “blind” bronchoalveolar lavage fluid. Am Rev Respir Dis 143:1121–1129
Fartoukh M, Maitre B, Honoré S, Cerf C, Zahar JR, Brun-Buisson C (2003) Diagnosing pneumonia during mechanical ventilation: the clinical pulmonary infection score revisited. Am J Respir Crit Care Med 168:173–179
Corrêa Rde A, Luna CM, Anjos JC, Barbosa EA, Rezende CJ, Rezende AP, Pereira FH, Rocha MO (2014) Quantitative culture of endotracheal aspirate and BAL fluid samples in the management of patients with ventilator-associated pneumonia: a randomized clinical trial. J Bras Pneumol 40(6):643–651
Feng DY, Zhou YQ, Zou XL, Zhou M, Zhu JX, Wang YH, Zhang TT (2019) Differences in microbial etiology between hospital-acquired pneumonia and ventilator-associated pneumonia: a single-center retrospective study in Guang Zhou. Infect Drug Resist 12:993–1000
Abdallah M, Alhababi R, Alqudah N, Aldyyat B, Alharthy A (2018) First report of carbapenem-resistant Providencia stuartii in Saudi Arabia. New Microbes New Infect 26:107–109. https://doi.org/10.1016/j.nmni.2018.09.007
Krake PR, Tandon N (2004) Infective endocarditis due to Providenca stuartii. South Med J 97(10):1022–1023
Tumbarello M, Citton R, Spanu T et al (2004) ESBL-producing multidrug-resistant Providencia stuartii infections in a university hospital. J Antimicrob Chemother 53(2):277–282
Rahav G, Pinco E, Silbaq F, Bercovier H (1994) Molecular epidemiology of catheter-associated bacteriuria in nursing home patients. J Clin Microbiol 32(4):1031–1034
Ferreira RL, da Silva BCM, Rezende GS, Nakamura-Silva R, Pitondo-Silva A, Campanini EB, Brito MCA, da Silva EML, Freire CCM, Cunha AF, Pranchevicius MC (2019) High prevalence of multidrug-resistant Klebsiella pneumoniae harboring several virulence and β-lactamase encoding genes in a Brazilian intensive care unit. Front Microbiol 9:3198. https://doi.org/10.3389/fmicb.2018.03198
Schnabel RM, Linssen CF, Guion N, van Mook WN, Bergmans DC (2014) Candida pneumonia in intensive care unit? Open Forum Infect Dis 1(1):ofu026. https://doi.org/10.1093/ofid/ofu026
Nseir S, Ader F (2008) Prevalence and outcome of severe chronic obstructive pulmonary disease exacerbations caused by multidrug-resistant bacteria. Curr Opin Pulm Med 14:95–100. https://doi.org/10.1097/MCP.0b013e3282f37a11
Raman G, Avendano E, Berger S, Menon V (2015) Appropriate initial antibiotic therapy in hospitalized patients with gram-negative infections: systematic review and meta-analysis. BMC Infect Dis 15:395. https://doi.org/10.1186/s12879-015-1123-5
Thom KA, Rock C, Jackson SS et al (2017) Factors leading to transmission risk of Acinetobacter baumannii. Crit Care Med 45:e633–e639. https://doi.org/10.1097/CCM.0000000000002318
Ang H, Sun X (2018) Risk factors for multidrug-resistant gram-negative bacteria infection in intensive care units: a meta-analysis. Int J Nurs Pract 24(4):12644. https://doi.org/10.1111/ijn.12644
Ekren PK, Ranzani OT, Ceccato A et al (2018) Evaluation of the 2016 Infectious Diseases Society of America/American Thoracic Society guideline criteria for risk of multidrug-resistant pathogens in patients with hospital-acquired and ventilator-associated pneumonia in the ICU. Am J Respir Crit Care Med 197(6):826–830. https://doi.org/10.1164/rccm.201708-1717LE
Ewan VC, Sails AD, Walls AW, Rushton S, Newton JL (2015) Dental and microbiological risk factors for hospital-acquired pneumonia in non-ventilated older patients. PLoS One 10(4):0123622
Ding C, Zhang Y, Yang Z et al (2017) Incidence, temporal trend and factors associated with ventilator-associated pneumonia in mainland China: a systematic review and meta-analysis. BMC Infect Dis 17(1):468. https://doi.org/10.1186/s12879-017-2566-7
Da Silveira F, Nedel WL, Cassol R, Pereira PR, Deutschendorf C, Lisboa T (2019) Acinetobacter etiology respiratory tract infections associated with mechanical ventilation: what impacts on the prognosis? A retrospective cohort study. J Crit Care 49:124–128. https://doi.org/10.1016/j.jcrc.2018.10.034
Siniscalchi A, Aurini L, Benini B et al (2016) Ventilator associated pneumonia following liver transplantation: etiology, risk factors and outcome. World J Transplant 6(2):389–395
Acknowledgements
The authors would like to thank all patients and their families for accepting participation in this study. Many thanks to the nursing team and residents of respiratory ICU, Tanta University Hospital, and to Mr. Ibrahim Rabie, for helping in statistical analysis.
Funding
Nil.
Author information
Authors and Affiliations
Contributions
AAE, collection, analysis, and interpretation of data, performing some cases of BAL, and writing, reviewing, and editing manuscript. MT, conception, design, and sharing in collection of data, performing some cases of BAL, and writing, reviewing, and editing manuscript. MMM, did cultures and microbiological analysis, writing methodology, and reviewing and editing the manuscript. AST and MAE, adjust therapeutic doses of antimicrobial agents and follow up drugs interactions. MSH and AHAEZ supervised manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Ethical Committee approval was taken from Faculty of Medicine, Tanta University, approval code (22/5/35500), and written consents were obtained from all patients and their families.
Consent for publication
Not applicable. Neither identifying images nor personal data were included in the manuscript.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Elkholy, A.A., Hantera, M.S., Abd El-Zaher, A.H. et al. Microbiological analysis of nosocomial pneumonia at Tanta University Chest Hospital. Egypt J Bronchol 17, 42 (2023). https://doi.org/10.1186/s43168-023-00215-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43168-023-00215-4