Abstract
Background
Trigger finger is one of the most common causes of pain and dysfunction. The aim of our study was to compare the effectiveness of shock wave therapy versus local corticosteroid injection in the treatment of trigger finger in diabetic patients.
Results
The two groups were matched on age, HbA1c, VAS score, and grip force at week zero. There was statistically significant improvement in both groups after treatment. In comparing the two groups after 6 weeks of starting treatment either by shockwave therapy or by local corticosteroid injection, there was a statistically significant improvement in group 1 as regards VAS pain score (p = 0.012), clinical signs (finger extension and locking) (p = 0.018), hand grip dynamometer (p < 0.001), and musculoskeletal ultrasound findings in comparison to group 2.
Conclusion
Trigger finger is a common complication of DM. Shockwave therapy was able to reduce pain and improve hand functional level and quality of life. Shockwave is an effective, safe, non-invasive method for conservative management of trigger finger.
Similar content being viewed by others
Background
Trigger finger is one of the most common causes of hand pain and dysfunction, with a prevalence of 3% in the population [1]. It is also known as stenosing tenosynovitis, characterized by hypertrophy at the intersection of the tendon with its pulley. This constriction of the tendon prevents smooth gliding through the ligament pulley, resulting in sudden finger release or locking during flexion, extension, or hand movement [2].
Trigger finger is primarily diagnosed clinically and is associated with risk factors such as aging and female gender. It is often encountered in the elderly population, especially due to the high prevalence of diabetes mellitus (DM) in this age group [3]. Commonly affected digits include the thumb and ring finger of the dominant hand. The classic presentation involves finger popping and locking, while patients with acute trigger finger often experience pain, swelling, and limited range of motion [4]. It can be either idiopathic or secondary to conditions such as rheumatoid arthritis, type 2 diabetes, or mucopolysaccharidoses. Some studies suggest a correlation between trigger finger and hand overuse or repetitive hand trauma [5].
Diabetes mellitus (DM) is a substantial risk factor for trigger finger, elevating the risk by up to 10%. DM leads to the accumulation of glycosylated proteins and less soluble collagen in connective tissues. These pathological changes can damage blood vessels and nerves, giving rise to musculoskeletal manifestations that may contribute to significant physical disability and a decline in quality of life [6].
Conservative treatment is recommended for uncomplicated patients referred after the onset of symptoms [7].
Local corticosteroid injections are typically the first-line treatment for trigger finger due to their convenience and effectiveness, with response rates ranging from 42 to 92% across all grades of the condition. Accurate injection into the flexor tendon sheath is crucial to maximize therapeutic effects and minimize side effects such as skin atrophy, tendon rupture, and skin discoloration. Corticosteroid injections exert their anti-inflammatory effects by inhibiting the formation of inflammatory mediators like cyclooxygenase and phospholipase A2 [8].
Extracorporeal shock wave therapy (ESWT) has appeared as an alternative and promising method for treating musculoskeletal disorders. ESWT utilizes pressure waves, resembling a rocket mechanism, to stimulate healing. Physically, a shockwave is described as the propagation of acoustic energy that disperses in three-dimensional spaces and can be transmitted, reflected, or absorbed. Shock wave therapy regulates growth and proliferation factors and reduces pain through substances like calcitonin gene-related peptides and substance P [9]. Numerous studies have demonstrated its effectiveness in various pathologies, including trigger finger [10]. Additionally, it can serve as an alternative conservative treatment option for diabetic patients who may not be suitable candidates for corticosteroid injections due to complications.
Musculoskeletal ultrasound is a non-invasive imaging technique for visualizing soft tissues, making it the preferred method for diagnosing wrist and hand pathologies. It provides dynamic imaging, offering valuable information about the thickness of annular pulleys, the structure of flexor tendons, irregularities in tendon margins, and fluid accumulation in tendon sheaths [11]. Our study aims to compare the effectiveness of shock wave therapy versus local corticosteroid injections in the treatment of chronic trigger finger in diabetic patients.
Methods
Patients, study design
Fifty patients with type II diabetes mellitus, who were nonsmokers and presented with chronic trigger finger grades II and III based on Green’s classification [12], were recruited from outpatient clinics at university hospitals. The Green classification categorized trigger finger as follows: grade I—a history of catching and tenderness over the A1 pulley; grade II—demonstrable catching with the patient’s ability to actively extend the digit; grade IIIA—demonstrable catching requiring passive extension; grade IIIB—demonstrable catching requiring passive flexion; and grade IV—fixed flexion contracture of the proximal interphalangeal joint [12].
The patients were randomly divided into two groups. Group 1 consisted of 25 patients who received six shockwave therapy sessions, one session per week, while the other 25 patients in group 2 received a single local corticosteroid injection into the affected flexor tendon sheath.
Inclusion criteria for this study encompassed patients with type II diabetes aged 50 years or older, diagnosed with chronic trigger finger based on the clinical presentation, which includes the presence of pain, tenderness, discomfort, or a palpable nodule over the flexor tendon sheath for months or years. Patients with Green’s classification grades I and IV, type I diabetes mellitus, uncontrolled diabetes (HbA1c > 7), rheumatological diseases, current pregnancy, traumatic trigger finger, local infection, a history of malignancy, or prior surgery for trigger finger were excluded from the study. Informed consent was obtained from all study participants, and the research received approval from the Research Ethics Committee.
Clinical assessment
All patients underwent a comprehensive medical history assessment along with general and local examinations. Finger pain was evaluated using a Visual Analogue Scale (VAS), where respondents choose a whole number on a scale of 0 to 10, with “0” representing no pain and “10” signifying extreme pain. Range of motion (ROM) assessments for all fingers included both active and passive movements, measured with a goniometer.
Grip force in the affected hand was quantified as the maximum grip strength using a baseline hand dynamometer. This measurement was taken with the patient seated, the shoulder at 0° abduction, in a neutral position, and the elbow flexed at 90°. A 30-s pause was allowed between measurements, and the mean value of three measurements was recorded in kilograms [13].
The Green classification was used to assess each trigger finger, categorized as either grade II or III. HbA1c levels were measured in all patients before starting treatment using an enzymatic assay.
Musculoskeletal ultrasound
High-resolution ultrasound was conducted using a linear transducer (12 MHz) (LOGIQ 9 Pro Series; GE Medical Systems, USA). Patients were seated in front of the examiner, with their hands and arms extended, resting on the examination table. The musculoskeletal ultrasound machine was positioned beside the patient. The patient’s hand was placed in a supine position, and a suitable amount of transmission gel was applied. A sterile field was maintained around the transducer’s edge.
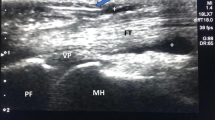
The scanning technique was carried out first in the transverse position and then in the longitudinal position. This allowed for the assessment of key parameters, including the thickness of the A1 pulley (with an average thickness of less than 0.5 mm) [14], the thickness of the synovial sheath surrounding the flexor tendons (with an average thickness of less than 1 mm) [15], and the use of power Doppler ultrasound to assess hypervascularity. Power Doppler ultrasound was initially used to confirm the diagnosis of trigger finger.
A reevaluation of both groups was conducted after 6 weeks from the initiation of treatment.
Extra-corporal shock wave therapy (ESWT)
The shockwave device (Technikwave GmbH & Co. KG, China) was utilized with a handheld probe to administer high-energy pulses, akin to sound waves, to the A1 pulley of the affected finger. This shock wave device delivered waves at an energy flux density of 0.2 mJ/mm2 and a frequency of 6 Hz.
In group 1, patients received a total of six sessions of extracorporeal shock wave therapy (ESWT) spanning 6 weeks, with one session conducted per week. Each session comprised 2000 impulses, and standard ultrasound gel was employed to facilitate coupling between the shock wave generator and the film drape.
Local corticosteroid injection
In group 2, patients received a single local corticosteroid injection guided by ultrasound. The patient was seated in a face-to-face position with the physician, and the physician palpated the flexor tendon nodule on the A1 pulley. The patient’s palm was sanitized with alcohol, and the affected finger was positioned in a flexed state. A 25-gauge needle was inserted at a 45° angle, deep into the flexor tendon, starting from the proximal to the distal part of the A1 pulley. Care was taken to ensure that the needle and puncture site did not come into contact with the ultrasound probe.
The finger was then moved through a range of bending and extending motions to confirm that the needle moved in tandem with the finger’s movements. The injection proceeded by repeating flexion and extension until the needle no longer moved. Subsequently, a mixture consisting of 0.5 ml of 40 mg/ml triamcinolone acetonide and 0.5 ml of lidocaine was injected into the flexor tendon sheath, beginning proximally and progressing distally at the nodule site. The success of intra-sheath injection was confirmed using musculoskeletal ultrasound, which also allowed monitoring of the injected solution’s flow beneath the A1 pulley.
After the injection, a small sterile adhesive dressing was applied, and the patient was allowed to move the finger immediately. Nonsteroidal anti-inflammatory drugs (NSAIDs) and anti-edematous drugs were prescribed post-injection.
Statistical methods
The collected data were reviewed, coded, tabulated, and entered into a computer using Statistical Package for the Social Sciences version 20 (SPSS-V20) in the USA. The data were then presented and analyzed according to the type of data obtained for each parameter.
For numerical data, the mean, standard deviation (SD), and range were calculated. Non-numerical data were presented as frequencies and percentages. To compare two groups of quantitative data, Student’s t-test was employed. The linear correlation coefficient was utilized to assess the strength of association between nonparametric variables within the same group.
An ANOVA test was applied to compare data at different time points within the same group for quantitative data. Results were considered statistically significant at p ≤ 0.05 and highly significant at p ≤ 0.001.
Results
Table 1 presents demographic, clinical, and laboratory data, along with scoring parameters for each group. The two groups were well-matched in terms of age, sex, and HbA1c.
When comparing the pre- and post-treatment assessments of group 1, there was a highly significant improvement in Green’s classification (p < 0.001), VAS pain score (p < 0.001), clinical signs (finger flexion, extension, and locking) (p < 0.001), hand grip strength (p < 0.001), presence of a palpable nodule (p = 0.027), and musculoskeletal ultrasound findings (thickness of A1 pulley and synovial sheath thickness) (Fig. 1, Tables 2 and 3).
When comparing the pre- and post-treatment assessments of Group 2, we found highly significant improvements in Green’s classification (p < 0.001), VAS pain score (p < 0.001), finger flexion and locking (p < 0.001), hand grip strength (p < 0.001), and musculoskeletal ultrasound findings (thickness of A1 pulley and synovial sheath thickness) (Tables 4 and 5).
When comparing the two groups after receiving treatment, group 1 exhibited statistically significant improvements compared to group 2 in terms of VAS pain score (p = 0.012), clinical signs (finger extension and finger locking) (p = 0.018), hand grip strength (p < 0.001), absence of palpable nodules (p = 0.021), and musculoskeletal ultrasound findings (size of A1 pulley) (p = 0.002) (Table 6).
Discussion
Diabetes mellitus is a chronic metabolic global disease that is rapidly increasing in prevalence, morbidity, and mortality [16]. DM affects the structure of the extracellular matrix, impairs cell viability, and causes pathological alterations [6].
Trigger finger is a clinical disorder that is a common complication of DM. It is characterized by pain and locking of the digits because of the disproportion between the diameter of the flexor tendons and the A1 pulley [17].
Musculoskeletal ultrasound has been proven to be an effective diagnostic tool for a variety of conditions and has been used for the evaluation of small, relevant structures of the wrist and hand. It is a highly dynamic imaging technique that allows an accurate evaluation of the tendons, joints, nerves, and vessels of the hand and wrist [18].
Treatment of trigger finger includes NSAIDS, physical therapy, corticosteroid injections, and recently, ESWT therapy [19]. The effectiveness of the best treatment is still lacking evidence. Steroid injections have an established role as the first-line treatment for trigger digit, but their efficacy varies with a slightly lower total efficacy with time and may need further injections, which increase the suitability of the occurrence of complications of injection such as hypopigmentation of the skin, digital necrosis, and fat atrophy [20]. ESWT is a safe conservative treatment as it seems to reduce pain and improve the functional level and quality of life [7]. Our study aimed to compare the effectiveness of shock wave therapy versus local corticosteroid injections in the treatment of chronic trigger finger in diabetic patients.
In our study, the mean age was 57.365.098 in group 1 and 57.246.092 in group 2, which agreed with Ko et al. [21], who reported that the mean age of diabetic patients in their study was 55 years. Also, our results agreed with Sarkar et al. [6], who showed that 61% of patients in their study were more than 55 years old. We revealed no statistically significant difference in the mean age between the two groups, which was in agreement with Ferrara et al. [2], who reported no difference in patients’ ages in their study groups.
In our study, the mean disease duration was 14.628.8 months in group 1 and 15.529.211 months in group 2, which was not in agreement with Hollins et al. [1], whose study was done in patients with a 2.5-month duration. In our study, a longer disease duration was chosen to verify the effect of the chosen therapy on chronic trigger finger and to have a larger number of patients participate in the study.
We reported that the affected hand with trigger finger was the dominant hand in the two groups, which was also reported by Rydberg et al. [22] who found that all affected hands with trigger finger were the dominant ones, while another study done by Ferrara et al. [2] showed that only 66% of the affected hands were their dominant hands. We found that the thumb was affected by 52% in group 1 and by 44% in group 2. This was in accordance with the study that was done by Ryzewiez et al. [23] who showed that the thumb finger was the most common affected digit with trigger finger.
As regards group 1 in our study who received 6 shockwave sessions, they showed improvement in VAS score (p 0.001), extension range (p 0.003), flexion range (p 0.001), locking of the finger (p 0.001), absence of palpable nodules (p 0.027), and grip strength by dynamometer (p 0.001). This was also reported by Dogru et al. [7], whose study showed a decrease in pain, an increase in grip strength, and an increase in the range of motion of the affected digit, showing the positive effects of shockwave therapy. Also, their study concluded that the treatment of trigger finger with shockwave might be a non-invasive option as shockwave therapy fastens the process of cell regeneration and increases perfusion, angiogenesis, and growth factor upregulation, which helps to regenerate musculoskeletal and vascular structures in the surrounding tissues. Furthermore, we revealed statistically significant improvements regarding musculoskeletal findings such as the thickness of the pulley (p 0.001) and synovial sheath thickness (p 0.001) after shockwave therapy.
Local hydrocortisone injection was the first method used in the treatment of trigger finger. In our study, group 2, who was treated by a single local steroid injection, reported statistically significant improvement as regards VAS pain score (p 0.001), locking, finger flexion, and hand grip strength (p 0.001), which was also reported by Massoud et al. [24] who revealed a statistically significant difference as regards flexion range (p 0.001), but there was no statistically significant difference as regards extension range.
We revealed that patients in group 2 showed a statistically significant difference in thickness of A1 pulley and synovial sheath thickness pre- and post-treatment with steroids injection, but there was no statistically significant difference in the presence of palpable nodules and hypervascularity detected by ultrasound, which was in agreement with Massoud et al. [24] who injected steroids with musculoskeletal ultrasound guidance, showed a decrease in size of A1 pulley and synovial sheath thickness, and suggested that accurate injection of steroids would increase the therapeutic results of steroids.
In comparing the results of the two groups, we found that there was a statistically significant improvement more in group 1 than in group 2 as regards VAS score (p = 0.012), as the role of shockwave in decreasing pain through stimulating soft tissue healing by inhibition of nociceptors decreases calcification, neovascularization, and hyperemia. Also, it has a direct effect on hyperstimulation, which blocks the gate control mechanism [7]. Ferrara et al. [2] suggested that shockwaves may facilitate the healing process by promoting the catabolic process, removing damaged matrix, and stimulating the synthesis of nitric oxide. Also, comparing the two groups, there was a statistically significant improvement regarding the size of the A1 pulley and synovial sheath thickness, which were detected by musculoskeletal ultrasound more in group 1, while Yildirim et al. [25] recommended that three sessions of shockwave could be as effective as steroid injection and suggested shockwave as an alternative treatment for patients who refuse injection or surgery. There was a statistically significant improvement in group 1 as regards the range of finger flexion and extension, while in group 2 there was a statistically significant improvement in the flexion range only and no statistically significant improvement in the extension range. This may be attributed to the improvement in pain, which was greater in group 1, as the prevalence of pain is one of the major causes of decreased joint movement. Shock wave therapy had a long-term effect on hand grip strength, which was proved by a study done by Dogru et al. [7]. Also, our study reported statistically significant improvements in hand grip strength in group 1 in comparison to group 2, which can be explained by the fact that pain decreases hand function and so improves hand grip strength.
Limitations of the study
One limitation of our study was the limited sample size, which was influenced by financial constraints. Additionally, there was a lack of long-term follow-up data due to poor patient compliance.
Conclusion
Trigger finger is a common complication of DM. The present study concluded that shock wave therapy was superior to local steroid injection in improving hand function and quality of life in patients with chronic trigger finger.
Recommendations
Shockwave therapy is an effective, safe, and non-invasive method for conservative management of trigger finger, especially in patients with uncontrolled DM or patients who refuse local steroid injections. Further studies are necessary to clarify the efficacy of different physical therapies on trigger finger.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- CTS:
-
Carpal tunnel syndrome
- DM:
-
Diabetes mellitus
- ESWT:
-
Extracorporeal shock wave therapy
- HBA1c:
-
Hemoglobulin A1c
- VAS:
-
Visual Analogue Scale
References
Hollins AW, Hein R, Atia A, Taskindoust M, Darner G, Shammas R et al (2022) Symptom duration and diabetic control influence success of steroid injection in trigger finger. Plast Reconstr Surg 150:357e–363e
Ferrara PE, Codazza S, Maccauro G, Zirio G, Ferriero G, Gianpaolo. (2020) Physical therapies for the conservative treatment of trigger finger: a narrative review. Orthop Rev (Pavia) 12(Suppl 1):86–80
Okita G, Hayashi M, Ikegami S, Iwakawa H, Haro H, Kato H (2022) The prevalence and risk factors for trigger digits in a random sampling of a Japanese population registry between 50 and 89 years of age. J Hand Surg 27(1):148–155
Sharma BD, Sah DNM (2017) The efficacy of local corticosteroid injection in the treatment of trigger finger. Janaki Med College J Med Sci 5(2):13–18
Lee M, Jung YR, Lee YK (2019) Trigger finger secondary to a neglected flexor tendon rupture. Medicine 98(1):e13980
Sarkar D, Morshed T, Hoque MA (2019) Association of diabetes mellitus with trigger finger. Clin Pract 16(6):1389–1391
Dogru, M., Erduran, M., & Narin, S. (2020). The effect of radial extracorporeal shock wave therapy in the treatment of trigger. Cureus, 12(6)
Saunders S, Fsom F, Longworth S (2018) Injection techniques in musculoskeletal medicine E book: a practical manual for clinicians in primary and secondary care. Elsevier
Romeo P, Lavanga V, Pagani D, Sansone V (2014) Extracorporeal shock wave therapy in musculoskeletal disorders: a review. Med Princ Pract 23:7–13
Ogden JA, Toth-Kischkat A, Schultheiss R (2001) Principles of shockwave therapy. Clin Orthop 387:8–17
Filippucci E, Mandl P, Balint PV, Grassi W (2018) Sonopathology: pathological findings (articular and periarticular). Ultrasonography of the Hand in Rheumatology. Springer, Cham, pp 121–149
Green DP, Hotchkiss RN, Pederson WC (1999) Green’s operative hand surgery, 4th edn. Ghurchill Livingstone, New York
Lin CL, Lee JS, Su WR, Kuo LC, Tai TW, Jou IM (2011) Clinical and ultrasonographic results of ultrasonographically guided percutaneous radiofrequency lesioning in the treatment of recalcitrant lateral epicondylitis. Am J Sports Med 39(11):2429–2435
Guerini H, Pessis E, Theumann N, Le Quintrec JS, Campagna R, Chevrot A et al (2008) Sonographic appearance of trigger fingers. J Ultrasound Med 27(10):1407–1413
Serafini G, Derchi LE, Quadri P, Martinoli C, Orio O, Cavallo A et al (1996) High-resolution sonography of the flexor tendons in trigger fingers. J Ultrasound Med 15(3):213–219
Huang, Q., Yan, P., Xiong, H., Shuai, T., Liu, J., Zhu, L., et al. (2020). Extracorporeal shock wave therapy for treating foot ulcers in adults with type 1 and type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Canadian Journal of Diabetes, 1–9
Fiorini, H. J., Tamaoki, M. J., Lenza, M., dos Santos, J. B. G., Faloppa, F., & Carlos Belloti, J. (2018). Surgery for trigger finger. Cochrane Database of Systematic Reviews, 2
Viviano SL, Chandler LK, Keith JD (2018) Ultrahigh-frequency ultrasound imaging of the hand: a new diagnostic tool for hand surgery. Hand 13(6):720–725
Earp BE (2018) Hand and wrist soft tissue conditions. Principles of Orthopedic Practice for Primary Care Providers. Springer, Cham, pp 207–229
Dala-Ali BM, Nakhdjevani A, Lloyd MA, Schreuder FB (2012) The efficacy of steroid injection in the treatment of trigger finger. Clin Orthop Surg 4(4):263–268
Ko SH, Kim DE, Lee TJ (2022) Steroid injection using tendon excursion for trigger finger: introduction to injection methods and analysis of treatment results. Archives Hand Microsurgery 27(1):33–40
Rydberg M, Zimmerman M, Gottsater A, Eeg-Olofsson K, Dahlin LB (2022) High HbA1c levels are associated with the development of trigger finger in type 1 and type 2 diabetes: an observational register-based study from Sweden. Diabetes Care 45(11):2669–2674
Ryzewiez M, Wolf JM (2006) Trigger digits: principles, management, and complications. J Hand Surg Am 31:135–146
Massoud AA, Fouaad AA, Abdelkareem MM, ElBaqary AMA (2018) Evaluation of the accuracy of trigger finger injection using ultrasound. Egyptian J Hospital Med 73(11):7988–7996
Yildirim P, Gultekin A, Yildirim A, Karahan AY, Tok F (2016) Extracorporeal shock wave therapy versus corticosteroid injection in the treatment of trigger finger: a randomized controlled study. J Hand Surg Eur Vol 41(9):977–983
Acknowledgements
Not applicable.
Funding
This study had no funding from any resource.
Author information
Authors and Affiliations
Contributions
SL wrote the paper with revision. RH followed the patients and collected the patients’ data. SL gave idea, put a study design, and revised the manuscript. NN revised the statistics. NG performed the therapeutic ultrasound before and after intervention and revised the methodology. They all approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Research Ethics Committee (REC) of the Faculty of Medicine at Ain Shams University in Egypt. Reference number of approval: FMASU R 126/ 2023. All patients included in this study gave written informed consent to participate in this research.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Leithy, S.A., Adly, N.N., Taha, R.M. et al. Extra-corporeal shock wave therapy versus local corticosteroid injection in treatment of chronic trigger finger in diabetic patients. Egypt Rheumatol Rehabil 50, 57 (2023). https://doi.org/10.1186/s43166-023-00219-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43166-023-00219-4