Abstract
Background
Infertility is a global problem that refers to the absence of pregnancy after 12 months of intercourse without using contraception. Assisted reproductive techniques (ART) are a treatment technique for infertile patients. Poor ovarian response to stimulation (POR) is one of the challenges in the field of ART. POR refers to people in whom, despite appropriate ovarian stimulation, the number of oocytes retrieved is less than expected. Several ovarian stimulation protocols are applied in POR patients, but the best protocol is not defined. One of the protocols used in POR patients is the "double stimulation in the same ovarian cycle" (DuoStim) protocol. During the DuoStim protocol, both follicular and luteal phases of an ovarian cycle are stimulated. It allows us to retrieve oocytes twice in one ovarian cycle. In the current study, ovarian stimulation in follicular and luteal phases in an ovarian cycle in patients with the poor ovarian response is compared.
Results
Twenty-six infertile patients with poor ovarian enrolled. They had a mean ± standard deviation age of 36.3 ± 3.15 years. The duration of follicular phase stimulation (FPS) was shorter than luteal phase stimulation (LPS) (pvalue = 0.003). The total dose of gonadotropin was significantly lower in FPS than in LPS (pvalue = 0.013). Significantly fewer total oocytes were retrieved after FPS than after LPS (pvalue = 0.001).
Conclusion
This study supports the putative benefits of LPS in infertile women with POR. Utilizing the DuoStim strategy in people with poor ovarian response caused more oocytes to be retrieved.
Trial registration: irct.ir identifier: IRCT20221001056068N1, 11/22/2022, https://www.irct.ir/trial/66187.
Similar content being viewed by others
1 Background
Infertility is a global problem that affects about 80 million couples worldwide [1]. Infertility refers to the absence of pregnancy after 12 months of intercourse without using contraception [2]. Infertility can be primary or secondary, with a prevalence of 2% and 10.5%, respectively [3].
Infertility due to female factors has various causes, including ovarian, anatomical, endocrine, and genetic causes. Assisted reproductive techniques (ART) are a treatment technique for infertile patients [2]. Poor ovarian response to stimulation (POR) is one of the challenges in the field of ART [4].
POR refers to people in whom, despite appropriate ovarian stimulation, the number of oocytes retrieved is less than expected. The prevalence of POR is between 9 and 25% is variable [5]. Accordingly, POR is defined as at least two of the following three characteristics must be present: advanced maternal age (≥ 40 years), a previously characterized POR cycle, or an abnormal ovarian reserve test [5, 6]. The physiology of POR is unknown, and probably oxidative stress and mitochondrial function disorders could be the main reasons [4].
Patient-oriented strategies encompassing individualized oocyte number (POSEIDON) criteria is a new approach to defining POR patients more precisely. It provides the best outlook for diagnosing and treating POR patients. The POSEIDON groups are as follows: The first group includes patients younger than 35 who had a weak response in the previous IVF cycle despite receiving the standard medications. The second group includes patients older than 35 who had a weak response in the previous IVF cycle despite receiving the standard medications. The third group includes patients younger than 35 with abnormal ovarian reserve test (antral follicle count (AFC) less than 5) and anti-Müllerian hormone (AMH) less than 1.2 nanograms/cc. The fourth group includes patients older than 35 with abnormal ovarian reserve test, AFC less than 5, and AMH less than 1.2 ng/cc [3, 4, 7, 8].
Studies show that about 50% of cancellation of cycles in ART occurs in POR people [9, 10]. Also, the number and quality of oocytes retrieved during ovarian stimulation are low. Therefore, a few transferable embryos could be obtained [4]. The pregnancy rate in these people is lower despite IVF [10, 11].
Several ovarian stimulation protocols are applied in POR patients, but the best protocol is not defined [9, 12]. One of the protocols used in POR patients is the "double stimulation in the same ovarian cycle" (DuoStim) protocol. During the DuoStim protocol, both follicular and luteal phases of an ovarian cycle are stimulated [13,14,15]. It allows us to retrieve oocytes twice in one ovarian cycle. Therefore, more oocytes are obtained in less time [9]. Also, the quality of the embryos obtained in the DuoStim protocol is better than the conventional ovarian stimulation methods [16]. In the current study, ovarian stimulation in follicular and luteal phases in an ovarian cycle (DuoStim) in patients with poor ovarian response is compared.
2 Methods
2.1 Study design
This study was conducted as a clinical trial (irct.ir identifier: IRCT20221001056068N1) on 26 infertile patients with poor ovarian response (POR) referred to the infertility center of Shahid Beheshti hospital, Isfahan, Iran. The study was approved by the research ethics committee of the Alzahra Research Centers (IR.ARI.MUI.REC.1401.183), and written informed consent was obtained from all patients.
2.2 Study participants
All patients with POR were assessed to include in the study, regardless of age. Inclusion criteria were: patients with poor response to ovarian stimulation according to POSEIDON 3 and 4 diagnostic criteria and undergoing the necessary treatment protocols for IVF.
Patients with partners suffering from azoospermia, a history of uterine surgery, severe endometriosis, hydrosalpinx, endocrine disorders, repeated abortions, recurrent implantation failures (≥ 3 times), and a body mass index of more than 30 were excluded.
2.3 Oocyte retrieval and fertilization
In the conventional method, ovarian stimulation is performed in just one ovarian phase (follicular or luteal). In the DuoStim protocol, ovarian stimulation is performed in both ovarian phases. From the third day of menstruation (the third day of the follicular phase), patients underwent ovarian stimulation with gonadotropins at a daily dose of 300 units-150 units of human menopausal gonadotropin (HMG) and 150 units of recombinant follicle-stimulating hormone (FSH). When the follicle diameter was higher than 14 mm, daily gonadotropin-releasing hormone (GnRH) antagonist (0.25 mg cetrorelix) was prescribed. Whenever at least two follicles with a diameter of 17 mm were observed through vaginal ultrasound, a single dose of 0.25 mg of human chorionic gonadotropin (hCG) was used to trigger the oocyte growth. Then transvaginal ultrasound-guided oocyte retrieval is performed. On the same day, a semen sample was taken from the partner. Eligible sperms were retrieved and microinjection was performed by an embryologist. Three days later (the third day of the luteal phase), a vaginal ultrasound was performed to check the ovaries for the presence of follicles, and the same drugs were prescribed. Then transvaginal ultrasound-guided oocyte retrieval is performed. On the same day, a semen sample was taken from the partner. Eligible sperms were retrieved and microinjection was performed by an embryologist.
2.4 Outcomes
The outcomes included fertilization rate, number of 2 pro-nuclei cells, follicles > 14 mm in diameter, total and metaphase II oocytes retrieved, gonadotropin dosage, stimulation duration, and quality of embryos in follicular and luteal phases. The quality of 3-day-old embryos was determined by an embryologist. Embryo quality parameters are the number of blastomeres (0, 1, 2, 3, and so on) and morphological characteristics in terms of the degree of fragmentation as 0, ≤ 10, 11–20, 21–50, more than 50% or completely fragmentation [17].
2.5 Statistical analysis
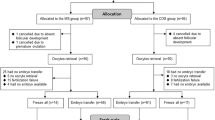
All data were analyzed using the Statistical Package for the Social Sciences for Windows (SPSS, Version 16.0, SPSS Inc., Chicago, IL, USA). Numerical variables were described with mean and standard deviation, and qualitative variables were described with frequency and percentage. Two-mode qualitative variables in two phases were compared using McNemar's test. Numerical variables were checked for normality by Shapiro–Wilk test and then compared according to the result of normality in two phases using paired t-test or its non-parametric equivalent, Wilcoxon. The significance was accepted for P < 0.05 (Fig. 1).
3 Results
In this study, 26 women with POR underwent the DuoStim protocol. They had a mean ± standard deviation (SD) age of 36.3 ± 3.15 years. The baseline characteristics of patients are presented in Table 1.
Approximately, luteal phase stimulation (LPS) was two days longer than follicular phase stimulation (FPS) (11.5 ± 1.6 days vs. 9.7 ± 1.8 days; pvalue = 0.003). The total dose of gonadotropin was significantly higher in LPS than FPS (2052 ± 395 IU vs. 1793.1 ± 272.1 IU; pvalue = 0.013). In FPS, the mean number of follicles bigger than 14 millimeters in diameter was lower than in LPS (3.5 ± 1.4 vs. 4.5 ± 1; pvalue = 0.002). On average, significantly fewer total oocytes were collected after FPS than after LPS (2.7 ± 1.5 vs. 3.9 ± 1.1; pvalue = 0.001).
There were no statistically significant differences in fertilization rate among both ovarian phases stimulation. Additionally, the number of metaphase II oocytes and top-quality day-3 embryos were similar in both FPS and LPS (Table 2).
4 Discussion
For those at risk of losing or rapidly declining fertility, time is a critical factor. Women with malignancy who require gonadotoxic treatment or removal of gonads have a brief time frame to preserve suitable oocytes. The time frame is as limited in patients with poor ovarian response. Nevertheless, more stimulation cycles are required to obtain the appropriate number of oocytes and subsequently top-quality embryos.
In our study, ovarian stimulation was performed by DuoStim protocol in one ovarian cycle in people with poor ovarian response. The duration of ovarian stimulation was shorter and the dose of gonadotropins was lower in the follicular phase than in the luteal phase (pvalue < 0.05). Also, the total amount of oocytes retrieved and the number of follicles with a diameter greater than 14 mm in the luteal phase were significantly higher than in the follicular phase (pvalue < 0.05). Significantly, the number of mature oocytes to the total number of oocytes ratio is higher in the follicular phase than in the luteal phase (pvalue < 0.05). However, fertilization rate, number and quality of the retrieved embryos, and the number of mature oocytes were similar in both ovarian phases.
In a retrospective case–control study, Liu et al. [18] found that the total dosage of gonadotropin and duration of ovarian stimulation was more in FPS than in LPS. However, the differences were not significant. Regarding the duration of ovarian stimulation and the total dose of gonadotropins, previous studies [9, 19, 20] were consistent with our survey. Induction of the similar dosage of GnRH agonist may make the FSH surge and luteinizing hormone (LH) surge much higher in the first trigger than in the second trigger. Notably, the ovarian sensitivity to the HMG stimulation is reduced during the luteal-phase ovarian stimulation. It could be inquired in further studies.
Due to previous stimulation in the follicular phase, patients had higher estrogen and progesterone levels in the luteal phase. Therefore, a significant difference was expected in the total number of retrieved oocytes and the ratio of metaphase 2 oocytes to total oocytes. In studies with stimulation of merely one ovarian phase, due to the absence of cumulative effect of estrogen and progesterone, the number of oocytes in two ovarian phases did not differ significantly. Also, participants in follicular and luteal phase groups were not adjusted for age and ovarian reserve [9, 12]. The number and quality of the embryos and the overall fertility rate of previous studies had similar findings [9, 12, 19, 20].
5 Limitations
Since 26 people participated in this research, it seems that more reliable results would have been obtained if more people participated. In case of follow-up of patients and examination of the number of live fetuses and the rate of abortion, more comprehensive results could be obtained about the effects of Dostim during pregnancy. Considering that all participants were evaluated in the embryology center by a unique embryologist, the data is more reliable than the data of multicenter studies.
6 Conclusion
Utilizing the DuoStim strategy in people with poor ovarian response in this study caused more oocytes to be retrieved in a shorter time frame and patients who needed ovarian stimulation were not placed in repeated ovarian cycles. This study supports the putative benefits of LPS in infertile women with POR. In addition, using GnRH antagonists prevented the effects of premature LH surge and the follicles grew more optimally.
Availability of data and materials
The datasets generated during and/or analyzed during the current study are not publicly available due to hospital ethics policies but are available from the corresponding author on reasonable request.
Abbreviations
- POR:
-
Poor ovarian response
- IVF:
-
In vitro fertilization
- DuoStim:
-
Double stimulation in the same ovarian cycle
- FPS:
-
Follicular phase stimulation
- LPS:
-
Luteal phase stimulation
- POSEIDON:
-
Patient-oriented strategies encompassing individualized oocyte number
- AFC:
-
Antral follicle count
- AMH:
-
Anti-Müllerian hormone
- hMG:
-
Human menopausal gonadotropin
- FSH:
-
Follicle-stimulating hormone
- GnRH:
-
Gonadotropin-releasing hormone
- hCG:
-
Human chorionic gonadotropin
- SPSS:
-
The Statistical Package for the Social Sciences for Windows
- SD:
-
Standard deviation
- BMI:
-
Body mass index
- IU:
-
International unit
References
Marchiani S, Tamburrino L, Benini F et al (2020) LH supplementation of ovarian stimulation protocols influences follicular fluid steroid composition contributing to the improvement of ovarian response in poor responder women. Sci Rep 10:12907. https://doi.org/10.1038/s41598-020-69325-z
Esteves SC, Humaidan P, Roque M, Agarwal A (2019) Female infertility and assisted reproductive technology. Panminerva Med 61(1):1–2. https://doi.org/10.23736/S0031-0808.18.03553-X
Cimadomo D, Vaiarelli A, Colamaria S et al (2018) Luteal phase anovulatory follicles result in the production of competent oocytes: intra-patient paired case-control study comparing follicular versus luteal phase stimulations in the same ovarian cycle. Hum Reprod 33(8):1442–1448. https://doi.org/10.1093/humrep/dey217
Xu Y, Nisenblat V, Lu C et al (2018) Pretreatment with coenzyme Q10 improves ovarian response and embryo quality in low-prognosis young women with decreased ovarian reserve: a randomized controlled trial. Reprod Biol Endocrinol. 16(1):29. https://doi.org/10.1186/s12958-018-0343-0
Özkan ZS (2019) Ovarian stimulation modalities in poor responders. Turk J Med Sci. 49(4):959–962. https://doi.org/10.3906/sag-1905-179
Ferraretti AP, La Marca A, Fauser BC et al (2011) ESHRE consensus on the definition of “poor response” to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod 26(7):1616–1624. https://doi.org/10.1093/humrep/der092
Blumenfeld Z (2020) What is the best regimen for ovarian stimulation of poor responders in ART/IVF? Front Endocrinol (Lausanne). 11:192. https://doi.org/10.3389/fendo.2020.00192
Song JY, Gao DD, Cao XL et al (2021) The role of traditional chinese formula ding-kun pill (DKP) in expected poor ovarian response women (POSEIDON Group 4) undergoing in vitro fertilization-embryo transfer: a multicenter, randomized, double-blind, placebo-controlled trial. Front Endocrinol (Lausanne). 12:675997. https://doi.org/10.3389/fendo.2021.675997
Llácer J, Moliner B, Luque L et al (2020) Luteal phase stimulation versus follicular phase stimulation in poor ovarian responders: results of a randomized controlled trial. Reprod Biol Endocrinol. 18(1):9. https://doi.org/10.1186/s12958-020-00570-7
Simopoulou M, Galatis D, Maziotis E et al (2022) Development of a predictive model for luteal phase oocyte retrieval in poor responders undergoing natural cycle IVF. Sci Rep. 12(1):7695. https://doi.org/10.1038/s41598-022-11602-0
Polat M, Mumusoglu S, Yarali Ozbek I, Bozdag G, Yarali H (2021) Double or dual stimulation in poor ovarian responders: where do we stand? Ther Adv Reprod Health. 15:26334941211024172. https://doi.org/10.1177/26334941211024172
Chen YC, Li JY, Li CJ et al (2021) Luteal phase ovarian stimulation versus follicular phase ovarian stimulation results in different human cumulus cell genes expression: a pilot study. Int J Med Sci. 18(7):1600–1608. https://doi.org/10.7150/ijms.55955
Gica C, Maxim BG, Botezatu R et al (2021) Double ovarian stimulation in the same ovarian cycle. Maedica (Bucur) 16(1):102–106. https://doi.org/10.26574/maedica.2020.16.1.97
Vaiarelli A, Cimadomo D, Petriglia C et al (2020) DuoStim - a reproducible strategy to obtain more oocytes and competent embryos in a short time-frame aimed at fertility preservation and IVF purposes. A systematic review. Ups J Med Sci. 125(2):121–130. https://doi.org/10.1080/03009734.2020.1734694
Glujovsky D, Pesce R, Miguens M, Sueldo CE, Lattes K, Ciapponi A (2020) How effective are the non-conventional ovarian stimulation protocols in ART? A systematic review and meta-analysis. J Assist Reprod Genet 37(12):2913–2928. https://doi.org/10.1007/s10815-020-01966-5
Vaiarelli A, Cimadomo D, Trabucco E et al (2018) Double stimulation in the same ovarian cycle (DuoStim) to maximize the number of oocytes retrieved from poor prognosis patients: a multicenter experience and SWOT analysis. Front Endocrinol (Lausanne). 9:317. https://doi.org/10.3389/fendo.2018.00317
Ziebe S, Lundin K, Janssens R, Helmgaard L, Arce JC; MERIT (Menotrophin vs Recombinant FSH in vitro Fertilisation Trial) Group. Influence of ovarian stimulation with HP-hMG or recombinant FSH on embryo quality parameters in patients undergoing IVF. Hum Reprod. 2007;22(9):2404–2413. https://doi.org/10.1093/humrep/dem221
Liu C, Jiang H, Zhang W, Yin H (2017) Double ovarian stimulation during the follicular and luteal phase in women ≥38 years: a retrospective case-control study. Reprod Biomed Online 35(6):678–684. https://doi.org/10.1016/j.rbmo.2017.08.019
Kuang Y, Chen Q, Hong Q et al (2014) Double stimulations during the follicular and luteal phases of poor responders in IVF/ICSI programmes (Shanghai protocol). Reprod Biomed Online 29(6):684–691. https://doi.org/10.1016/j.rbmo.2014.08.009
Ubaldi FM, Capalbo A, Vaiarelli A et al (2016) Follicular versus luteal phase ovarian stimulation during the same menstrual cycle (DuoStim) in a reduced ovarian reserve population results in a similar euploid blastocyst formation rate: new insight in ovarian reserve exploitation. Fertil Steril 105(6):1488-1495.e1. https://doi.org/10.1016/j.fertnstert.2016.03.002
Acknowledgements
We would like to thank all the participants for taking part in this study and acknowledge Shahid Beheshti Hospital staff for their cooperation.
Funding
None declared.
Author information
Authors and Affiliations
Contributions
All authors have accepted responsibility for the entire content of this manuscript and approved its submission. E.N. conceived of the presented idea. E.N. and R.A.H. designed the study. N.H. and S.S. performed the measurements and gathered the data. V.S. and E.N. were involved in planning and supervised the work. N.B. processed the experimental data, performed the analysis. V.S. designed the tables. All authors discussed the results and commented on the manuscript. S. Salehi took the lead in writing the manuscript with support from E.N. and N.H. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The local Institutional Review Board deemed the study exempt from review. The study was approved by the research ethics committee of the Alzahra Research Centers (IR.ARI.MUI.REC.1401.183). Written informed consent was obtained from all individuals participating in this study.
Consent for publication
Not applicable.
Competing interests
Authors state no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Naghshineh, E., Aalami-harandi, R., Heydari, N. et al. Comparison of ovarian stimulation in follicular and luteal phases in an ovarian cycle (DuoStim) in patients with poor ovarian response. Beni-Suef Univ J Basic Appl Sci 12, 39 (2023). https://doi.org/10.1186/s43088-023-00377-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43088-023-00377-6