Abstract
Background
The principal cause of renal graft loss after the first year is chronic allograft nephropathy which is represented histologically by tubulo-interstitial fibrosis. Its early diagnosis and treatment are crucial to prevent late graft failure. Ultrasound is unequivocally the first-line imaging modality for the evaluation of renal transplants in the immediate postoperative period and for long-term follow-up. Ultrasound shear wave elastography is an imaging technique based on estimation of the elastic properties of tissues.
Elastography is performed in the same clinical setting with conventional B-mode ultrasonography. Tissue elasticity is displayed as an absolute number and color-coded real-time estimation. So, it can be used in screening and diagnosing chronic allograft nephropathy. However, the accurate diagnosis and prognosis of renal parenchymal complications still relies on tissue biopsy. Many studies have proved the high specificity of ultrasound elastography in decreasing the number of unnecessary biopsies.
Results
In our study, we included 36 patients with biopsy-proven chronic allograft nephropathy. All patients had a B-mode ultrasound examination and followed by ultrasound shear wave elastography in the same session. The results were compared to the histopathological results.
Time since transplantation was directly correlated with mean renal stiffness, revealing that with longer time of transplantation renal stiffness and interstitial fibrosis and tubular atrophy (IF/TA) percentage increased with r = 0.72, 0.90 and p value < 0.001.
Antero-posterior (AP) diameter of the renal graft was significantly correlated with mean renal stiffness as the larger the AP diameter, the higher the mean kidney stiffness with r = 0.47, 0.73 and p value 0.001.
Sensitivity analysis showed that US shear wave elastography through mean kidney stiffness can significantly predict moderate Banff score of renal fibrosis using cutoff value 28.67 kPa with sensitivity 87.5%, specificity 90%, AUC 0.91 and p value < 0.001.
Conclusion
Shear wave elastography (SWE) may be useful for the prediction of fibrosis in renal transplant patients, especially in the case of moderate Banff score, where the accuracy reached 87.5% using a cutoff value 28.67 kPa.
We conclude that US SWE can be of great help during the regular follow-up of renal transplant patients. It can act as a screening tool to identify patients with stiffness values that suggest moderate tubulo-interstitial fibrosis, so eventually helping in the early diagnosis, management and help in selecting patients who are candidate for biopsy and in avoiding the repeated unnecessary biopsies for others.
Similar content being viewed by others
Background
The observation of acute graft rejections has decreased during the last two decades by the introduction of more effective immunosuppressive therapy. Long-term renal graft survival, however, has not improved in the same way. The pathophysiology of late graft failure after the first year is still unexplained, and several confluent immunologic and non-immunologic variables are most likely implicated [1].
Unfortunately, a rise in serum creatinine is a late sign for renal graft impairment, and it is frequently detected at a later stage of fibrosis, when the damage becomes permanent [2]. Only after the loss of 60% of renal function, a significant shift in serum creatinine level can be detected [3].
Currently, renal biopsy is the gold standard of diagnosis of graft dysfunction. Protocol biopsies are done on schedule, usually during the first year after transplantation, to closely monitor and early identify any pathological alterations in the absence of clinical events. While renal biopsy is of a great benefit, it is not preferred due to its invasiveness and high cost. Also, it can result in complications such as infections and bleeding [4].
As a result, noninvasive imaging approaches have attracted a lot of attention and research, with the potential to replace or at least reduce the use of biopsy in identifying graft dysfunction [4].
Being a very easy to perform, affordable, and non-nephrotoxic imaging modality, ultrasound is commonly utilized for the early assessment of renal graft function in the postoperative phase as well as for long-term follow-up [5].
Recently, ultrasound elastography, which evaluates the viscoelastic properties of tissue in various organs, was developed to detect parenchymal alterations [6]. For renal stiffness evaluation, several studies using different elastography techniques, such as real-time and shear wave-based elastography (SWE), have been established [7].
SWE technique estimates the speed of shear waves to provide a quantitative estimate of tissue elasticity by generating shear waves at a focal point in the tissue, where the velocity of the wave provides an estimate of tissue stiffness [7, 8]. Because of the benefits of SWE and B-mode imaging guiding, this technique can give a more precise evaluation of tissue stiffness [9].
SWE works on the basis of a radiation force generated into tissues by focused ultrasonic beams and a very high frame rate US imaging sequence capable of capturing the propagation of resultant shear waves in real time [10, 11].
The US system captures the generated shear waves, to capture them in sufficient details, frame rates of a few thousand images per second are required. This ultrafast imaging technique captures raw data at a high frame rate of up to 5000 frames per second [12].
The shear wave speed is then calculated across a specific region of interest (ROI). The tissue stiffness is calculated using the shear wave speed [9, 10, 12].
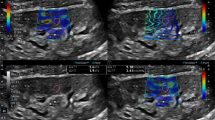
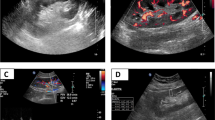
The mean value of KS is calculated and expressed in kilopascals (kPa) and considered indicative of fibrosis (Figs. 1 and 2).
E = 3ρ Vs2, where E is Young’s modulus (kPa), ρ is tissue density (kg/m3), and Vs indicates shear wave velocity (m/s) [11, 13, 14]. In order to calculate Young’s modulus, SWE uses for referencing the density of water (1000 kg/m3). The elasticity is represented by a color-coded image displayed on a B-mode image, where the stiffer tissues are shown in red and softer tissues are shown in blue (Fig. 1) [9, 12].
A low speed denotes a soft medium, whereas a high speed denotes a stiff medium.
The aim of our study is to find a correlation between the renal allograft parenchymal stiffness through ultrasound shear wave elastography and the histopathological changes described in patients with biopsy-proven chronic allograft nephropathy (CAN).
Methods
All patients wrote a prior consent of inclusion in the study. All patients were subjected to full history taking, clinical examination, laboratory investigation, and graft evaluation using a conventional ultrasound examination and shear wave elastography examination.
Selection of the patients
Inclusion criteria
Recipients of living donor renal transplants of any sex and age-group above 18 years old, with diagnosis of chronic allograft nephropathy as evidenced by histopathological findings.
Exclusion criteria
Age range: below 18 years old, time less than 3 months out from transplant surgery, presence of perinephric fluid collections, hydronephrosis, renal cysts, or other parenchymal disease.
Technique of supersonic shear wave elastography
Ultrasound of the graft and SWE examinations were done using TOSHIBA Aplio 500 machine. Initially, B-mode ultrasound examination was performed for renal allograft with (6C1) curvilinear probe and then shear wave elastographic examination using (14L5) linear probe.
Patients were examined in the supine position. The transducer was covered with ultrasound gel and placed on the skin in a stable position for about 3 s, perpendicularly to better minimize the compression artifact, as shear wave elastography relies on the automatic generation and analysis of shear waves.
Elasticity was then qualitatively assessed using color-coded image superimposed on a B-mode image. At the same time, kidney stiffness (KS) was quantitatively measured; the mean KS value in the region of interest (ROI), as well as the standard deviation (SD) of the measured elasticity, was displayed on the screen and expressed in kilopascals (KPa).
When the cineloop was stable showing parallel lines or parallel circles, we froze the image. Ten measurements of KS using a pre-determined circular regions of interest (ROI = 5 mm) were placed at the nearest region from the skin with a well-represented renal parenchyma.
The mean value of KS was calculated and expressed in kilopascals (kPa) and considered indicative of fibrosis. The information was quantified either as shear wave velocity or as Young’s modulus value expressed in kilopascal (kPa).
The statistical methods
Data were coded and entered using the Statistical Package for the Social Sciences (SPSS) version 26 (IBM Corp., Armonk, NY, USA). Data were summarized using mean, standard deviation, median, minimum, and maximum in quantitative data and using frequency (count) and relative frequency (percentage) for categorical data. Comparisons between quantitative variables were made using the nonparametric Mann–Whitney test. For comparing categorical data, chi-square test was performed. Exact test was used instead when the expected frequency is less than 5. Correlations between quantitative variables were done using Spearman correlation coefficient. ROC curve was constructed with area under curve analysis performed to detect best cutoff value of elastography for detection of fibrosis. p values less than 0.05 were considered as statistically significant.
Results
Thirty-six renal transplant recipients with biopsy-proven chronic allograft nephropathy histological findings, after at least 3 months since transplantation, were recruited. The study was conducted from February 2021 to August 2021.
Renal allografts were biopsied by US-guided interventional procedures by true cut needle biopsy (via 18-gauge automated biopsy device).
Chronic pathological findings (IF/TA) were then interpreted and categorized based on Banff classification. Grade I: Mild interstitial fibrosis (6–25% of cortical area) and mild tubular atrophy (≤ 25% of the area of the cortical tubules). Grade II: Moderate interstitial fibrosis (26–50% of cortical area) and moderate tubular atrophy (26–50% of the area of the cortical tubules). Grade III: Severe interstitial fibrosis (< 50% of cortical area) and severe tubular atrophy (< 50% of the area of the cortical tubules).
Renal allografts mean stiffness value was correlated with the histopathological findings (Table 1). On the basis of the histological examinations of the 36 patients who underwent biopsy, there were 20 patients with mild chronic interstitial fibrosis and tubular atrophy (IF/TA) (55.6%) and 16 patients with moderate–severe chronic IF/TA (44.4%).
IF/TA percentage was significantly higher among patients with moderate–severe Banff score when compared to patients with mild Banff score, with p value < 0.001.
IF/TA percentage was positively correlated with mean kidney stiffness with r = 0.57 and p value 0.013, which refer to the higher the stiffness of the renal transplant, the higher the IF/TA percentage (Fig. 3).
Time since transplantation was directly correlated with mean renal stiffness, revealing that with longer time of transplantation renal stiffness and IF/TA percentage increased with r = 0.72, 0.90 and p value < 0.001 (Fig. 4).
The graft antero-posterior diameter was significantly correlated with mean renal stiffness as the larger the AP dimension, the higher the mean kidney stiffness with r = 0.47, 0.73 and p value 0.001 (Fig. 5).
Sensitivity analysis showed that US shear wave elastography through mean kidney stiffness can significantly predict moderate Banff score of renal fibrosis (IF/TA) using cutoff value 28.67 kPa with sensitivity 87.5%, specificity 90%, AUC 0.91, and p value < 0.001.
Discussion
Chronic allograft nephropathy (CAN) is the most common cause of graft failure beyond the first year after renal transplantation and is highly prevalent in renal transplant recipients, with moderate to severe chronic allograft nephropathy [15].
The accurate diagnosis and early treatment of CAN are important for renal allograft salvage. Histologically, CAN is defined by interstitial fibrosis and tubular atrophy (IF/TA) which is further categorized according to Banff scoring system into three grades based to severity (1 = mild, 2 = moderate, and 3 = severe) [2].
Renal biopsy is the gold standard for diagnosis. However, biopsy cannot always be performed, for both anatomical and coagulation-related issues; moreover, major complications have to be always considered; therefore, new noninvasive methods have to be developed to obtain new data on the cause of graft dysfunction [16].
Elastography has emerged as a new technique that uses an intrinsic tissue property, the elasticity of which is the capacity to deform and to return to the initial shape when a stress is applied [16].
Supersonic shear wave elastography (SWE) has been documented to be strongly correlated with grade of fibrosis, providing an assessment of the tissue's stiffness [8, 17].
In our study, IF/TA percentage was positively correlated with SWE mean kidney stiffness with r = 0.57 and p value 0.013, which means that as renal parenchymal stiffness increases, the percentage of interstitial fibrosis and tubular atrophy (IF/TA) also increases.
Our results were consistent with the results reported by Chiocchini et al. and Yang et al. who stated that when the histological features of the Banff classification were categorized (mild vs moderate/severe), renal stiffness was higher in the presence of moderate/severe injury rather than in mild injury for interstitial fibrosis and tubular atrophy [2, 18].
Our results revealed that the time since transplantation was directly correlated with IF/TA percentage and mean renal stiffness, revealing that the longer the time since transplantation, the more stiffness, and the higher IF/TA percentage of the renal allograft with r = 0.72, 0.90 and p value 0.001, < 0.001, respectively, which is consistent with studies conducted on a similar group of patients highlighting that longer time since transplantation (> 12 months) had higher IF/TA percentage and renal stiffness [2, 20].
In the current study, the antero-posterior diameter of the renal graft was significantly correlated with IF/TA percentage and mean kidney stiffness, as the greater the antero-posterior diameter, the higher IF/TA percentage and mean kidney stiffness with r = 0.47, 0.73 and p value 0.048 and 0.001, respectively. This correlation was not evident in the study conducted by Yang et al. that included 82 patients with renal transplantation with p value > 0.05 [18].
In our study, sensitivity analysis showed that SWE mean kidney stiffness can significantly predict grade II Banff score with moderate renal fibrosis (IF/TA) using cutoff 28.67 kPa with sensitivity 87.5%, specificity 90%, AUC 0.91, and p value < 0.001.
Our results were consistent with a Chinese study conducted on 31 patients who showed renal dysfunction post-renal transplantation. Their results revealed that using cutoff 29.01 kPa significantly predicted grade 2–3 Banff score with moderate–severe renal fibrosis with sensitivity 93.5%, specificity 91.7%, and AUC 0.97 [18].
Our results also found that SWE mean kidney stiffness was able to distinguish between the grade I and grade II Banff score with mild-to-moderate interstitial fibrosis and tubular atrophy. However, we could not effectively differentiate between the grade II and grade III groups with moderate-to-severe IF/TA, probably due to insufficient number of patients with grade III Banff score. These results were consistent with the results reported by López Novoa et al. (19).
We did not have a patient group of grade III Banff score with severe interstitial fibrosis and tubular atrophy. This factor was likely the result of the fact that these patients were detected by serological results and had clinical deterioration compatible with graft failure, therefore precluding the need for biopsy.
Conclusion
Shear wave elastography may be useful for the prediction of fibrosis in renal transplant patients, especially in the case of moderate Banff score, when the accuracy reaches 87.5% using a cutoff value 28.67 kPa.
At this time, US shear wave elastography cannot replace renal biopsy, as it is still under investigation, and a standard technique for performing examinations should be established.
However, we think that US shear wave elastography (SWE) can be of great help during the regular follow-up of renal transplant patients. It can act as a screening tool to identify patients with stiffness values that suggest moderate tubulo-interstitial fibrosis, so eventually helping in the early diagnosis and management and help in selecting patients who are candidate for biopsy and in avoiding the repeated unnecessary biopsies for others.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AR:
-
Acute rejection
- CAN:
-
Chronic allograft nephropathy
- Fig:
-
Figure
- IF/TA:
-
Interstitial fibrosis and tubular atrophy
- kPa:
-
Kilopascals
- KS:
-
Kidney stiffness
- ROI:
-
Region of interest
- SWE:
-
Shear wave elastography
- UE:
-
Ultrasound elastography
- US:
-
Ultrasound
References
El Ters M, Grande JP, Keddis MT, Rodrigo E, Chopra B, Dean PG, Cosio FG (2013) Kidney allograft survival after acute rejection, the value of follow-up biopsies. Am J Transplant 13(9):2334–2341
Chiocchini ALC, Sportoletti C, Comai G, Brocchi S, Capelli I, Baraldi O, Bruno P, Conti F, Serra C, Meola M, Zompatori M, La Manna G (2017) Correlation between renal cortical stiffness and histological determinants by point shear-wave elastography in patients with kidney transplantation. Prog Transplant 27(4):346–353
Ishikawa Y, Akasaka Y, Kiguchi H, Akishima-Fukasawa Y, Hasegawa T, Ito K, Ishii T (2006) The human renal lymphatics under normal and pathological conditions. Histopathology 49(3):265–273
Hollis EM (2017) Diffusion-weighted magnetic resonance imaging in diagnosing graft dysfunction: a non-invasive alternative to renal biopsy. Electronic Theses and Dissertations. Paper 2661. https://ir.library.louisville.edu/etd/2661/
Lukenda V, Mikolasevic I, Racki S, Jelic I, Stimac D, Orlic L (2014) Transient elastography: a new noninvasive diagnostic tool for assessment of chronic allograft nephropathy. Int Urol Nephrol 46(7):1435–1440
Ferraioli G, Tinelli C, Dal Bello B, Zicchetti M, Filice G, Filice C, Liver fibrosis Study Group (2012) Accuracy of real time shear wave elastography for assessing liver fibrosis in chronic hepatitis C: A pilot study. Hepatology 56(6):2125–2133
Leong SS, Wong JHD, Md Shah MN, Vijayananthan A, Jalalonmuhali M, Chow TK, Sharif NHM, Ng KH (2021) Shear wave elastography accurately detects chronic changes in renal histopathology. Nephrology 26(1):38–45
Xia S, Ren X, Ni Z, Zhan W (2019) A noninvasive method—Shear-wave elastography compared with transient elastography in evaluation of liver fibrosis in patients with chronic hepatitis B. Ultrasound Q 35(2):147–152
Ferraioli G, Parekh P, Levitov AB, Filice C (2014) Shear wave elastography for evaluation of liver fibrosis. J Ultrasound Med 33(2):197–302
Bamber J, Cosgrove D, Dietrich CF, Fromageau J, Bojunga J, Calliada F, Cantisani V, Correas JM, D’Onofrio M, Drakonaki EE, Fink M, Friedrich-Rust M, Gilja OH, Havre RF, Jenssen C, Klauser AS, Ohlinger R, Saftoiu A, Schaefer F, Sporea I, Piscaglia F (2013) EFSUMB guidelines and recommendations on the clinical use of ultrasound elastography. Part 1: Basic principles and technology. Ultraschall Med 34:169–184
Muller M, Aït-Belkacem D, Hessabi M, Gennisson J-L, Grangé G, Goffinet F, Lecarpentier E, Cabrol D, Tanter M, Tsatsaris V (2015) Assessment of the cervix in pregnant women using shear wave elastography: a feasibility study. Ultrasound Med Biol 41(11):2789–2797
Sporea I, Bota S, Gradinaru-Tascau O, Sirli R, Popescu A (2014) Comparative study between two point shear wave elastographic techniques: acoustic radiation force impulse (ARFI) elastography and ElastPQ. Med Ultrason 16(4):309–314
Yang JR, La Q, Ding XM, Song Y, Wang YY (2020) Using real-time sound touch elastography to monitor changes in transplant kidney elasticity. Clin Radiol 75(12):963-e1
Ferraioli G, Wong VWS, Castera L, Berzigotti A, Sporea I, Dietrich CF, Barr RG (2018) Liver ultrasound elastography: an update to the world federation for ultrasound in medicine and biology guidelines and recommendations. Ultrasound Med Biol 44(12):2419–2440
Ghonge NP, Mohan M, Kashyap V, Jasuja S (2018) Renal allograft dysfunction: evaluation with shear-wave sonoelastography. Radiology 288(1):146–152
Yoo MG, Jung DC, Oh YT, Park SY, Han K (2017) Usefulness of multiparametric ultrasound for evaluating structural abnormality of transplanted kidney: Can we predict histologic abnormality on renal biopsy in advance? Am J Roentgenol 209(3):W139–W144
Dong F, Wu H, Zhang L, Tian H, Liang W, Ye X, Xu J (2019) Diagnostic performance of multimodal sound touch elastography for differentiating benign and malignant breast masses. J Ultrasound Med 38(8):2181–2190
Yang JR, Wang FQ, Li N, Pan WQ, Ai H (2016) Application value of shear wave elastography technique in diagnosis of chronic allograft nephropathy. Int J Clin Exp Med 9(6):9449–9454
López-Novoa JM, Rodríguez-Peña AB, Ortiz A, MartínezSalgado C, Hernández FJL (2011) Etiopathology of chronic tubular, glomerular and renovascular nephropathies: clinical implications. J Transl Med 9(1):1–26
Arndt R, Schmidt S, Loddenkemper C, Grünbaum M, Zidek W, Van Der Giet M, Westhoff TH (2010) Noninvasive evaluation of renal allograft fibrosis by transient elastography - A pilot study. Transpl Int 23(9):871–877
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
NRB contributed to results and statistics and manuscript writing. AAH helped in the idea of the research and revising the manuscript. AEE contributed to the idea of the research, collecting cases, and manuscript writing. MFM provided results and statistics revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Research Ethics Committee of the Faculty of Medicine at Cairo University in Egypt. Written informed consent was signed by all patients who participated in this study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Barsoum, N.R., Elsisy, A.E., Mohamed, M.F. et al. Role of shear wave elastography in assessment of chronic allograft nephropathy. Egypt J Radiol Nucl Med 53, 100 (2022). https://doi.org/10.1186/s43055-022-00778-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43055-022-00778-0