Abstract
Background
Wearable sensors have become a complementary means for evaluation of body function and gait in lower limb osteoarthritis. This study aimed to review the applications of wearable sensors for gait analysis after total knee arthroplasty (TKA).
Methods
Five databases, including Web of Science Core Collection, Embase, Cochrane, Medline, and PubMed, were searched for articles published between January 2010 and March 2023, using predetermined search terms that focused on wearable sensors, TKA, and gait analysis as broad areas of interest.
Results
A total of 25 articles were identified, involving 823 TKA patients. Methodologies varied widely across the articles, with inconsistencies found in reported patient characteristics, sensor data and experimental protocols. Patient-reported outcome measures (PROMs) and gait variables showed various recovery times from 1 week postoperatively to 5 years postoperatively. Gait analysis using wearable sensors and PROMs showed differences in controlled environments, daily life, and when comparing different surgeries.
Conclusion
Wearable sensors offered the potential to remotely monitor the gait function post-TKA in both controlled environments and patients’ daily life, and covered more aspects than PROMs. More cohort longitudinal studies are warranted to further confirm the benefits of this remote technology in clinical practice.
Similar content being viewed by others
Background
Knee osteoarthritis (KOA) is a common degenerative joint disease affecting articular cartilage, menisci, capsule, and other soft tissues [1]. It significantly reduces the quality of life in approximately 10% of KOA patients aged over 60 years [2]. Total knee arthroplasty (TKA) is the most effective treatment for severe knee joint diseases, but selecting an appropriate tool for the assessment of postoperative outcomes is challenging [3].
The patient-reported outcome measures (PROMs) are the commonly used questionnaires to assess postoperative knee pain, keen function, patient satisfaction, etc. However, PROMs should be used in randomized controlled trials due to inherent ceiling effects, poor patient-clinician communication, and unrepeatable results [2]. Optical gait analysis is an objective and quantitative tool to provide detailed kinematic measurements, but the complexity, cost and inconvenience associated with the method impede its widespread application in clinical practice [4]. Wearable sensors are miniaturized and low-cost monitoring devices for real-time detection of movements and posture. Biomechanical data can be collected from accelerometers, gyroscopes, and magnetometers attached to different parts of the body [5]. Therefore, gait analysis, in combination with the use of wearable sensors provides a convenient, efficient, and inexpensive means for data collection, allowing for high-accuracy gait feature extraction for analysis [6]. To our knowledge, only a few systematic reviews [7, 8] have pooled the available evidence for the assessment of post-TKA rehabilitation by combining the technologies, but they have not focused on TKA or gait analysis.
This systematic review aimed to present more data about the application of gait analysis in combination with wearable sensor technologies in post-TKA rehabilitation.
Materials and methods
Inclusion and exclusion criteria
The scoping review was conducted in accordance with the preferred reporting items of the meta-analysis (PRISMA) guidelines [9]. Articles that satisfied all of the following criteria were included in this study: (1) gait analyses with wearable sensors; (2) post-TKA management; and (3) studies published between January 2010 and March 2023. Articles that met one of the following criteria were excluded (1) conference abstracts; (2) review articles; (3) non-TKA treatment; (4) the lack of gait or biomechanical data; (5) studies on technological evaluation; (6) the lack of wearable sensors; (7) robot-assisted rehabilitation or the use of a surgical navigation system; (8) non-independent walking; and (9) the absence of full text.
Search and selection strategies
A systematic search was conducted in the Web of Science Core Collection, Embase, Cochrane, Medline and PubMed. The pre-determined search terms for this review were: wearable electronic devices, total knee arthroplasty and gait analysis. Search strategies for each database were detailed in Appendix. In addition, other relevant articles were also searched in order to find relevant references.
Upon comprehensive searching, duplicate articles were automatically removed using the Endnote software, and the duplicates were verified by the first author (Y.G.F.). Using the software, two authors (Y.G.F. and Y.L.) selected the articles by reviewing the titles and abstracts. The articles were finally confirmed on the basis of review of the main text. The first author (Y.G.F.) collected the data from the main text of the articles. The data were validated by the second author (Y.L.). Disagreements were resolved by comparing notes and reaching a consensus between the two authors (Y.G.F. and Y.L.) and the third author (Y.X.).
Results
Selected articles
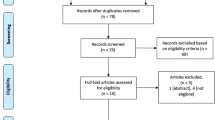
We identified 542 articles (537 articles from the databases and 5 articles from other sources) by using the aforementioned search strategy. We removed 156 duplicate articles. We excluded 361 articles according to the exclusion criteria. Finally, 25 articles were included in the study. The PRISMA flowchart is shown in Fig. 1.
Patient characteristics
We finally identified 25 articles (823 patients), including unilateral [10,11,12,13,14,15,16,17,18,19,20,21], bilateral [14], cruciate-retaining [4] and posterior-stabilized [4, 18], bi-condylar [22], bi-cruciate retaining and bi-cruciate stabilized [23, 24], fixed-bearing prostheses and mobile-bearing prostheses [25], medial-pivot [18], posterior stabilized [24], minimally invasive [26], revision [17] and primary TKA [11, 16, 17, 27, 28], as well as “unknown techniques” [29,30,31] (Table 2).
Sensors
Sensory data varied widely (Table 1). Sampling frequencies ranged from 32 Hz to 1,149 Hz. Frequencies were less than 100 Hz in 2 articles [11, 30]. No frequency was reported in 7 studies [21, 23,24,25,26, 28, 31]. The most common location of sensors was the torso (n = 17) [4, 11, 15, 18,19,20,21,22,23,24,25,26,27,28,29,30, 32], followed by the foot (n = 7) [12, 15, 17, 21, 22, 24, 29], thigh (n = 7), lower leg (n = 6) [13, 16, 22, 25, 31, 33] and head (n = 1) [18].
Study designs
Walking was different in 25 articles. Non-walking protocols were reported in 4 articles [11, 14, 32, 33]. Walking distances ranged from 10 to 100 m in 19 articles. 6-min walk time was reported in 4 articles [10, 13, 20, 28]. Furthermore, the most common experimental environment was indoors (n = 19), followed by outdoors (n = 4) [10, 11, 14, 32], and then by both indoors and outdoors (n = 2) [17, 20]. The study design is detailed in Table 1.
PROMs and gait outcomes
More than one PROMs were employed in most of the 25 articles. The most common PROMs were EuroQol Five-Dimensions Questionnaire (EQ-5D), Knee Injury and Osteoarthritis Outcomes Score (KOS), Knee Society Score (KSS), Oxford Knee Score (OKS), Visual Analogue Scale (VAS), and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) (Table 2).
Gait parameters included the length, width, speed, and frequency of stride. Other parameters included the range of motion (ROM) of the knee (n = 9) [10, 16, 22, 24, 25, 28, 29, 31, 33], acceleration (n = 8) [13, 15, 18, 26, 28,29,30, 32], the step time symmetry (n = 6) [4, 12, 18, 19, 27, 29], coefficient of variability (n = 6) [4, 16, 22, 23, 27, 29] and kinetic parameters (n = 1) [10] (Table 1).
In 13 studies, PROMs improved from the second postoperative week [19] to the 12th postoperative month [4, 17, 22, 27, 29, 31], but gait analysis showed that improvement varied with the different follow-up time points. Storey et al. [28] reported improvement beginning at the 1st-month post-surgery, while Senden et al. [29] observed the change from the 2nd month postoperatively, and yet Tsuji et al. [19] started their observation from the 3rd month. Besides, Çankaya et al. [30] and Tomite et al. [13] pointed out that the patient’s gait returned to normal level 6 months after operation but the notion was not supported by some articles [15, 17, 21]. In most studies, gait parameters improved at 12 months after surgery [4, 22, 27, 29, 30], but Kluge et al. [17] and Rahman et al. [31] didn’t agree with the findings (Table 2).
In 6 comparison articles [12, 18, 23,24,25,26], wearable sensors generated different gait parameters in different groups, and 2 of the articles reported PROMs [24, 25].
Improved PROMs were found at all follow-up time points. In 6 articles [10, 11, 14, 20, 32, 33], the findings of remote gait assessment differed but all sensors captured the changes in gait parameters at all follow-up time points (Table 2).
Time points
In the articles, gait was evaluated from 1 week to 5 years postoperatively, with the most common follow-up time points being 6 weeks, 3 months, 6 months and 1 year after surgery. However, in few studies, follow-up lasted for more than 1 year postoperatively (Table 2).
Discussion
In this review study, we extracted post-TKA data about patient features, sensor data, study protocols, PROMs, and gait variables at various follow-up time points. However, so far, no standard testing method is available for the assessment of wearable sensor-based gait. Kinematic parameters are the most common gait variables. The post-TKA PROMs showed continuous improvement from 1 week to 5 years, but wearable sensors-based gait outcomes varied substantially with different testing protocols used and other relevant factors [34]. In addition, surgeons can track a wide range of daily gait parameters using remote wearable sensors. These parameters are more sensitive and objective than PROMs. Improvement in PROMs is not consistent across the gait parameters due to the lack of high correlation [4, 16, 19, 21, 27]. In addition, wearable sensor assessments show different TKA techniques produce different gait parameters, and some are not covered by PROMs [12, 23, 26, 27]. Those findings suggested that the functional assessment using PROMs may not accurately reflect a patient’s true mobility, and wearable sensor-based gait assessment can serve as a supplement to make up for the PROM insufficiency [35].
As the most important biomechanical assessment, kinematic analysis is effective in interpreting and predicting the recovery of postoperative movement [36,37,38]. However, we found that different post-TKA kinematic parameters were used in the follow-up periods. We found many articles reporting the accuracy, consistency, and responsiveness of wearable sensor-based gait analysis, but they mainly focused on the evaluation of healthy gait based on inconsistent protocols [39,40,41,42]. Item-Glatthorn et al. [43] took issue with the use of certain gait parameters (such as walking speed and stride length) since the sensors, test methods, and parameter definitions restricted the comparability of the findings [7]. Hafer et al. [44] also expressed concerns about those inconsistent and unreliable protocols. Similarly, Kobsar et al. [7] suggested that the reliability of wearable devices be verified. Therefore, it is imperative to establish a standardized and generally-accepted testing protocol to yield reliable and comparable results.
As important gait indicators, kinetic parameters are used in gait training and surgical planning [37, 45, 46]. Surprisingly, no article reported the use of wearable system-based kinematic parameters for the assessment of post-TKA gait. Emery et al. emphasized avoiding this limitation and making wearable systems more accessible in clinical practice [1]. Currently, many studies examined the feasibility of dynamic assessments using kinematic parameters. Youn et al. [47] extracted 11 inertial gait variables from accelerometers and successfully predicted four kinetic gait variables (maximum knee flexion moment, maximum knee inversion moment, vertical ground reaction force, and maximum ground reaction force). Konrath et al. [48] proposed a musculoskeletal model based on the data derived from wearable sensors. They assessed knee movement in older adults during activities of daily living. They found that the accuracy of internal knee moments measured using wearable sensors was comparable to that of optical motion capture. He et al. developed a wearable sensory training system and successfully predicted the changes in knee internal joint moments during a walking test in elderly KOA patients [49]. In summary, the relevant articles confirmed that joint torque and related load estimation methods were valid on the basis of wearable sensors, and they provided a novel approach for assessing dynamic parameters and led to improved gait training and surgical planning.
The “white coat effect” was observed in rehabilitation assessments, where gait movements conducted in the presence of surgeons or researchers differed from those conducted in their absence [17]. Emmerzaal et al. [30] reported gait differences between the clinical settings and daily environments. PROMs may not represent daily gait behaviors. Wearable sensors have the advantage of allowing physicians to perform remote unsupervised assessments, both in and out of the clinic, throughout the rehabilitation process [50, 51]. Our results suggest that remote measurement using wearable sensors is more informative than PROMs in terms of a patient’s daily gait. It reduces the number of patients’ clinical visits and optimizes rehabilitation training [52]. In order to assess daily gait function better, Chapman et al. [33] suggested that post-TKA rehabilitation and follow-up periods should be longer than 1 to 2 years [32].
Our review has limitations. First, the small patient groups in some articles and different testing methods compromised the power of the evidence in assessing walking ability after TKA. In the future, wearable sensors may be used to monitor real-life physical activities and gait outcomes in TKA. Second, PROMs and gait parameters are subject to some limitations, which affect measurement accuracy.
Conclusion
This systematic review confirmed that wearable sensors can be used to monitor post-TKA gait function in unsupervised mode and on remote basis, providing additional clinical measurement methods and diagnostic approaches. More longitudinal cohort studies using wearable sensors could help further improve the assessment of gait function and post-TKA rehabilitation.
Availability of data and materials
The dataset analyzed in this study is available from the corresponding author on reasonable request.
References
Emery CA, Whittaker JL, Mahmoudian A, Lohmander LS, Roos EM, Bennell KL, et al. Establishing outcome measures in early knee osteoarthritis. Nat Rev Rheumatol. 2019;15(7):438–48. https://doi.org/10.1038/s41584-019-0237-3.
Stevens-Lapsley JE, Schenkman ML, Dayton MR. Comparison of self-reported knee injury and osteoarthritis outcome score to performance measures in patients after total knee arthroplasty. PM R. 2011;3(6):541–9. https://doi.org/10.1016/j.pmrj.2011.03.002.
Marmon AR, Zeni JA, Snyder-Mackler L. Comparing younger and older patients after TKA: impairments, functional performance and pain. Osteoarthritis Cartilage. 2016;24:S475–6. https://doi.org/10.1016/j.joca.2016.01.869.
Bolink S, Grimm B, Heyligers IC. Patient-reported outcome measures versus inertial performance-based outcome measures: A prospective study in patients undergoing primary total knee arthroplasty. Knee. 2015;22(6):618–23. https://doi.org/10.1016/j.knee.2015.04.002.
Ma CC, Li WF, Cao JJ, Du J, Li QM, Gravina R. Adaptive sliding window based activity recognition for assisted livings. Inf Fusion. 2020;53:55–65. https://doi.org/10.1016/j.inffus.2019.06.013.
Kobsar D, Charlton JM, Tse CT, Esculier J-F, Graffos A, Krowchuk NM, et al. Validity and reliability of wearable inertial sensors in healthy adult walking: a systematic review and meta-analysis. J Neuroeng Rehabil. 2020;17:1–21. https://doi.org/10.1186/s12984-020-00685-3.
Kobsar D, Masood Z, Khan H, Khalil N, Kiwan MY, Ridd S, et al. Wearable inertial sensors for gait analysis in adults with osteoarthritis-A scoping review. Sensors. 2020;20(24):7143. https://doi.org/10.3390/s20247143.
Small SR, Bullock GS, Khalid S, Barker K, Trivella M, Price AJ. Current clinical utilisation of wearable motion sensors for the assessment of outcome following knee arthroplasty: a scoping review. BMJ Open. 2019;9(12):e033832. https://doi.org/10.1136/bmjopen-2019-033832.
Moher D, Liberati A, Tetzlaff J, Altman DG, Grp P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010;8(5):336–41. https://doi.org/10.1016/j.ijsu.2010.02.007.
Bolam SM, Batinica B, Yeung TC, Weaver S, Cantamessa A, Vanderboor TC, et al. Remote patient monitoring with wearable sensors following knee arthroplasty. Sensors (Basel). 2021;21(15):5143. https://doi.org/10.3390/s21155143.
Brandes M, Ringling M, Winter C, Hillmann A, Rosenbaum D. Changes in physical activity and health-related quality of life during the first year after total knee arthroplasty. Arthritis Care Res (Hoboken). 2011;63(3):328–34. https://doi.org/10.1002/acr.20384.
Cankaya D, Akti S, Unal SB, Sezgin EA. Unicompartmental knee arthroplasty results in a better gait pattern than total knee arthroplasty: gait analysis with a smartphone application. Jt Dis Relat Surg. 2021;32(1):22–7. https://doi.org/10.5606/ehc.2021.79635.
Christiansen CL, Bade MJ, Paxton RJ, Stevens-Lapsley JE. Measuring movement symmetry using tibial-mounted accelerometers for people recovering from total knee arthroplasty. Clin Biomech (Bristol, Avon). 2015;30(7):732–7. https://doi.org/10.1016/j.clinbiomech.2015.04.013.
Daugaard R, Tjur M, Sliepen M, Lipperts M, Grimm B, Mechlenburg I. Are patients with knee osteoarthritis and patients with knee joint replacement as physically active as healthy persons? J Orthop Translat. 2018;14:8–15. https://doi.org/10.1016/j.jot.2018.03.001.
Hiyama Y, Asai T, Wada O. Effect of improvement in quadriceps strength asymmetry on trunk movement asymmetry after total knee arthroplasty. Gait Posture. 2020;79:21–5. https://doi.org/10.1016/j.gaitpost.2020.04.008.
Hiyama Y, Asai T, Wada O, Maruno H, Nitta S, Mizuno K, et al. Gait variability before surgery and at discharge in patients who undergo total knee arthroplasty: a cohort study. PLoS One. 2015;10(1):e0117683. https://doi.org/10.1371/journal.pone.0117683.
Kluge F, Hannink J, Pasluosta C, Klucken J, Gassner H, Gelse K, et al. Pre-operative sensor-based gait parameters predict functional outcome after total knee arthroplasty. Gait Posture. 2018;66:194–200. https://doi.org/10.1016/j.gaitpost.2018.08.026.
Lo CWT, Brodie MA, Tsang WWN, Lord SR, Yan CH, Wong AYL. Pain, balance, and mobility in people 1 year after total knee arthroplasty: a non-randomized cross-sectional pilot study contrasting posterior-stabilized and medial-pivot designs. Pilot Feasibility Stud. 2022;8(1):131. https://doi.org/10.1186/s40814-022-01094-0.
Senden R, Grimm B, Meijer K, Savelberg H, Heyligers IC. The importance to including objective functional outcomes in the clinical follow up of total knee arthroplasty patients. Knee. 2011;18(5):306–11. https://doi.org/10.1016/j.knee.2010.07.008.
Storey AS, Myrah AM, Bauck RA, Brinkman DM, Friess SN, Webber SC. Indoor and outdoor mobility following total knee arthroplasty. Physiother Can. 2013;65(3):279–88. https://doi.org/10.3138/ptc.2012-36
Zhang HH, Yan SH, Fang C, Guo XY, Zhang K. Clinical evaluation and gait characteristics before and after total knee arthroplasty based on a portable gait analyzer. Orthop Surg. 2016;8(3):360–6. https://doi.org/10.1111/os.12270.
Calliess T, Bocklage R, Karkosch R, Marschollek M, Windhagen H, Schulze M. Clinical evaluation of a mobile sensor-based gait analysis method for outcome measurement after knee arthroplasty. Sensors (Basel). 2014;14(9):15953–64. https://doi.org/10.3390/s140915953.
Amemiya K, Kaneko T, Omata M, Igarashi T, Takada K, Ikegami H, et al. Anatomical bi-cruciate retaining TKA improves gait ability earlier than bi-cruciate stabilized TKA based on triaxial accelerometery data: a prospective cohort study. Asia Pac J Sports Med Arthrosc Rehabil Technol. 2021;25:35–41. https://doi.org/10.1016/j.asmart.2021.05.004.
Tomite T, Saito H, Kijima H, Ishikawa N, Hatakeyama Y, Tazawa H, et al. Evaluation of anteroposterior accelerometric change after bi-cruciate stabilized total knee arthroplasty and posterior stabilized total knee arthroplasty. Knee. 2021;32:121–30. https://doi.org/10.1016/j.knee.2021.08.012.
Jolles BM, Grzesiak A, Eudier A, Dejnabadi H, Voracek C, Pichonnaz C, et al. A randomised controlled clinical trial and gait analysis of fixed- and mobile-bearing total knee replacements with a five-year follow-up. J Bone Joint Surg Br. 2012;94(5):648–55. https://doi.org/10.1302/0301-620X.94B5.27598.
Tsuji S, Tomita T, Fujii M, Laskin RS, Yoshikawa H, Sugamoto K. Is minimally invasive surgery-total knee arthroplasty truly less invasive than standard total knee arthroplasty? A quantitative evaluation. J Arthroplasty. 2010;25(6):970–6. https://doi.org/10.1016/j.arth.2009.06.016.
Fransen BL, Mathijssen NMC, Slot K, de Esch NHH, Verburg H, Temmerman OPP, et al. Gait quality assessed by trunk accelerometry after total knee arthroplasty and its association with patient related outcome measures. Clin Biomech (Bristol, Avon). 2019;70:192–6. https://doi.org/10.1016/j.clinbiomech.2019.10.007.
Lee SY, Kim BR, Kim SR, Han EY, Nam KW, Park Y-G. Influence of preoperative physical function on gait 1 month after total knee arthroplasty. Ann Geriatr Med Res. 2017;21(4):188–96. https://doi.org/10.4235/agmr.2017.21.4.188.
Boekesteijn R, Smolders J, Busch V, Keijsers N, Geurts A, Smulders K. Objective monitoring of functional recovery after total knee and hip arthroplasty using sensor-derived gait measures. PeerJ. 2022;10:e14054. https://doi.org/10.7717/peerj.14054
Emmerzaal J, Corten K, van der Straaten R, De Baets L, Van Rossom S, Timmermans A, et al. Movement quality parameters during gait assessed by a single accelerometer in subjects with osteoarthritis and following total joint arthroplasty. Sensors (Basel). 2022;22(8):2955. https://doi.org/10.3390/s22082955.
Rahman J, Tang Q, Monda M, Miles J, McCarthy I. Gait assessment as a functional outcome measure in total knee arthroplasty: a cross-sectional study. BMC Musculoskelet Disord. 2015;16:66. https://doi.org/10.1186/s12891-015-0525-2.
Fransen BL, Pijnappels M, Butter IK, Burger BJ, van Dieen JH, Hoozemans MJM. Patients’ perceived walking abilities, daily-life gait behavior and gait quality before and 3 months after total knee arthroplasty. Arch Orthop Trauma Surg. 2022;142(6):1189–96. https://doi.org/10.1007/s00402-021-03915-y.
Chapman RM, Moschetti WE, Van Citters DW. Is clinically measured knee range of motion after total knee arthroplasty ‘good enough?’: a feasibility study using wearable inertial measurement units to compare knee range of motion captured during physical therapy versus at home. Med Novel Technol Devices. 2021;11:100085. https://doi.org/10.1016/j.medntd.2021.100085.
Gianzina E, Kalinterakis G, Delis S, Vlastos I, Platon Sachinis N, Yiannakopoulos CK. Evaluation of gait recovery after total knee arthroplasty using wearable inertial sensors: a systematic review. Knee. 2023;41:190–203. https://doi.org/10.1016/j.knee.2023.01.006.
Boonstra MC, Schwering PJA, Malefijt M, Verdonschot N. Sit-to-stand movement as a performance-based measure for patients with total knee arthroplasty. Phys Ther. 2010;90(2):149–56. https://doi.org/10.2522/ptj.20090119.
Benson LC, Clermont CA, Bosnjak E, Ferber R. The use of wearable devices for walking and running gait analysis outside of the lab: a systematic review. Gait Posture. 2018;63:124–38. https://doi.org/10.1016/j.gaitpost.2018.04.047.
Erhart-Hledik JC, Favre J, Andriacchi TP. New insight in the relationship between regional patterns of knee cartilage thickness, osteoarthritis disease severity, and gait mechanics. J Biomech. 2015;48(14):3868–75. https://doi.org/10.1016/j.jbiomech.2015.09.033.
Mills K, Hunt MA, Ferber R. Biomechanical deviations during level walking associated with knee osteoarthritis: a systematic review and meta-analysis. Arthritis Care Res. 2013;65(10):1643–65. https://doi.org/10.1002/acr.22015.
Boonstra MC, van der Slikke RMA, Keijsers NLW, van Lummel RC, Malefijt MCD, Verdonschot N. The accuracy of measuring the kinematics of rising from a chair with accelerometers and gyroscopes. J Biomech. 2006;39(2):354–8. https://doi.org/10.1016/j.jbiomech.2004.11.021.
Faber GS, Kingma I, Bruijn SM, van Dieen JH. Optimal inertial sensor location for ambulatory measurement of trunk inclination. J Biomech. 2009;42(14):2406–9. https://doi.org/10.1016/j.jbiomech.2009.06.024.
Ishigaki N, Kimura T, Usui Y, Aoki K, Narita N, Shimizu M, et al. Analysis of pelvic movement in the elderly during walking using a posture monitoring system equipped with a triaxial accelerometer and a gyroscope. J Biomech. 2011;44(9):1788–92. https://doi.org/10.1016/j.jbiomech.2011.04.016.
Reininga IHF, Stevens M, Wagenmakers R, Boerboom AL, Groothoff JW, Bulstra SK, et al. Compensatory trunk movements in patients with hip osteoarthritis accuracy and reproducibility of a body-fixed sensor-based assessment. Am J Phys Med Rehabil. 2011;90(8):681–7. https://doi.org/10.1097/PHM.0b013e31820f955e.
Item-Glatthorn JF, Casartelli NC, Petrich-Munzinger J, Munzinger UK, Maffiuletti NA. Validity of the intelligent device for energy expenditure and activity accelerometry system for quantitative gait analysis in patients with hip osteoarthritis. Arch Phys Med Rehabil. 2012;93(11):2090–3. https://doi.org/10.1016/j.apmr.2012.06.018.
Hafer JF, Provenzano SG, Kern KL, Agresta CE, Grant JA, Zernicke RF. Measuring markers of aging and knee osteoarthritis gait using inertial measurement units. J Biomechan. 2020;99:109567. https://doi.org/10.1016/j.jbiomech.2019.109567.
Hunt MA, Simic M, Hinman RS, Bennell KL, Wrigley TV. Feasibility of a gait retraining strategy for reducing knee joint loading: Increased trunk lean guided by real-time biofeedback. J Biomech. 2011;44(5):943–7. https://doi.org/10.1016/j.jbiomech.2010.11.027.
Hatfield GL, Hubley-Kozey CL, Wilson JLA, Dunbar MJ. The effect of total knee arthroplasty on knee joint kinematics and kinetics during gait. J Arthroplasty. 2011;26(2):309–18. https://doi.org/10.1016/j.arth.2010.03.021.
Youn IH, Youn JH, Zeni JA, Knarr BA. Biomechanical gait variable estimation using wearable sensors after unilateral total knee arthroplasty. Sensors (Basel). 2018;18(5):1577. https://doi.org/10.3390/s18051577.
Konrath JM, Karatsidis A, Schepers HM, Bellusci G, de Zee M, Andersen MS. Estimation of the knee adduction moment and joint contact force during daily living activities using inertial motion capture. Sensors. 2019;19(7):1681. https://doi.org/10.3390/s19071681.
He ZX, Liu T, Yi JG. A wearable sensing and training system: towards gait rehabilitation for elderly patients with knee osteoarthritis. IEEE Sens J. 2019;19(14):5936–45. https://doi.org/10.1109/JSEN.2019.2908417.
Schotanus MGM, Bemelmans YFL, Grimm B, Heyligers IC, Kort NP. Physical activity after outpatient surgery and enhanced recovery for total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2017;25(11):3366–71. https://doi.org/10.1007/s00167-016-4256-1.
van Lummel RC, Walgaard S, Pijnappels M, Elders PJM, Garcia-Aymerich J, van Dieen JH, et al. Physical performance and physical activity in older adults: associated but separate domains of physical function in old age. PLoS One. 2015;10(12):e0144048. https://doi.org/10.1371/journal.pone.0144048.
Ramkumar PN, Haeberle HS, Ramanathan D, Cantrell WA, Navarro SM, Mont MA, et al. Remote patient monitoring using mobile health for total knee arthroplasty: validation of a wearable and machine learning-based surveillance platform. J Arthroplasty. 2019;34(10):2253–9. https://doi.org/10.1016/j.arth.2019.05.021.
Acknowledgements
None.
Funding
No funding has been received for this study.
Author information
Authors and Affiliations
Contributions
Y.G.F.: Study design, literature review, data analysis and manuscript writing. Y.L. and F.D.: Literature review and data analysis. Y.X.: Study design and supervisor. J.C., Y.F. and J.L: Interpretation and manuscript editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
All authors have no competing interests for this study to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Complete search strategy
Search strategy individually optimized for each database based on the three broad topics of total knee arthroplasty, wearable electronic devices, and gait analysis joined using the "AND" search command/function.
PubMed
Total knee arthroplasty: ("Arthroplasty, Replacement, Knee"[Majr] OR Arthroplasties, Knee Replacement[Title/Abstract] OR Arthroplasties, Replacement, Knee[Title/Abstract] OR Arthroplasty, Knee Replacement[Title/Abstract] OR Arthroplasty, Replacement, Partial Knee[Title/Abstract] OR Knee Arthroplasty[Title/Abstract] OR Knee Arthroplasty, Total[Title/Abstract] OR Knee Replacement Arthroplasties[Title/Abstract] OR Knee Replacement Arthroplasty[Title/Abstract] OR Knee Replacement, Total[Title/Abstract] OR Partial Knee Arthroplasty[Title/Abstract] OR Partial Knee Replacement[Title/Abstract] OR Replacement Arthroplasties, Knee[Title/Abstract] OR Replacement Arthroplasty, Knee[Title/Abstract] OR Replacement, Total Knee[Title/Abstract] OR Total Knee Replacement[Title/Abstract] OR Unicompartmental Knee Arthroplasty[Title/Abstract] OR Unicompartmental Knee Replacement[Title/Abstract] OR Unicondylar Knee Arthroplasty[Title/Abstract] OR Unicondylar Knee Replacement[Title/Abstract]).
Wearable electronic devices: ("Wearable Electronic Devices"[Majr] OR Electronic Skin[Title/Abstract] OR Wearable Devices[Title/Abstract] OR Wearable Technology[Title/Abstract]) OR wearable sensor*[Title/Abstract] OR wearable technology[Title/Abstract] OR motion sensor*[Title/Abstract] OR inertial sensor*[Title/Abstract] OR inertial motion capture[Title/Abstract] OR inertial measurement unit*[Title/Abstract] OR body sensor network*[Title/Abstract] OR body worn sensor*[Title/Abstract] OR sensor fusion[Title/Abstract] OR IMU[Title/Abstract] OR MEMS*[Title/Abstract] OR acceleromet*[Title/Abstract] OR gyroscop*[Title/Abstract] OR magnetomet*[Title/Abstract] OR Device, Wearable Electronic[Title/Abstract] OR Electronic Device, Wearable[Title/Abstract] OR Wearable Electronic Device[Title/Abstract] OR Technology, Wearable[Title/Abstract] OR Wearable Devices[Title/Abstract] OR Device, Wearable[Title/Abstract] OR Electronic Skin[Title/Abstract] OR Skin, Electronic[Title/Abstract]).
Gait analysis: ("Gait Analysis" [Majr] OR Analysis, Gait [Title/Abstract] OR Gait Analyses [Title/Abstract] OR biomechanic*[Title/Abstract] OR walk*[Title/Abstract] OR spatiotemporal [Title/Abstract] OR kinematic*[Title/Abstract] OR acceleration*[Title/Abstract]).
Embase
Total knee arthroplasty: ('total knee arthroplasty'/exp OR 'arthroplasty, replacement, knee' OR 'arthroplasties, knee replacement':ab,ti OR 'arthroplasties, replacement, knee':ab,ti OR 'arthroplasty, knee replacement':ab,ti OR 'arthroplasty, replacement, partial knee':ab,ti OR 'knee arthroplasty':ab,ti OR 'knee arthroplasty, total':ab,ti OR 'knee replacement arthroplasties':ab,ti OR 'knee replacement arthroplasty':ab,ti OR 'knee replacement, total':ab,ti OR 'partial knee arthroplasty':ab,ti OR 'partial knee replacement':ab,ti OR 'replacement arthroplasties, knee':ab,ti OR 'replacement arthroplasty, knee':ab,ti OR 'replacement, total knee':ab,ti OR 'total knee replacement':ab,ti OR 'unicompartmental knee arthroplasty':ab,ti OR 'unicompartmental knee replacement':ab,ti OR 'unicondylar knee arthroplasty':ab,ti OR 'unicondylar knee replacement':ab,ti).
Wearable electronic devices: ('wearable computer'/exp OR 'Wearable Electronic Devices' OR 'Electronic Skin':ab,ti OR 'Wearable Devices':ab,ti OR 'Wearable Technology':ab,ti OR 'wearable sensor*':ab,ti OR 'wearable technology':ab,ti OR 'motion sensor*':ab,ti OR 'inertial sensor*':ab,ti OR 'inertial motion capture':ab,ti OR 'inertial measurement unit*':ab,ti OR 'body sensor network*':ab,ti OR 'body worn sensor*':ab,ti OR 'sensor fusion':ab,ti OR 'IMU':ab,ti OR 'MEMS*':ab,ti OR 'acceleromet*':ab,ti OR 'gyroscop*':ab,ti OR 'magnetomet*':ab,ti OR 'Device, Wearable Electronic':ab,ti OR 'Electronic Device, Wearable':ab,ti OR 'Wearable Electronic Device':ab,ti OR 'Technology, Wearable':ab,ti OR 'Wearable Devices':ab,ti OR 'Device, Wearable':ab,ti OR 'Electronic Skin':ab,ti OR 'Skin, Electronic':ab,ti).
Gait analysis: ('gait’/exp OR 'Gait Analysis' OR 'Analysis, Gait':ab,ti OR 'Gait Analyses':ab,ti OR 'biomechanic*':ab,ti OR 'walk*':ab,ti OR 'spatiotemporal':ab,ti OR 'kinematic*':ab,ti OR 'acceleration*':ab,ti).
Cochrane
Total knee arthroplasty: ((Arthroplasties, Knee Replacement):ti,ab,kw OR (Arthroplasties, Replacement, Knee):ti,ab,kw OR (Arthroplasty, Knee Replacement):ti,ab,kw OR (Arthroplasty, Replacement, Partial Knee):ti,ab,kw OR (Knee Arthroplasty):ti,ab,kw OR (Knee Arthroplasty, Total):ti,ab,kw OR (Knee Replacement Arthroplasties):ti,ab,kw OR (Knee Replacement Arthroplasty):ti,ab,kw OR (Knee Replacement, Total):ti,ab,kw OR (Partial Knee Arthroplasty):ti,ab,kw OR (Partial Knee Replacement):ti,ab,kw OR (Replacement Arthroplasties, Knee):ti,ab,kw OR (Replacement Arthroplasty, Knee):ti,ab,kw OR (Replacement, Total Knee):ti,ab,kw OR (Total Knee Replacement):ti,ab,kw OR (Unicompartmental Knee Arthroplasty):ti,ab,kw OR (Unicompartmental Knee Replacement):ti,ab,kw OR (Unicondylar Knee Arthroplasty):ti,ab,kw OR (Unicondylar Knee Replacement):ti,ab,kw)).
Wearable electronic devices: ((Electronic Skin):ti,ab,kw OR (Wearable Devices):ti,ab,kw OR (Wearable Technology):ti,ab,kw OR (wearable sensor*):ti,ab,kw OR (wearable technology):ti,ab,kw OR (motion sensor*):ti,ab,kw OR (inertial sensor*):ti,ab,kw OR (inertial motion capture):ti,ab,kw OR (inertial measurement unit*):ti,ab,kw OR (body sensor network*):ti,ab,kw OR (body worn sensor*):ti,ab,kw OR (sensor fusion):ti,ab,kw OR (IMU):ti,ab,kw OR (MEMS*):ti,ab,kw OR (acceleromet*):ti,ab,kw OR (gyroscop*):ti,ab,kw OR (magnetomet*):ti,ab,kw OR (Device, Wearable Electronic):ti,ab,kw OR (Electronic Device, Wearable):ti,ab,kw OR (Wearable Electronic Device):ti,ab,kw OR (Technology, Wearable):ti,ab,kw OR (Wearable Devices):ti,ab,kw OR (Device, Wearable):ti,ab,kw OR (Electronic Skin):ti,ab,kw OR (Skin, Electronic):ti,ab,kw)).
Gait analysis: ((Analysis, Gait): ti,ab,kw OR (Gait Analyses):ti,ab,kw OR (biomechanic*):ti,ab,kw OR (walk*):ti,ab,kw OR (spatiotemporal):ti,ab,kw OR (kinematic*):ti,ab,kw OR (acceleration*):ti,ab,kw)).
Web of Science Core Collection/Medline
Total knee arthroplasty: TS = (total knee arthroplasty OR arthroplasty, replacement, knee OR arthroplasties, knee replacement OR arthroplasties, replacement, knee OR arthroplasty, knee replacement OR arthroplasty, replacement, partial knee OR knee arthroplasty OR knee arthroplasty, total OR knee replacement arthroplasties OR knee replacement arthroplasty OR knee replacement, total OR partial knee arthroplasty OR partial knee replacement OR replacement arthroplasties, knee OR replacement arthroplasty, knee OR replacement, total knee OR total knee replacement OR unicompartmental knee arthroplasty OR unicompartmental knee replacement OR unicondylar knee arthroplasty OR unicondylar knee replacement).
Wearable electronic devices: TS = (Wearable Electronic Devices OR wearable sensor* OR wearable technology OR motion sensor* OR inertial sensor* OR inertial motion capture OR inertial measurement unit* OR body sensor network* OR body worn sensor* OR sensor fusion OR IMU OR MEMS* OR acceleromet* OR gyroscop* OR magnetomet*).
Gait analysis: TS = (gait OR analysis, gait OR gait Analyses or biomechanic* OR walk* OR spatiotemporal OR kinematic OR acceleration).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Feng, Y., Liu, Y., Fang, Y. et al. Advances in the application of wearable sensors for gait analysis after total knee arthroplasty: a systematic review. Arthroplasty 5, 49 (2023). https://doi.org/10.1186/s42836-023-00204-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s42836-023-00204-4