Abstract
Background
Climate change has caused several alterations in the frequency, intensity, and severity of wildfires globally, particularly in the western United States. Wildfire can dramatically change the microclimate experienced by animals who inhabit fire-prone areas, with implications for energy expenditure, particularly for heterothermic species. Heterothermic mammals manage energy expenditure by selecting optimal microclimates and regulating their body temperature. Because bats frequently use torpor, they are a useful model organism to understand how wildfires affect heterothermic mammal communities. In 2020, Sequoia and Kings Canyon National Parks were subjected to a severe wildfire that spanned 3636 ha within the boundaries of the park. We tracked eight California myotis (Myotis californicus) captured in Sequoia National Park to 22 roost sites in a gradient of burn severities in the summer from June–August 2021. We also quantified the torpor expression of bats on this post-burn landscape by measuring the roosting skin temperature of four M. californicus captured in a fire-affected site using temperature-sensitive radio telemetry.
Results
M. californicus showed preference for taller and larger diameter trees with more exfoliating bark, although they did not show preference for whether individual trees were burned or unburned. At the roost habitat-scale, 21 out of 22 M. californicus roosts were within or < 5 m from the burn mosaic, despite availability of unburned landscape. M. californicus most frequently roosted in low-severity burned areas, as opposed to unburned or severely burned areas. Additionally, bats avoided areas without a canopy and used areas with taller trees in proportion to their availability. Myotis californicus used torpor during the coolest periods of the morning before sunrise and regularly used torpor in low-severity burn area roosts.
Conclusions
This research indicates that while fire may be beneficial to some bat species, those that are clutter-tolerant could be negatively affected by severe wildfires that cause major reductions in vegetative complexity. Bats in our study appear to use both habitat selection and torpor to manage their energy and water budgets. Protecting large diameter trees with exfoliating bark, like those used by bats in our study, could be key to promoting the persistence of M. californicus on this landscape as severe wildfire may reduce these roosts. In addition, restoring historical mixed-severity fire regimes, thereby creating a fire mosaic containing low severity burned areas, may be critical for maintaining both roosting and foraging habitat suitable for our study species in the fire-adapted coniferous forests of North America’s Sierra Nevada.
Resumen
Antecedentes
El cambio climático ha causado diferentes alteraciones en la frecuencia, intensidad, y severidad de los incendios a nivel global, particularmente en el oeste de los EEUU. Los fuegos de vegetación pueden alterar dramáticamente el microclima que experimentan los animales que habitan en áreas proclives al fuego, con implicancias en el gasto de energía, en particular por especies hetero-térmicas. Los mamíferos hetero-térmicos manejan su gasto de energía mediante la selección de microclimas óptimos y la regulación de su temperatura corporal. Dado que los murciélagos usan frecuentemente la quiescencia o inactividad para ese propósito, representan un organismo modelo para comprender cómo los fuegos afectan a las comunidades de mamíferos hetero-térmicos. En 2020, los parques nacionales Sequoia y King Canyon fueron objeto de fuegos severos que cubrieron 3.636 ha dentro de los límites de esos parques. Registramos el movimiento de ocho murciélagos-ratones de California (Myotis californicus) capturados en el Parque Nacional Sequoia en 22 lugares de percheo, en un gradiente de severidad de fuegos en el verano de junio a agosto de 2021. También cuantificamos la expresión de inactividad los murciélagos en este paisaje post fuego mediante la medición de la temperatura de la piel de cuatro murciélagos-ratones en los lugares donde permanecen colgados durante su inactividad (percheo) en un sitio afectado por fuegos usando radio telemetría sensible a la temperatura.
Resultados
Los murciélagos-ratones de California mostraron preferencia por árboles altos y de gran diámetro y con corteza muy exfoliada, aunque no mostraron preferencias por árboles individuales que estuviesen o no quemados. A escala de percheo, 21 de 22 murciélagos-ratones de California estaban a 5 o menos metros de altura sobre el mosaico del suelo quemado, a pesar de disponer de áreas del paisaje no quemadas. Este murciélago se colgaba para su inactividad en áreas quemadas con baja severidad, en oposición a áreas severamente o directamente no quemadas. Adicionalmente, los murciélagos evitaban áreas sin doseles superiores, y usaban áreas con árboles altos en proporción su disponibilidad. El murciélago-ratón de California usaba el período de inactividad durante los períodos más fríos antes de la salida del sol y regularmente en áreas de percheo ubicadas en sitios postfuego de baja severidad.
Conclusiones
Esta investigación indica que, aunque el fuego puede ser beneficioso para algunas especies de murciélagos, aquellas que son tolerantes a cierto grado de desorden pueden ser negativamente afectadas por fuegos severos que causan una reducción en la complejidad de la vegetación. Los murciélagos en nuestro estudio parecen usar tanto la selección de hábitat y la inactividad en el percheo para manejar sus balances de agua y energía. La protección de árboles de gran diámetro con corteza exfoliada, como los usados por los murciélagos en nuestro estudio, puede ser la clave para promover la persistencia de M. californicus en este paisaje en el que incendios severos pueden reducir estos lugares de percheo. Adicionalmente, la restauración de regímenes de fuegos de severidad mixta, creando de esa manera mosaicos de fuego que contengan áreas de baja severidad, puede ser crítica para mantener tanto los hábitats de percheo como forrajeo para nuestra especie en estudio, en bosques de coníferas adaptadas al fuego en la Sierra Nevada de Norte América.
Similar content being viewed by others
Background
Anthropogenic effects including climate change, historical fire exclusion, and cessation of Indigenous fire regimes have caused changes to the frequency, intensity, and severity of wildfires (Di Virgilio et al. 2019; Williams et al. 2019; Hanan et al. 2021; Pausas and Keeley 2021). In California, large fires (> 10,000 ha) have become more common (Keeley and Syphard 2021), in part due to increased periods of drought (Chen 2022). Wildfire is now a concern in California outside of the “typical” fire season, more frequently extending into cooler autumn months due to increased temperatures coupled with dry vegetation (Goss et al. 2020). Alarmingly, reburn of forests (when new fires burn recently burned areas at a much shorter interval than expected) have become more frequent (Buma et al. 2020), which may have significant effects on the ability of vegetation to regenerate.
The effects of wildfire on wildlife are now a topic of great interest and concern. In the short-term, wildfire poses risks to wildlife through exposure to hazardous pollution, which, among other effects, may cause carbon monoxide poisoning, respiratory disease, and immunosuppression (Sanderfoot et al. 2022). Post-wildfire habitat can be beneficial to some carnivorous or insectivorous species that opportunistically seek food resources on scorched land (e.g., Geary et al. 2020; Doherty et al. 2023) or may trigger the use of torpor in heterothermic species to avoid predation (Geiser et al. 2018). Carnivores, such as the spotted-tail quoll (Dasyurus maculatus), may shift dietary preference based on post-fire prey availability (Dawson et al. 2007), while herbivores, such as the white-tailed deer (Odocoileus virginianus) and snowshoe hare (Lepus americanus), may temporarily shift habitat preference to unburned habitat until burned land regenerates sufficiently for feeding (Cave et al. 2021).
For highly mobile animals such as bats, the effects of wildfire are complex and often species-dependent (Loeb and Blakey 2021). While bats may be able to fly away from fire, their escape response is dependent on the timing of fire; for example, fires that occur in cooler weather may reduce the ability of a bat to respond to smoke cues while in torpor (Doty et al. 2018; Layne et al. 2021). The height of the roost at the time of fire also influences bat behavior, in that bats roosting lower on a tree are more likely to evacuate (Jorge et al. 2021) or experience injury (Dickinson et al. 2010). Following fire, bats may capitalize on insects attracted to open habitat created by wildfire, even shortening torpor bouts to do so (Doty et al. 2016). The short-term effects of fires on bats may be beneficial in that they may create suitable roosts (Johnson et al. 2010; Ford et al. 2016) or clear vegetation that normally precludes access for less-maneuverable bats with high wing-loading. Bats adapted to open habitats are more active in areas with more frequent and severe fires (Armitage and Ober 2012; Blakey et al. 2019). A reduction in tree density may also be beneficial for bats in winter that seek increased sun exposure (Boyles and Aubrey 2006), although fire may collapse tree cavities, placing species that rely on these trees at risk (Lindenmayer et al. 2012). In some cases, increased heterogeneity caused by fire is linked to increased occupancy and bat richness, indicating the resiliency of some bats to fire-affected landscapes (Steel et al. 2019, Blakey et al. 2021), although bats may be sensitive to extreme wildfires that largely reduce preferred habitat (Law et al. 2022).
Acoustic research indicates that clutter-adapted bats may be more susceptible to severe fire when foraging (Blakey et al. 2019). Though research on roosting ecology in landscapes affected by wildfire is scarce, there is evidence that clutter-adapted bats may both avoid or be unaffected by wildfire-affected areas (Snider et al. 2013; Law et al. 2018), whereas areas managed with low severity prescribed fire provide suitable roosting habitat for an Australian clutter-adapted species (Gonsalves et al. 2022), while having minimal effect on bat foraging (Law et al. 2018). As prescribed fires generally burn at lower severity than wildfires, burn severity, rather than the type of burn (wildfire or prescribed fire) may be the key factor determining suitability of burned landscapes as roosting habitat for clutter-adapted bats (Loeb and Blakey 2021).
Bats are uniquely adapted among mammals to both escape fire (using flight) and to cope with resource scarcity after fire through the use of torpor. Most research on torpor in bats has been biased towards winter months when bats hibernate, when there is a decrease in temperature, food, and water availability (Ruf and Geiser 2015). However, more recent research has highlighted the bat use of torpor, even in summer during heatwaves (Bondarenco et al. 2014), or when conditions are mild (Levin et al. 2012). Changes in resource availability, foraging habitat, and roosting opportunities due to wildfire could trigger bats to use torpor even during summer, when possible, to save energy or reduce water loss. This is particularly true for bats that typically roost in thermally buffered sites (e.g., cavities and crevices) such as clutter-adapted bats, who are less capable of tolerating high heat compared to bats that roost in more open areas (e.g., foliage; Czenze et al. 2020). In burned landscapes, roosts with greater sun exposure may be beneficial in winter to facilitate passive rewarming, yet detrimental to bats during summer, particularly in hot areas, such as the Sierra Nevada foothills of California. Thus, understanding how clutter-tolerant bats cope with tree- and habitat-scale changes due to wildfire both in terms of their physiology and ecology is important in both making informed fire management decisions and predicting bat community response to ongoing climate change.
In August 2020, lightning-caused wildfires known as the Castle and Shotgun fires occurred in the Sierra Nevada range in Central California. The fires, later deemed the SQF complex, spanned 69,337 ha and encompassed regions of Sequoia National Forest, the Giant Sequoia National Monument, Inyo National forest, land managed by the Bureau of Land Management, state land, private land, and Sequoia National Park (3636 ha). This event provided the opportunity to study the post-fire roosting behavior of a common clutter-tolerant bat species, the California myotis (Myotis californicus). Reproductive females of this species roost in snags, under bark, or in cavities of coniferous trees in varying stages of decay in British Columbia (Brigham et al. 1997; Barclay and Brigham 2001). However, there is little to no understanding of their response to fire, with the exception of acoustic research (e.g., Blakey et al. 2019, Steel et al. 2019).
The aim of our project was to (1) determine roost tree and roost habitat selection of M. californicus in a wildfire-affected landscape that provides areas of varying fires severity and (2) understand the torpor expression of bats in a post-wildfire landscape. We hypothesized that both tree and habitat selection would be affected by fire severity, and that torpor expression would also be affected by these choices. We predicted that (1) M. californicus, a clutter-tolerant species, would select roosts in unburned or low-severity burn habitat, (2) that M. californicus would avoid areas with low canopy cover and would also avoid severely burned trees due to the increased daytime thermal fluctuations in an area that regularly experiences extremely hot temperatures, and (3) that M. californicus would use torpor, when possible, and more frequently in larger, unburned trees to reduce daytime energy expenditure and water loss in response to increased summer temperatures.
Methods
Study area
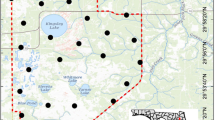
Our study took place in Sequoia and Kings Canyon National Parks, California, USA (Fig. 1), from June to August 2021. We also present physiological data from two pallid bats (A. pallidus) captured at the Potwisha campground, near the foothill visitor center entrance of the park (see Supplemental Material). Capture locations were situated in the foothill-woodland vegetation zone of the Sierra Nevada mountain range, characterized by blue oak (Quercus douglasii), interior live oak (Q. chrysolepis), California black oak (Q. kelloggii), and California buckeye (Aesculus californica), with understory plants including Dogwood (Cornus spp.) and Redbud (C. occidentalis). Elevation in our study area ranged from 1086 to 1658 m. Mean temperature in the foothill area during summer ranges from mean low of 16 °C to a mean high of 36 °C (data provided by the National Park Service). Precipitation is rare and is generally restricted to short afternoon summer showers. Fire severity surrounding the South Fork campground was classified from 1 (not burned) to 4 (severely burned) according to the Rapid Assessment of Vegetation Condition after Wildfire (RAVG) portal (RAVG; https://burnseverity.cr.usgs.gov/ravg/; See burn variables and environmental covariates subsection for more information).
The study area within the Sierra Nevada Mountain range (green shading, right inset) of California (black line, right inset). The capture location (larger circle) and roost locations (smaller circles, n = 22) are shown within Sequoia National Park. The area burned by the Castle Fire (August 19, 2020) is outlined by a black dashed line and shaded lighter. The left inset shows a zoomed in version of the area near the roost (marked with a black rectangle on the main map). The aerial imagery was collected post-fire (May, 2022) by Airborne Snow Observatories, Inc
Bat capture and radiotracking
Bats were captured with mist nets (Avinet, Portland, ME, USA) and harp traps (Faunatech, Rydalmere, NSW, AU) placed at dusk across paths, forest edges, or across the South Fork of the Kaweah River in a central location at the South Fork Campground. All fire severities were present in the proximity of the South Fork capture location, and thus, M. californicus bats had opportunity to choose preferred roosting habitat over a range of vegetation complexities.
All bats captured were assessed for body mass, forearm length, sex, reproductive status, age, wing damage, and parasite load. Targeted bats were fitted with temperature-sensitive radio transmitters, with individual transmitter frequencies (0.32 g, LB-2XT, Holohil Systems Inc., Carp, ON, Canada) to assess skin temperature (Tsk) and torpor patterns over a period of up to 2 weeks. Due to the difficulty of implanting internal transmitters and to increase transmitter range, external transmitters were used. Tsk of small heterothermic mammals is usually < 2.0 °C cooler than core body temperature (Barclay et al. 1996) and thus a reliable measurement of torpor patterns in species with large body temperature fluctuations like bats. Transmitters were glued using a latex adhesive (Osto-Bond, Montreal Ostomy, Canada) to the mid-dorsal skin region after removing a patch of fur. Prior to fitting, the temperature-sensitive transmitters were calibrated in a water bath over a range of temperatures from 0 to 40 °C in ~ 7 °C increments.
The morning following release, targeted bats were radio-tracked to their roost location every day using a 3- or 5-element Yagi antennae (Part numbers 13860 or 13,864, Advanced Telemetry Systems, Isanti, MN) connected to a portable receiver (Model R410, Advanced Telemetry Systems, Isanti, MN). Following successful roost location, characteristics of the roost, random trees, and habitat were recorded (see burn variables and environmental covariates for more details). The locations of the bats were recorded with a GPS unit (GPSMap 66; Garmin, Olathe, KS).
Tsk of bats was recorded continually in 10-min intervals using a datalogger (manufactured by Gerhard Körtner, University of New England, Armidale, AU) connected to either a 3-element or 5-element Yagi antennae set within strong reception of the bat transmitter to collect information on torpor and activity patterns. The position of the bats and reception of transmitter signal by the receivers/loggers were assessed every day when possible, and, if necessary, the receivers/loggers were moved to ensure data collection. Data were loaded onto a laptop computer approximately every 3–5 days. Ambient temperature (Ta) was measured with temperature data loggers (± 0.5 °C, iButton thermochron DS1921G, Maxim Integrated Products, Inc., Sunnyvale, CA, USA) in the shade 1 m above the ground, placed in the vicinity of the roosts of M. californicus.
Analytical approach
We used a resource selection approach to investigate whether bats were selecting roosts based on burn characteristics at the roost tree and roost habitat-scales. At the roost tree scale, we used Design III (Manly et al. 2007; Thomas and Taylor 1990), where individuals are identified and availability is measured for each individual, however, in this case, “individual” refers to individual roost locations (n = 19) rather than individual bats (n = 8). For each individual roost, the closest tree ≥ 5 cm DBH in each of the four cardinal directions (North, East, South, and West) were identified as “available trees”; these ranged from 1 to 20 m away from the roost tree (mean = 5.5 m ± 0.4 SE). It is possible that bats that we were not tracking were using the other “available” trees; however, this is unlikely to affect our results given the high (4:1) available-used ratio. At the roost habitat-scale, we used Design II, where individual animals (in this case, individual bats) are identified, but availability is measured at the population scale (Manly et al. 2007; Thomas and Taylor 1990), which we defined as a circular buffer around the capture location, with a radius equal to the farthest roost from the capture location (2512 m).
Burn variables and environmental covariates
We collected burn variables and environmental covariates for the roost-tree scale (among trees) analysis and the roost habitat-scale (landscape) analysis. Roost tree-scale variables included variables that were measured for individual roost trees (Table 1), whereas roost habitat-scale variables varied spatially across the landscape, measured at a coarser grain than an individual tree (Table 2). We collected 13 tree structure and type variables (Table 1) and two burn variables for the roost tree-scale analysis. We used 10 of the 13 tree structure and type variables in the analysis and used the other three to provide contextual information.
We identified each roost tree to species, allowing us to also categorize leaf type (deciduous, evergreen) and tree type (conifer, hardwood). Roost type was visually determined as one of four categorical variables: exfoliating bark, crevice, cavity, or unknown, and crown class was determined as one of four categories: dominant, co-dominant, intermediate, suppressed. We measured diameter at breast height (DBH) using a fabric diameter tape measure (Model 283D/20F, Forestry Suppliers, Jackson, MS) and estimated tree height using a clinometer (PM-5/360, Suunto, Vantaa, Finland). We visually estimated sun exposure by determining, during peak daylight hours, whether the roost tree was fully exposed, partially exposed or not exposed to sunlight at the crown. We also estimated the percent of remaining and exfoliating bark on trees. Percent canopy cover was estimated using a densiometer (Concave Model C, Forestry Suppliers, Jackson, MS), directly underneath the roost tree. Snags were counted as all dead tress > 5 cm DBH within a circular plot with a radius of 17.8 m around the roost tree. We estimated tree burn severity visually, as a categorical variable with five levels: unburned (alive or dead), minor bark burned (base only), major bark burned (up to midpoint of trunk), hardwood burned (tree has burned through the bark into the wood, but was not dead), or burned and dead. We also grouped burn severity variables into burned/unburned categories. Due to access issues, we were only able to estimate these variables for 19 of the 22 roost tree sets, though these included roosts for all 8 individuals.
For the roost habitat-scale analysis, we collected two forest structure variables (tree height and canopy cover) and two burn variables (burn severity and distance to unburned area). Tree height, canopy cover, and elevation were based on a raster product (3 m resolution) provided by Airborne Snow Observatories Inc. (Painter et al. 2016), taken from Lidar flown on May 17, 2022, after the Castle Fire (2020); the horizontal accuracy of this product is < 1.5 m (Ferraz et al. 2018). We estimated maximum tree height (hereafter: tree height) for each roost and random location by extracting the maximum tree height around each location with a circular buffer of 30 m. We used 30 m because this is the coarsest resolution of raster products in our study. We binned tree heights into four categories based on the structure of the forest including shrub (< 5 m), small (5–10 m), medium (10–20 m), and tall (> 20 m). We estimated canopy cover from the tree height raster by coding all pixels with a tree height ≥ 10 m as 1, and all pixels with tree height < 10 m as 0. Similarly, we estimated the proportion of canopy cover for each roost and random location by extracting the mean within 30 m buffers around each location. We binned canopy cover into three categories based on the structure of the forest including: no canopy (0), medium (< 25%), and high (> 25%). We also extracted elevation for each roost location, using mean elevation around roosts with a circular buffer of 30 m, for contextual information.
We collected variables for the roost habitat-scale analysis for all 22 roosts. We measured burn severity using the composite burn index raster from the RAVG portal for the Castle Fire which burned August 19, 2020 (RAVG; https://burnseverity.cr.usgs.gov/ravg/; Key et al. 2006). The composite burn index raster (30 m resolution) coded burn severity as five categories increasing in severity, ranging from 0 (unburned) to 4 (high severity burn). Based on field observations, we re-coded these into three categories: 0–1 (unburned = 1), 2 (low severity = 2), and 3–4 (high severity = 3). We extracted burn severity categories for each roost and random location using a buffer of 30 m, in case locations fell across two pixels. We then used these rasters to make a binary raster with burned areas (burn severity categories 2 and 3) coded as 1 and unburned areas coded as 0. We then created a raster of the distance to unburned areas with 3 m pixels, using the raster v3.5–29 and rgeos v0.5–9 packages in R (R Development Core Team 2022). We extracted the distance to unburned areas for the 21 roosts within the fire perimeter (shaded white, Fig. 2) as the 3 m pixel that overlapped each (roost and random) location and then binned the variable into four categories. These included: unburned, < 50 m, 50–100 m, > 100 m (from unburned area).
Relationships between probability of roost tree use and tree-scale variables including A diameter at breast height (cm) and B percentage of trunk covered in exfoliating bark (%). Relationships were fitted using generalized linear mixed effects models and gray bands show confidence intervals estimated around fixed effects only. Diameter at breast height and exfoliating bark were not correlated (R = 0.03)
How do bats select roosts among trees?
We used generalized mixed effects models with the binomial family to estimate probability of roost selection at the tree scale. We expressed roost selection as a Bernoulli distributed response variable, coding selected roost locations as one, and available (the four alternative trees in the four cardinal directions closest to the roost tree) as zero. We first fitted a model that described the base environmental variables that were likely to influence roost selection at the tree-scale and then added the burn variables to the model to see if burn characteristics explained any more of the variability in roost selection, in addition to factors already influencing bat roost choice. We used the 10 base environmental variables that varied among individual trees (see Table 1). Since including all variables would result in an overparameterized model given our small sample size (n = 95) and could also bias coefficient estimation since some variables were correlated (Dormann et al. 2013), we first fit single covariate models including each environmental covariate separately, but with the same model structure as the full model, including roost (0,1) as the response variable and roost ID (the group of roost tree plus four closest available trees in each ordinal direction) as a random effect.
We used an information theoretic approach to rank the models by the Akaike’s information criterion corrected for small sample sizes (AICc) (Burnham and Anderson 2002). We took the top 3 uncorrelated variables, representing the most parsimonious models, as the variables for our base model. We then compared the base model, to the base model plus the burn variables using ΔAICc. If the burn model had a lower AICc than the base model by greater than 2 AICc, we retained this model as the most parsimonious model; if not, we retained the base environmental model. Fitted relationships are presented with 95% confidence intervals, which were estimated around the fixed effects only, using parametric bootstrapping.
How do bats select roosts across the landscape?
We evaluated bat selection of roost habitat by comparing used and available habitat with Manly selectivity measures. To characterize used roost locations, we summed the number of roost locations for each individual bat, within each category of the four roost habitat-scale variables. For example, we counted how many roosts of each individual bat fell within unburned, low severity burned, and high severity burned areas. We characterized available habitat within a (19.8 km2 area) a circle around the capture location, with a radius equal to the farthest roost from the capture location (2512 m). This gave us measures of use for each category of each variable (tree height, canopy cover, burn severity, distance to unburned) that differed for each bat individual and measures of availability of the same variables that were the same across all bat individuals (Design II). For the distance to unburned area, we restricted the available area to the fire perimeter (shaded white, Fig. 1), because the majority of bats roosted within the fire perimeter, so we were interested in whether they selected for burned areas close to the edge of this perimeter. We note that elevation was uncorrelated to distance to unburned area (R = − 0.11) and distance to capture location was also uncorrelated to distance to unburned area (R = − 0.08). We then calculated Manly selectivity measures for each category of each variable and estimated 95% confidence intervals using Bonferroni-corrected standard errors of the selection ratios. All roost habitat-scale analyses were implemented in the package adehabitatHS v0.3.15 (Calenge, 2006). Results were interpreted as indicating selection for a habitat if the ratio and its confidence interval was > 1 and selection against a habitat if the ratio and its confidence interval were < 1.
Torpor expression
To quantify the torpor expression of bats, we extracted a suite of ecologically relevant response variables for each 24-h day including the presence/absence of torpor, number of torpor bouts, time spent torpid (minutes per day), and torpor depth (maximum drop in Tsk below 30 °C). We further calculated the daily Heterothermy Index for each bat (Boyles et al. 2011). The Heterothermy Index results in a single value describing the overall extent of heterothermy expressed by an animal and is cross-taxa comparable. High Heterothermy Index values indicate greater heterothermic expression (e.g., extensive torpor use) and low values indicate defense of a stable high body temperature (Tb). To differentiate between torpid and active bat Tsk, we considered bats to be torpid if their Tsk dropped below 30 °C for at least 30 min, which is within the range of values commonly used to define torpor in bats (e.g., between 28 and 32 °C, Turbill 2006; Geiser et al. 2011; Johnson and Lacki 2014). Additionally, we calculated the Tb of torpor onset for each individual bat according to Willis (2007). The resulting temperature of torpor onset for all bats ranged from 32 to 33 °C. Because there may be a difference of ~ 2 °C between skin and body temperature (Barclay et al. 1996), we lowered the torpor threshold to 30 °C to account for this possibility. From our daily Tsk data, we labeled days as representative and included them in our analysis if the trace encompassed the entire day roosting period or if we were confident that the Tsk trace was representative of the bat’s thermoregulatory behavior for that day with no extensive active or torpid bouts presumed missing. Tsk data were deemed not representative and not used in analysis if they were incomplete traces deemed likely to be missing extensive torpid or active Tsk bouts. Due to a small sample size, we do not formally analyze the Tsk data of bats in relation to fire severity. However, we provide summary statistics describing the thermoregulatory scope of M. californicus. We report mean values as mean ± SD.
Results
We identified 22 roosts for 8 individual M. californicus bats (2 M (nonreproductive) and 6 F (postlactating); mass = 4.5 ± 0.2 g,) tracked over an average of 4.1 ± 3.9 days with a median of 2 roosts per bat; characteristics of roosts and associated habitats are provided in Tables 1 and 2. Roosts consisted of crevices and spaces underneath bark. We did not observe any maternity roosts. We were also able to collect physiological data from 4 individual M. californicus (1 M (nonreproductive), 3 F (postlactating). Out of 21 roosts (environmental characteristics were not recorded for one roost at the site-level), M. californicus roosted in White alder (Alnus rhombifolia) 3 times, California black oak (Quercus kelloggii) 6 times, Canyon live oak (Quercus chyrysolepis) 8 times, Incense cedars (Calocedrus spp.) twice, California bay (Umbellularia californica) once, and White fir (Abies concolor) once. Of the 6 bats for which multiple roost days were available, all occupied multiple roosts, ranging from 2 to 7 separate roosts. One bat (148.588) used a California black oak twice, but roosted different parts of the same tree on consecutive days. This tree diverged at the base at ground level and thus was functionally considered a separate roost. Bats roosted at elevations ranging from 1093 m, directly adjacent to the capture site, to 1594 m, in a California black oak among a giant Sequoia (Sequoiadendron giganteum) grove. Bats roosted on average 792 m away from the capture site, ranging from 78 to 2512 m.
How do bats select roosts among trees?
We found no evidence that bats selected roosts based on whether trees were burned or unburned or based on how severely a tree had burned (Table 3). We did find a positive relationship between probability of roost use and DBH and percentage of exfoliating bark (Fig. 2). For every 10 cm in DBH, bats were 35% more likely to use a tree as a roost (Fig. 2A). Similarly, for every 10% increase in exfoliating bark coverage, bats were 75% more likely to use a tree as a roost; however, confidence was lower for this relationship (Fig. 2B). We did not find any evidence for spatial autocorrelation in our most parsimonious model (Moran’s I: observed < 0.001, expected = − 0.011, SD = 0.024, p = 0.660). DBH and percentage exfoliating bark were not correlated (R = 0.03) and were the top two most parsimonious models when fitting tree-scale variables as separate predictors (Table 4), after removing tree height which was correlated with DBH (R = 0.68) and bark which was correlated with percentage exfoliating bark (R = − 0.63).
How do bats select roosts across the landscape?
We found evidence that bats’ roosting choices were influenced by both burn and vegetation structure variables. Bats were more likely to roost in low severity burned areas, rather than unburned or areas burned with high severity (Fig. 3A). Importantly, 21 of the 22 roosts were located in or within 5 m of the burn mosaic, despite availability of unburned landscape. Within this mosaic, we did not find evidence that bats selected for unburned areas (Fig. 3B). Bats used areas with the tallest trees (maximum tree height > 20 m) in proportion to their availability (Fig. 3C). Bats avoided areas with no canopy, and areas where maximum tree height was below 10 m when roosting, instead selecting for areas with canopy cover above 10% and medium tree heights (10–20 m) (Fig. 3D).
Population level Manly selection ratios (black points) showing positive (> 1) and negative (< 1) selection for categories of A burn severity (dNBR), B distance to unburned area within the fire perimeter (m), C tree height (m), and D canopy cover (%) by roosting M. californicus based on 22 roost locations observed from 8 individuals from 29 June to 14 August 2021. Error bars show Bonferroni-corrected 95% confidence intervals, gray points show selection ratios for each individual bat, with size of the point proportional to the number of roosts collected for that individual (median of 2 (1–7) roosts per bat)
Torpor expression
For the four M. californicus we collected Tsk data from, mean Tsk was 32.1 ± 4.8 °C and ranged from 19.4 to 41.3 °C (mode = 35.7 °C; Fig. 4). From our representative days of data, we found that M. californicus used torpor on 94% of days (15 of 16 days) and that bats typically employed 1–2 torpor bouts per day (mean = 1.8 ± 0.9 bouts per day), which typically occurred in the morning hours (e.g., Fig. 5A, B). Myotis californicus on average spent 310 ± 255 min in torpor each day (range: 0–940 min). Torpor depth averaged 5.9 ± 2.8 °C below the torpor onset threshold of 30 °C and ranged from 0 to 10.6 °C. The mean Heterothermy Index for M. californicus was 5.5 ± 2.0 °C and ranged from 2.9 to 10.9 °C.
Skin temperature (Tsk) traces of two individual (A, B) post-lactating female Myotis californicus exhibiting consistent morning torpor bouts. Colored lines and points indicate the bat’s Tsk and the black line indicates ambient temperature. Different colored Tsk traces delineate separate days. The horizontal green line represents an approximate torpor onset threshold at 30 °C. Gray shaded regions indicate approximate overnight hours (2000–0800 h). A This post-lactating female used two roost trees in low severity burn areas on 29 June through 03 July 2021. The roost tree location was not confirmed for 27 June 2021. B This post lactating female did not have confirmed roost locations due to site inaccessibility
Of the four M. californicus bats tracked with temperature-sensitive telemetry, we were able to confirm eight roost trees for two individuals. Of these eight trees, two were in a high-severity burn area, five were in low-severity burn areas, and one was in an unburned area. From these trees, we have representative Tsk data for four roosts (eight total days of Tsk data), all of which were located in a low severity burn area. The mean Tsk for bats in these low-severity burn area roosts was 32.9 ± 4.9 °C and ranged from 20.5 to 41.3 °C. Bats used torpor on 88% of these days (7 of 8 days; e.g., Fig. 5A). Bats used 0–4 torpor bouts in these roosts (mean = 2.0 ± 1.2 bouts per day). These bats spent 0–940 min in torpor per day (mean = 303 ± 290 min per day), and their torpor depth ranged from 0 to 10.6 °C (mean = 5.5 ± 3.3 °C). The Heterothermy Index for bats roosting in these low-severity burn area trees ranged from 2.9 to 10.9 (mean = 5.5 ± 2.5 °C).
Discussion
Our study demonstrates that wildfires, and specifically burn severity, are an important driver of roost and habitat selection of M. californicus in the South Fork Area of Sequoia National Park. We also recorded the use of torpor by bats during summer in a burned landscape, potentially uncovering one of the mechanisms underpinning bat resilience to fire.
Myotis californicus did not show preference for whether a tree was burned or unburned. However, at the roost habitat scale (within 30 m of the roost tree), bats tended to roost in low-severity burn sites, as opposed to unburned or severely burned areas. The authors believe that M. californicus, though clutter-tolerant, did not select for unburned sites because the dense vegetation of the unburned areas was not preferred over the moderate complexity of low-severity sites. Low-severity burn sites did not typically express more than minor burns at the base of some trees, which would presumably not meaningfully affect bats as they tended to roost higher in trees. Similarly, Gonsalves et al. (2022) found a clutter tolerant bat in south-eastern Australia (Nyctophilus corbeni) selected low-severity areas for roosting, avoiding areas impacted by high severity burns. These results differ from a study by Doty et al. (2016) that showed the lesser long-eared bat (Nyctophilus geoffroyi) from south-eastern Australia regularly roosted in severely burned trees and habitat. However, this study was conducted in autumn when daytime temperatures were cooler and thus those bats may have capitalized on the thermal variability afforded by dark, burned, bark to facilitate passive rewarming. The SQF complex fire left large patches of habitat completely burned, with little canopy cover and trees that were completely burned through. It is not surprising that these clutter-tolerant bats did not prefer roosting in these areas, as the dark, burned trees would experience more fluctuation in daytime temperature due to increased absorption of solar radiation, which may not be physiologically suitable for bats in areas where summer temperatures regularly reach over 30 °C. Additionally, because the trees were in direct sunlight, this would leave bats at even greater risk for overheating. Unfortunately, very little research has been conducted on post-wildfire roost selection (see Snider et al. 2013; Schwab 2006), though a greater number of studies have focused on roost selection following prescribed fire (see Loeb and Blakey 2021). Post-wildfire acoustic research is challenging to compare with our study because areas where bats forage and roost are not necessarily the same (e.g., Popa-Lisseanu et al. 2009).
In the landscape (at the roost habitat-scale), bats preferred to roost within the burn mosaic, as 21 out of 22 roosts were located in or within 5 m of the burn boundary (the fire boundary, Fig. 1), despite the capture location being near the border of the burned area, with presumably equal access to unburned landscapes. While this pattern of landscape use may be based on a number of other factors, including social interactions, we do not believe bats are selecting these roost locations based on water resources, given their capture site was at a large water source (South Fork of the Kaweah River), nor elevation, which was uncorrelated with distance to unburned areas. In the field, an author observed no obvious barriers or deterrents that would prevent bats from flying north into the unburned areas. We suspect that the burn mosaic provided a balance of roost habitats with appropriate microclimates (unburned or lightly burned areas with large trees and exfoliating bark) and proximity to productive foraging habitat, as M. californicus, though clutter tolerant, may use edge habitat within heterogeneously burned areas to minimize energy expenditure (Steel et al. 2019).
While roost selection may favor sun-exposed or insulated roosts based on season, the foraging activity of bats is complicated by agility, in that both clutter- and open- adapted bats may exploit different patches of landscape created by fire based on morphological traits (Starbuck et al. 2020). Soon after wildfire, bats that are adapted to vegetative complexity may reduce activity in areas that have been severely burned and increase activity after regrowth occurs (Ancillotto et al. 2021). Research conducted on land with historical wildfires in the central Appalachians showed that activity largely did not depend on burn severity when at least 3 years had passed since fire (Austin et al. 2020). Additionally, burn severity may not be the only important landscape predictor of roost or foraging habitat selection. Starbuck et al. (2020) demonstrated that both elevation and water density were more important predictors of bat activity than fire severity. While water is clearly an important driver of roost selection for some bats, as those bats in our study all stayed relatively close to the water source, it is particularly interesting that the majority of bats did not choose to roost at higher and cooler elevations during summer, only occasionally selecting roosts at high elevation among a grove of giant Sequoia trees (Seqouiadendron giganteum). The mean elevation over which bats roosted was not very different between unburned (1196.9 ± 99.7 m), low severity (1275.8 ± 54.5 m), and high severity (1125.2 ± 0.0 m) roost sites; thus, we are confident that roost selection was not affected by elevation alone. Nardone et al. (2015) demonstrated that male Daubenton’s bats (M. daubentonii) roosting at higher altitude during summer partially exhibited poorer body condition than bats roosting at lower altitude, potentially due to reduced prey access. However, high altitude bats were more capable of entering torpor and reducing energy expenditure. Thus, it is possible that bats in our study roosted at lower elevation sites for easier access to prey, with a tradeoff of more shallow and shorter torpor bouts. Bats in the study were either post-lactating females or non-reproductive males, so they were not experiencing their highest energy and water demand of the season (which is during lactation for females).
Bats also roosted in trees with larger DBH and percentage of exfoliating bark compared to what was available. Trees with larger DBH are commonly preferred by tree-roosting bats (Kalcounis-Rüppell et al. 2005). These results are also consistent with Brigham et al. (1997), wherein M. californicus were more likely to choose roost trees that were larger in diameter and roosted both under exfoliating bark or in holes or cracks in trees. While we cannot confidently assume that bats always roosted underneath exfoliating bark or in crevices, as many roost trees had sufficient holes or cracks for roosting, the presence of exfoliating bark appears to be an important consideration for roost location for this species. The studies by Brigham et al. (1997) and Barclay and Brigham (2001) indicate that M. californicus switch roosts frequently, either because of declining suitability of previously used roosts or that suitable roost trees are abundant. Although bats in the previous studies were mostly reproductive females and bats from our study were either nonreproductive males or postlactating females, our results also demonstrate frequent roost-switching, likely due to the abundance of appropriate roosts within their habitat. Of the 6 bats that were tracked on multiple days, individuals switched roosts every 1.3 ± 1.0 (s.d.) days. This study is also the first to provide evidence for M. californicus roosting in deciduous trees on the landscape in addition to evergreen. Although this is unsurprising given the wide availability of oak species in the study area, the result demonstrates the flexibility of this species in roost selection, as the bats used 6 different species of trees, of which only 2 were evergreen, whereas Brigham et al. (1997) determined the species to select for patches of ponderosa pine. Supporting the ecology of the clutter-adapted M. californicus, the species also avoided roosting in habitat with little canopy and preferred roosting in areas with taller trees relative to their availability. Chosen roost trees typically experienced moderate sun exposure (see Table 1) due to their height, although roosts were almost exclusively lower than the tallest point of the tree.
In line with our third hypothesis, M. californicus used torpor frequently during the coolest part of the day and did so regularly in low severity burn area roosts. However, due to our low sample size, we could not statistically correlate torpor use with burn severity or roost tree diameter. Future work is needed to elucidate torpor use in relation to fire severity and roost tree characteristics. Our study supports the idea that torpor expression appears to be widespread across the Chiroptera regardless of climate zone (Fjelldal et al. 2022) and supplements previous studies that indicate bats readily use torpor in summer when possible (e.g., Turbill et al. 2003, Wojciechowski et al. 2007, Bondarenco et al. 2016). While body temperature during torpor in insectivorous bats typically reaches low levels in winter (Stawski et al. 2014), our species were constrained by the hot summer temperatures up to a maximum of 41.3 °C, and thus M. californicus exhibited only shallow bouts of torpor, on average 5.9 ± 2.8 °C below the torpor onset threshold of 30 °C and exhibited 1–2 bouts of torpor on most study days. Another bat species of western North America, the fishing myotis (M. vivesi), infrequently expressed short bouts of torpor during summer in the cooler parts of the day (Salinas et al. 2014), possibly to cope with challenging weather conditions that reduced prey availability.
We believe M. californicus exhibited bouts of torpor to deal with water loss energy expenditure associated with the hot and dry summer conditions in the foothills of Sequoia National Park. Myotis californicus appears to use torpor when possible and adjusts to the altered landscape by selecting thermally beneficial roosts and habitat within the burn mosaic. Alston et al. (2022) recently demonstrated that a species of heterothermic bat (Myotis thysanodes) do not select for microhabitats with specific temperature profiles and instead adjust torpor expression to meet energetic needs. However, this study focused on male bats of only one species and was conducted in South Dakota where mean high summer temperatures range between 22 and 27 °C, substantially cooler than our study site. Thus, we suggest that microhabitat selection may still be an important driver of torpor expression in hot, dry areas like the foothills Sequoia National Park. Additionally, evaporative water loss of euthermic bats is high, even for those bats that occupy arid regions. The expression of torpor reduces evaporative water loss in bats (Hosken and Withers 1997), and because the high summer temperatures of Sequoia and Kings Canyon National Parks may preclude the use of torpor in the afternoon, expression is limited to early hours of the day. Further replication of studies similar to ours is also needed to reveal how torpor patterns differ between burned and unburned landscapes and whether burn severity affects torpor expression in bats.
Conclusions
Due to the intensity of wildfires in the western United States, understanding how and if bats roost in severely burned habitat in the years following fires is essential to understanding whether bats are truly resilient within these landscapes. Our research demonstrates that 10 months following wildfire, clutter-tolerant bats avoided unburned and severely burned habitat, instead demonstrating preference for low-severity burn areas and large diameter trees with exfoliating bark. Given parts of North America now experience such large-scale and severe fires that climatic regeneration thresholds are at risk of being crossed (Steel et al. 2022; Davis et al. 2019), reduction in roost and foraging habitat may occur for multiple species, particularly those that are clutter-adapted. Restoring historical mixed-severity fire regimes and creating a fire mosaic may be critical for maintaining both roosting and foraging habitat for bats in this region’s fire-affected forests. Wildfire may affect the torpor patterns (Doty et al. 2016) and subsequent energetics savings of bats; therefore, considering the physiological responses of bats in addition to roost and foraging habitat selection will be vital to understanding the energetic consequences of wildfire on North American heterotherms.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Alston, J. M., M. E. Dillon, D. A. Keinath, I. M. Abernathy, and J. R. Goheen. 2022. Daily torpor reduces the energetic consequences of microhabitat selection for a widespread bat. Ecology 103: e3677. https://doi.org/10.1002/ecy.3677.
Ancillotto, L., L. Bosso, P. Conti, and D. Russo. 2021. Resilient responses by bats to a severe wildfire: Conservation implications. Animal Conservation 24 (3): 470–481. https://doi.org/10.1111/acv.12653.
Armitage, D. W., and H. K. Ober. 2012. The effects of prescribed fire on bat communities in the longleaf pine sandhills ecosystem 93(1):102-114. https://doi.org/10.1644/11-MAMM-A-169.1
Austin, L.V., A. Silvis, W.M. Ford, and K.E. Powers. 2020. Effects of historic wildfire and prescribed fire on site occupancy of bats in Shenandoah National Park, Virginia, USA. Journal of Forestry Research 31: 1255–1270. https://doi.org/10.1007/s11676-019-00923-y.
Barclay, R.M.R., and R.M. Brigham. 2001. Year-to-year reuse of tree-roosts by California bats (Myotis californicus) in Southern British Columbia. The American Midland Naturalist 146 (1): 80–85.
Barclay, R.M.R., M.C. Kalcounis, L.H. Crampton, C. Stefan, M.J. Vonhof, and R.M. Brigham. 1996. Can external radiotransmitters be used to assess body temperature and torpor and bats? Journal of Mammalogy 77 (4): 1102–1106. https://doi.org/10.2307/1382791.
Blakey, R.V., E.B. Webb, D.C. Kesler, R.B. Siegel, D. Corocoran, and M. Johnson. 2019. Bats in a changing landscape: Linking occupancy and traits of a diverse montane bat community to fire regime. Ecology and Evolution 9 (9): 5324–5337. https://doi.org/10.1002/ece3.5121.
Blakey, R.V., E.B. Webb, D.C. Kesler, R.B. Siegel, D. Corcoran, J.S. Cole, and M. Johnson. 2021. Extent, configuration and diversity of burned and forested areas predict bat richness in a fire-maintained forest. Landscape Ecology 36 (4): 1101–1115. https://doi.org/10.1007/s10980-021-01204-y.
Bondarenco, A., G. Körtner, and F. Geiser. 2014. Hot bats: Extreme thermal tolerance in a desert heat wave. Naturwissenschaften 101: 679–685. https://doi.org/10.1007/s00114-014-1202-2.
Bondarenco, A., G. Körtner, and F. Geiser. 2016. How to keep cool in a hot desert: Torpor in two species of free-ranging bats in summer. Temperature 3 (3): 476–483. https://doi.org/10.1080/23328940.2016.1214334.
Bosso, L., L. Ancillotto, S. Smeraldo, S. D’Arco, A. Migliozzi, P. Conti, and D. Russo. 2018. Loss of potential bat habitat following a severe wildfire: A model-based rapid assessment. International Journal of Wildland Fire 27 (11): 756–769. https://doi.org/10.1071/WF18072.
Boyles, J.G., and D.P. Aubrey. 2006. Managing forests with prescribed fire: Implications for a cavity-dwelling bat species. Forest Ecology and Management 222: 108–115. https://doi.org/10.1016/j.foreco.2005.09.024.
Boyles, J.G., and B. Smit. 2011. A new comparative metric for estimating heterothermy in endotherms. Physiological and Biochemical Zoology 84 (1): 115–123. https://doi.org/10.1086/656724.
Brigham, R.M., M. J. Vonhof, R. M. R. Barclay, and J.C. Gwilliam. 1997. Roosting behavior and roost-site preferences of forest-dwelling bats (Myotis californicus). Journal of Mammalogy 78 (4): 1231–1239. https://doi.org/10.2307/1383066.
Buma, B., Weiss, S., Hayes, K., and M. Lucas. 2020. Wildland fire reburning trends across the US West suggests only short-term negative feedback and differing climatic effects. Environmental Research Letters 15: 034026. https://doi.org/10.1088/1748-9326/ab6c70
Burnham, K. P., and D. R. Anderson. 2002. Model selection and inference: A practical information-theoretic approach. 2nd edition. Springer-Verlag.
Calenge, C. 2006. The package adehabitat for the R software: a tool for the analysis of space and habitat use by animals. Ecological Modelling 197:516-519.
Cave, H., M. Adams, T. Jaeger, T. Peet, L. Staats, D. Garneau, and M. Lesser. 2021. Wildlife response to wildfire in a northern New York jack pine barrens. Forests 12 (6): 676. https://doi.org/10.3390/f12060676.
Chen, A. 2022. Evaluating the relationships between wildfires and drought using machine learning. International Journal of Wildland Fire 31 (3): 230–239. https://doi.org/10.1071/WF21145.
Czenze, Z. J., Naidoo, S., Kotze, A., and A. E. McKechnie. 2020. Bat thermoregulation in the heat: Limits to evaporative cooling capacity in three southern African bats. Journal of Thermal Biology 89: 102542. https://doi.org/10.1016/j.jtherbio.2020.102542
Davis, K.T., S.Z. Dobrowski, and P.E. Higuera. 2019. Wildfires and climate change push low-elevation forests across a critical climate threshold for tree regeneration. Proceedings of the National Academy of Science 116 (13): 6193–6198. https://doi.org/10.1073/pnas.181510711.
Dawson, J.P., A.W. Claridge, B. Triggs, and D.J. Paull. 2007. Diet of a native carnivore, the spotted-tailed quoll (Dasyurus maculatus): Before and after an intense wildfire. Wildlife Research 34 (5): 342–351. https://doi.org/10.1071/WR05101.
Di Virgilio, G., J.P. Evans, S.A.P. Blake, M. Armstrong, A.J. Dowdy, J. Sharples, and R. McRae. 2019. Climate change increases the potential for extreme wildfires. Geophysical Research Letters 46 (14): 8517–8526. https://doi.org/10.1029/2019GL083699.
Dickinson, M.B., J.C. Norris, A.S. Bova, R.L. Kremens, V. Young, and M. J. Lacki. 2010. Effects of wildland fire smoke on a tree-roosting bat: Integrating a plume model, field measurements, and mammalian dose-response relationships. Canadian Journal of Forest Research 40: 2187–2203. https://doi.org/10.1139/X10-148.
Doherty, T.S., D.J. Watchorn, V. Miritis, A.J. Pestell, and W.L. Geary. 2023. Cats, foxes and fire: Quantitative review reveals that invasive predator activity is most likely to increase shortly after fire. Fire Ecology 19 (1): 22. https://doi.org/10.1186/s42408-023-00183-6.
Dormann, C.F., J. Elith, S. Bacher, C. Buchmann, G. Carl, G. Carré, J.R.G. Marquéz, B. Gruber, B. Lafourcade, P.J. Leitão, T. Münkemüller, C. Mcclean, P.E. Osborne, B. Reineking, B. Schröder, A.K. Skidmore, D. Zurell, and S. Lautenbach. 2013. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 36 (1): 027–046. https://doi.org/10.1111/j.1600-0587.2012.07348.x.
Doty, A.C., C. Stawski, B.S. Law, and F. Geiser. 2016. Post-wildfire physiological ecology of an Australian microbat. Journal of Comparative Physiology B 186: 937–946. https://doi.org/10.1007/s00360-016-1003-3.
Doty, A.C., S.E. Currie, C. Stawski, and F. Geiser. 2018. Can bats sense smoke during deep torpor? Physiology and Behavior 185 (1): 31–38. https://doi.org/10.1016/j.physbeh.2017.12.019.
Ferraz, A., S. Saatchi, K.J. Bormann, and T.H. Painter. 2018. Fusion of NASA Airborone Snow Obseratory (ASO) lidat time series over mountain forest landscapes. Remote Sensing 10 (2): 164. https://doi.org/10.3390/rs10020164.
Fjelldal, M. A., R. Sørås, and C. Stawski. 2022. Universality of torpor expression in bats. Physiological and Biochemical Zoology 95(4): 326-339. https://doi.org/10.1086/720273.
Ford, W.M., A. Silvis, J.B. Johnson, J.W. Edwards, and J. Karp. 2016. Northern long-eared bat day-roosting and prescribed fire in the Central Appalachians, USA. Fire Ecology 12: 13–27. https://doi.org/10.4996/fireecology.1202013.
Geary, W.L., T.S. Doherty, D.G. Nimmo, A.I. Tulloch, and E.G. Ritchie. 2020. Predator responses to fire: A global systematic review and meta-analysis. Journal of Animal Ecology 89 (4): 955–971. https://doi.org/10.1111/1365-2656.13153.
Geiser, F., C. Stawski, A. Bondarenco, and C. R. Pavey. 2011. Torpor and activity in a free-ranging tropical bat: implications for the distribution and conservation of mammals? Naturwissenschaften 98:447-452. https://doi.org/10.1007/s00114-011-0779-y.
Geiser, F., C. Stawski, A. C. Doty, C. E. Cooper, and J. Nowack. 2018. A burning question: What are the risks and benefits of mammalian torpor during and after fires? Conservation Physiology 6(1): coy057. https://doi.org/10.1093/conphys/coy057
Goforth, N. D. 2015. Short-term effects of wildfire on bat activity on the Valles Caldera National Preserve, New Mexico. Doctoral dissertation, Texas Tech University, USA.
Gonsalves, L., B. Law, T. Brassil, I. Kerr, and C. O-Loughlin. 2022. Roost selection in relation to a patchy, mosaic management burn by a threatened clutter-adapted bat. Forests 2022 (13): 1327. https://doi.org/10.3390/f13081327.
Goss, M. A., D. L. Swain, J. T. Abatzoglou, A. Sarhadi, C. A. Kolden, A. P. Williams, and N. S. Diffenbaugh. 2020. Climate change is increasing the likelihood of extreme autumn wildfire conditions across California. Environmental Research Letters 15, 094016. https://doi.org/10.1088/1748-9326/ab83a7
Hanan, E. J., J. Ren, C. L. Tague, C. A. Kolden, J. T. Abatzoglou, R. R. Bart, M. C. Kennedy, M. Liu, and J. C. Adam. 2021. How climate change and fire exclusion drive wildfire regimes at actionable scales. Environmental Research Letters 16: 024051. https://doi.org/10.1088/1748-9326/abd78e
Hosken, D.J., and P.C. Withers. 1997. Temperature regulation and metabolism of an Australian bat, Chalinolobus gouldii (Chiroptera: Vespertilionidae) when euthermic and torpid. Journal of Comparative Physiology B 167 (1): 71–80. https://doi.org/10.1007/s003600050049.
Johnson, J.S., and M.L. Lacki. 2014. Effects of reproductive condition, roost microclimate, and weather patterns on summer torpor use by a vespertilionid bat. Ecology and Evolution 4 (2): 157–166. https://doi.org/10.1002/ece3.913.
Johnson, J.B., W.M. Ford, J.L. Rodrigue, J.W. Edwards, and C.M. Johnson. 2010. Roost selection by male Indiana Myotis following forest fires in Central Appalachian hardwood forests. Journal of Fish and Wildlife Management 1 (2): 111–121. https://doi.org/10.3996/042010-JFWM-007.
Jorge, M. H., W. M. Ford, S. E. Sweeten, S. R. Freeze, M. C. True, M. J. St. Germain, H. Taylor, K. M. Gorman, E. P. Garrison, and M. J. Cherry. 2021. Winter roost selection of Lasiurine tree bats in a pyric landscape. PLoS ONE 16(2): e0245695. https://doi.org/10.1371/journal.pone.0245695.
Kalcounis-Rüppell, M.C., J.M. Psyllakis, and R.M. Brigham. 2005. Tree roost selection by bats: An empirical synthesis using meta-analysis. Wildlife Society Bulletin 33 (3): 112–1132.
Keeley, J.E., and A.D. Syphard. 2021. Large California wildfires: 2020 fires in historical context. Fire Ecology 17: 17–22. https://doi.org/10.1186/s42408-021-00110-7.
Key, C. H., and N. C. Benson. 2006. FIREMON – Landscape assessment. Pages LA 1–55 In: Lutes, D. C. R. E. Keane, J. F. Caratti, C. H. Key, N. C. Benson, S. Sutherland, and L. J. Gangi. 2006. FIREMON: Fire effects monitoring and inventory system. General Technical Report RMRS-GTR-164-CD. Fort Collins, CO: USDA Forest Service, Rocky Mountain Research Station.
Law, B., A. Doty, M. Chidel, and T. Brassil. 2018. Bat activity before and after a severe wildfire in Pilliga forests: Resilience influenced by fire extent and landscape mobility? Austral Ecology 43 (6): 706–718. https://doi.org/10.1111/aec.12617.
Law, B.S., G. Madani, A. Lloyd, L. Gonsalves, L. Hall, A. Sujaraj, T. Brassil, and C. Turbill. 2022. Australia’s 2019–20 mega-fires are associated with lower occupancy of a rainforest-dependent bat. Animal Conservation 26 (1): 103–114. https://doi.org/10.1111/acv.12805.
Layne, J.T., D. Green, A. Scesny, and L.W. Robbins. 2021. Eastern red bat responses to fire during winter torpor. Forests 12 (10): 1347. https://doi.org/10.3390/f12101347.
Levin, E., A. Ar, Y. Yom-Tov, and N. Kronfeld-Schor. 2012. Summer torpor and sexual segregation in the subtropical bat Rhinopoma microphyllum. In Living in a Seasonal World: Thermoregulatory and Metabolic Adaptations, ed. T. Ruf, C. Bieber, W. Arnold, and E. Mellsi, 167–174. Berling, Heidelberg: Springer.
Lindenmayer, D. B., W. Blanchard, L. McBurney, D. Blair, S. Banks, G. E. Likens, J. F. Franklin, W. F. Laurance, J. A. R. Stein, and P. Gibbons. 2012. Interacting factors driving a major loss of large trees with cavities in a forest ecosystem. PLoS One 7(10): e41864. https://doi.org/10.1371/journal.pone.0041864
Loeb, S.C., and R. V. Blakey. 2021. Bats and fire: A global review. Fire Ecology 17: 29. https://doi.org/10.1186/s42408-021-00109-0
Manly, B. F. J., L. L. McDonald, D. L. Thomas, T. L. McDonald, and W. P. Erickson. 2007. Resource selection by animals: Statistical design and analysis for field studies 2nd edition. Springer Science & Business Media. https://doi.org/10.1007/0-306-48151-0
Mollie, E., V. Benthem, J. Koen, W. Casper, J. Hans, M. Benjamin, M. E. Brooks, K. Kristensen, K. J. V. Benthem, A. Magnusson, C. W. Berg, A. Nielsen, H. J. Skaug, M. Mächler, and B. M. Bolker. 2017. GlmmTMB Balances Speed and Flexibility Among Packages for Zero-inflated Generalized Linear Mixed Modeling.
Nardone, V., L. Cistrone, I. Di Salvo, A. Ariano, A. Migliozzi, C. Allegrini, L. Ancillotto, A. Fulco, and D. Russo. 2015. How to be a male at different elevations: ecology of intra-sexual segregation in the trawling bat Myotis uratusniid. PLoS ONE 10(7):e0134573. https://doi.org/10.1371/journal.pone.0134573
Painter, T.H., D.F. Berisford, J.W. Boardman, K.J. Bormann, J.S. Deems, F. Gehrke, A. Hedrick, M. Joyce, R. Laidlaw, D. Marks, C. Mattmann, B. McGurk, P. Ramirez, M. Richardson, S.M.K. Skiles, F.C. Seidel, and A. Winstral. 2016. The Airborne Snow Observatory: Fusion of scanning lidar, imaging spectrometer, and physically-based modeling for mapping snow water equivalent and snow albedo. Remote Sensing of Environment 184: 139–152. https://doi.org/10.1016/j.rse.2016.06.018.
Pausas, J.G., and J.E. Keeley. 2021. Wildfires and global climate change. Frontiers in Ecology and the Environment 19 (7): 387–395. https://doi.org/10.1002/fee.2359.
Popa-Lisseanu, A.G., F. Bontadina, and C. Ibáñez. 2009. Giant noctule bats face conflicting constraints between roosting and foraging in a fragmented and heterogeneous landscape. Journal of Zoology 278 (2): 126–133. https://doi.org/10.1111/j.1469-7998.2009.00556.x.
R Development Core Team. (2022). R: A language and environment for statistical computing. R Foundation for Statistical Computing. http://www.r-project.org/
Ruf, T., and F. Geiser. 2015. Daily torpor and hibernation in birds and mammals. Biological Reviews 90: 891–926. https://doi.org/10.1111/brv.12137.
Salinas R., V. B., L. G. Herrera M., J. J. Flores-Martínez, and D. S. Johnston. 2014. Winter and summer torpor in a free-ranging subtropical desert bat: The fishing myotis (Myotis vivesi). Acta Chiroterologica 16 (2): 327–336. https://doi.org/10.3161/150811014X687288.
Sanderfoot, O. V., S. B. Bassing, J. L. Brusa, R. L. Emmet, S. J. Gillman, K. Swift, and B. Gardner. 2022. A review of the effects of wildfire smoke on the health and behavior of wildlife. Environmental Research Letters, 16(12): 123003. https://doi.org/10.1088/1748-9326/ac30f6
Schwab, N. A. 2006. Roost-site selection and potential prey sources after wildland fire for two insectivorous bat species (Myotis evotis and Myotis lucifugus) in mid-elevation forests of western Montana. Graduate thesis, University of Montana, 82 pp.
Snider, E.A., P.M. Cryan, and K.R. Wilson. 2013. Roost selection by western long-eared myotis (Myotis evotis) in burned and unburned piñon juniper woodlands of southwestern Colorado. Journal of Mammalogy 94 (3): 540–649. https://doi.org/10.1644/11-MAMM-A-153.1.
Starbuck, C. A., E. S. Considine, and C. L. Chambers. 2020. Water and elevation are more important than burn severity in predicting bat activity at multiple scales in a post-wildfire landscape. PLoS ONE 15(4): e0231170. https://doi.org/10.1371/journal.pone.0231170
Stawski, C., C.K.R. Willis, and F. Geiser. 2014. The importance of temporal heterothermy in bats. Journal of Zoology 292 (2): 86–100. https://doi.org/10.1111/jzo.12105.
Steel, Z. L., G. M. Jones, B. M. Collins, R. Green, A. Koltunov, K. L. Purcell, S. C. Sawyer, M. R. Slaton, S. L. Stephens, P. Stine, and C. Thompson. 2022. Mega-disturbances cause rapid decline of mature conifer forest habitat in California. Ecological Applications 33(2): e2763. https://doi.org/10.1002/eap.2763.
Steel, Z. L., B. Campos, W. F. Frick, R. Burnett, and H. D. Safford. 2019. The effects of wildfire severity and pyrodiversity on bat occupancy and diversity in fire-suppressed forests. Scientific Reports 9: 16300. https://doi.org/10.1038/s41598-019-52875-2.
Thomas, D.L., and E.J. Taylor. 1990. Study designs and tests for comparing resource use and availability. Journal of Wildlife Management 54: 322–330. https://doi.org/10.2307/3809050.
Turbill, C., B. S. Law, and F. Geiser. 2003. Summer torpor in a free-ranging bat from subtropical Australia. Journal of Thermal Biology 28(3): 223-226. https://doi.org/10.1016/S0306-4565(02)00067-0.
Turbill, C. 2006. Roosting and thermoregulatory behaviour of male Gould’s long-eared bats, Nyctophilus gouldi: Energetic benefits of thermally unstable roost trees. Australian Journal of Zoology 54: 57–60. https://doi.org/10.1071/ZO05068.
Williams, A.P., J.T. Abatzoglou, A. Gershunov, J. Guzman-Morales, D.A. Bishop, J.K. Balch, and D.P. Lettenmaier. 2019. Observed impacts of anthropogenic climate change on wildfire in California. Earth’s Future 7 (8): 892–910. https://doi.org/10.1029/2019EF001210.
Willis, C.K.R. 2007. An energy-based body temperature threshold between torpor and normothermia for small mammals. Physiological and Biochemical Zoology 80 (6): 643–651. https://doi.org/10.1086/521085.
Wojciechowski, M.S., M. Jefimow, and E. Tegowska. 2007. Environmental conditions, rather than season, determine torpor use and temperature selection in large mouse-eared bats (Myotis myotis). Comparative Biochemistry and Physiology Part a: Molecular & Integrative Physiology 147 (4): 828–840. https://doi.org/10.1016/j.cbpa.2006.06.039.
Acknowledgements
We would like to thank all of the people who, without their help, this study would not have been possible. Thank you to Dr. Tyler Coleman for assistance in planning and implementing the study and to Gabe Reyes for providing study design advice and recommendations. Additionally, a huge thank you to the field technicians who braved the Sequoia Foothills summer heat to help capture and track bats—Conrad Lucas, Autumn Corrow, Austin Waag, and Jonathan Janes. Thank you to Dr. Kat Bormann and Airborne Snow Observatories Inc. for supplying the data for post-fire canopy cover and tree height. Finally, we thank two anonymous reviewers, whose insights greatly improved the draft manuscript.
Funding
This research was funded by Save the Redwoods Foundation and the Sequoia Science and Learning Center. Funding bodies did not contribute to the design of the study, data collection, analysis, or interpretation, nor the writing of the manuscript.
Author information
Authors and Affiliations
Contributions
AD conceived the ideas and designed the methodology; AD collected the data; RB and RC analyzed the data; AD, RB, and RC led the writing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was carried out in accordance with federal requirements for capture and handling of wildlife. The protocol was approved by the Animal Care and Use Committee of California State University Bakersfield (Protocol # 20–01) and permitted by the United States Department of the Interior—National Park Service (Permit # SEKI-2019-SCI-0054).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Supplemental Material. Fig. S1. Density plot showing the distribution of skin temperature (pink shaded region) and ambient temperature (black line) experienced by 2 roosting Antrozous pallidus over a total of 10 days.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Doty, A.C., Crawford, R.D. & Blakey, R.V. Bat roosting strategies and torpor expression in a wildfire-affected landscape during summer. fire ecol 19, 39 (2023). https://doi.org/10.1186/s42408-023-00199-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s42408-023-00199-y