Abstract
Objectives
To evaluate analgesic and hemodynamic outcome of fentanyl or midazolam as additives to local anesthetic mixture (LAM) for peribulbar block (PBB) during posterior chamber surgery.
Methods
One hundred thirty-two ASA status I to III adult patients aged 50–75 years scheduled for posterior segment surgery (intraocular foreign body and elective vitrectomy) were enrolled in this prospective, randomized, double-blind trial from which 12 patients were excluded. After signing a written fully informed consent for study participation, patients were grouped into 3 groups (40 patients in each group); group C received local anesthetic mixture plus 1 ml plain saline, group F received local anesthetic mixture plus 25 μg fentanyl in 1 ml saline and group M received local anesthetic mixture plus 1 mg midazolam in 1 ml saline. The primary outcome was the onset time of eyelid and globe akinesia. Also, the duration of the block was assessed in the three studied groups. Intraoperative and postoperative hemodynamic measures were assessed. Postoperative analgesia was hourly-assessed using Visual analogue scale (VAS) and rescue analgesia was provided at visual analogue score of > 3.
Results
The number of patients who had fast eyelid and globe akinesia was significantly higher with significantly lower total 15-min score in group F than the other groups. Intraoperative and postoperative hemodynamic measures were non-significantly different between studied groups. Duration of the block was significantly longer in groups F and M than group C with significantly longer duration in group F. The number of patients who required postoperative rescue analgesia was significantly lower with significantly lower number of requests in group F than the other groups.
Conclusion
Additives to local anesthetic mixture during peribulbar block provided satisfactory anesthetic outcome than local anesthetic mixture alone. Fentanyl was superior to midazolam in terms of significantly speed up onset, longer block duration with significantly longer postoperative analgesia and lesser consumption of rescue analgesia. Both additives provided adjusted hemodynamic measures comparable to the control group.
Trial registration
Pan African Clinical Trials Registry (PACTR201708002496243) registered 03/08/2017 retrospectively.
Similar content being viewed by others
Background
Additives to local anesthetic mixture (LAM) during regional anesthesia had been tried for various surgical procedures as documented in recent studies (Li et al. 2016), to increase potency, extent, and improve onset of local anesthesia (Gentili et al. 2016; Pierce et al. 2016).
Fentanyl is considered as one of the most successful opioid analgesics. It is a synthetic, lipophilic phenylpiperidine opioid agonist with analgesic and anesthetic properties. Fentanyl selectively binds to the mu-receptor in the central nervous system (CNS) (Chen and Gupta 2014). Stimulation of the mu-subtype opioid receptor stimulates the exchange of GTP for GDP on the G-protein complex and subsequently inhibits adenylate cyclase, so decreasing in intracellular cAMP and leads to a reduction in the release of neurotransmitters (Celver et al. 2004). The analgesic effect of fentanyl is by two actions: it induces the opening of potassium channels and blocks the opening of N-type voltage-gated calcium channels, resulting in hyperpolarization and reduced neuronal excitability (Rodrigues et al. 2005; Kuhar et al. 2015).
Midazolam is a short-acting benzodiazepine derivative with anxiolytic, amnestic, hypnotic, anticonvulsant, and sedative properties (Griffin 3rd et al. 2013). Midazolam binds to the benzodiazepine receptor at the gamma-aminobutyric acid (GABA) receptor in CNS leading to increased opening of chloride channels, membrane hyperpolarization, and inhibitory effect of GABA in the CNS (Khom et al. 2006; Beltrán González et al. 2014). Midazolam may also interfere with the reuptake of GABA, thereby causing accumulation of GABA in the synaptic cleft (Baburin et al. 2008). The GABA receptors are crucial for induced sedation through α1-receptors and the anxiolytic effects through α2-receptors (Naguib et al. 1995; Newman et al. 2015) and have a role in the regulation of Ca channel activity at legends of peripheral benzodiazepine receptors and in CNS at nanomolar concentration leading to Ca-mediated increase in K conductance in CA1 cells in hippocampal slices with adenosine benzodiazepine actions via adenosine uptake inhibition or enhancing adenosine release. Neuraxial midazolam acts on the benzodiazepine receptors on the gray matter of the spinal cord, the highest concentration of which is found on the lamina II of the dorsal horn. The analgesic effect of neuraxial midazolam is caused by the spinal suppression of sensory functions and its antinociceptive effect mediated by GABAergic mechanisms (Edwards et al. 1990; Lötsch et al. 2013).
We hypothesized that the addition of either fentanyl or midazolam will affect the quality of the peribulbar block, prolonging its anesthetic and analgesic duration. This will later help in providing adequate anesthesia, analgesia, and comfort during lengthy ophthalmic operations. The aim of this study was to evaluate the analgesic and hemodynamic outcome of fentanyl or midazolam as an additive to local anesthetic mixture (LAM) for peribulbar anesthesia during posterior segment surgery.
Methods
The study was approved by the Local Ethics Committee and registered with the Pan African Clinical Trials Registry (PACTR201708002496243) and conducted at Anesthesia Department, Cairo University Hospitals, from January 2017 to August 2017. One hundred thirty-two ASA status I to III adult patients, aged 50–75 years scheduled for posterior segment surgery (intraocular foreign body and elective vitrectomy) were included in this prospective, randomized, double-blind trial. The patients signed written fully informed consent for study participation where 12 patients were excluded from the study.
Complete ophthalmological examination was done by the ophthalmologist to exclude complicated vitreous hemorrhage, diagnosis of any associated disorders as well as ophthalmic ultrasound and biometry was done for all cases to measure the axial length and diagnose posterior staphyloma if it was present. All patients were clinically evaluated for fitness for anesthesia and categorized according to ASA grade and patients who had comorbidities underwent preoperative control and continued their treatment during postoperative (PO) course. Patients with impaired orbital/periorbital sensation had history of abnormal bleeding or allergy to local anesthetics and had posterior staphyloma or with axial length > 30 mm were excluded from the study.
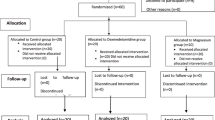
Patients were randomized using computer-generated numbers and were assigned randomly using sealed opaque envelopes prepared by a blinded assistant (consort flow diagram)
Patients were categorized into 3 groups (40 for each) according to the type of additive used with the LAM (2 ml of mepivacaine 3%, 1 ml of hyaluronidase containing 150 IU, 3 ml of bupivacaine 0.5%, 1 ml of lidocaine 2%). as follows:
-
1-
Group C (control group): included patients assigned to receive 1 ml plain saline.
-
2-
Group F (fentanyl group): 1 ml saline containing 25 μg fentanyl was used instead of plain saline.
-
3-
Group M (midazolam group):1 ml saline containing 1 mg midazolam was used instead of plain saline.
Anesthetic procedure
No sedative premedication was administered in the preoperative period. In the operating room, an intravenous line was secured and standard monitoring (heart rate, electrocardiography, oxygen saturation, and noninvasive blood pressure) was applied to all the patients. The study solution for peribulbar block was prepared and performed by the anesthesiologist who was not involved in the study. Then, 3 ml of LAM assigned for each group was injected with needle 3-cm length and 27 G inserted at angle of 45° between the caruncle and medial canthal angle till the tip of the needle touched the ethmoid bone then the direction of the needle was changed to 90° with the hub of the needle at level of the iris. Another 3 ml of LAM was injected at the extreme inferotemporal border of the orbit with the same needle directed downward and medially below the globe. Light orbital compression was applied for 1 min; then eye was evaluated 1, 3, 5, and 10 min for the appearance of proptosis and chemosis. After satisfactory sensory and motor block, oxygen 4 L/min was delivered through a nasal cannula to the patient. Surgery was then allowed to proceed.
Study outcomes
Primary outcomes
The onset of eye block (eyelid and globe akinesia) was assessed and recorded throughout 15 min after LAM injection using a three-point scoring or ocular movement scale (0–complete akinesia–limited movement–normal movement) (Sarvela et al. 1993).
Secondary outcomes
-
1.
Duration of the block was calculated since 15-min assessment after LAM injection till complete recovery.
-
2.
Hemodynamic variables were assessed every 15 min during the entire procedure and every 30 min during the first two postoperative (PO) hours. Hypotension and bradycardia were defined as a 20% decrease in MAP and HR in relation to baseline measures.
-
3.
PO analgesia was assessed by using visual analogue score (VAS) every hour for 6 h PO and was scored as 0 if no pain up to 10 which indicates intolerable pain sensation. PO analgesia was provided if VAS was > 3
Statistical analysis
Sample size calculated according to the standard nomogram for power calculation (Kraemer and Theimann 1987) defined a sample size of 36 patients per group giving the trial 80% power and is sufficient to detect a difference at the 5% significance level. Sample size and power were re-calculated and assured using Power and Sample Size Calculation Software program provided by the Department of Biostatistics, Vanderbilt University. Forty patients were included in each group to compensate for dropouts. Obtained data were presented as mean ± SD, ranges, numbers, and ratios. Results were analyzed using one-way ANOVA with post hoc Tukey HSD test and chi-square test (χ2 test). Statistical analysis was conducted using the SPSS (Version 15, 2006) for Windows statistical package. P value < 0.05 was considered statistically significant (Murphy and Myors 2003)..
Results
One hundred thirty-two patients scheduled for posterior segment surgical procedures were enrolled in the study. They were divided randomly into 3 equal groups (40 patients in each group); group C received LAM plus 1 ml plain saline, group F received LAM plus 25 μg fentanyl in 1 ml saline and group M received LAM plus 1 mg midazolam in 1 ml saline. Mean age of studied patients was 58.4 ± 6.6; range 50–75 years. Sixty-eight patients were males and 52 patients were females. Mean body mass index of studied patients was 31.4 ± 3.2; range 24.6–37.5 kg/m2. Forty patients ASA grade I, 64 patients ASA grade II, and 16 patients ASA grade III. Details of enrolment data of patients categorized according to the received local anesthetic mixture (LAM) are shown in Table 1.
Injection of LAM was conducted successfully for all groups, patients of group F responded significantly (P < 0.05) faster than patients of the other groups with significantly (P < 0.05) fast onset of eyelid and globe akinesia. On the contrary, group M showed non-significantly (P > 0.05) fast onset of eyelid and globe akinesia compared to group C (Fig. 1).
Mean of total score of eyelid akinesia recorded throughout 15-min after LAM injection showed significantly (P < 0.05) progressing decrease in the three groups with significantly (P < 0.05) lower score in group F, but non-significantly (P > 0.05) in group M compared to group C with non-significantly (P > 0.05) lower score in group F compared to group M (Fig. 1). Similarly, mean of total score of globe akinesia recorded throughout 15 min after LAM injection showed significantly (p < 0.05) progressing decrease in the three groups with significantly (P < 0.05) lower score in group F, but non-significantly (P > 0.05) in group M compared to group C with non-significantly (P > 0.05) lower score in group F compared to group M (Fig. 2).
All surgeries were conducted uneventfully without intraoperative (IO) morbidities within a mean operative time of 96 ± 6.5; range 80–120 min with non-significant (P > 0.05) difference between studied groups despite being shorter in group F. Preoperative, intraoperative, and 2-h PO hemodynamic measures showed non-significant (P > 0.05) difference between studied groups; despite being lowest in group M (Tables 2 and 3).
Mean duration till complete recovery of eyelid and globe akinesia was significantly (P < 0.05) longer in groups F and M compared to group C with significantly (P < 0.05) longer duration in group F compared to groups M. Only 46 patients (38.3%) required PO rescue analgesia; 6 patients in groups C and M required it twice, while 40 patients among the three groups required it once with significantly (P < 0.05) lower frequency of patients required rescue analgesia in group F compared to groups C and M and non-significantly (P > 0.05) lower frequency in group M compared to group C. Moreover, the mean number of requests was significantly (P < 0.05) lower in group F compared to group C, but non-significantly (P > 0.05) lower than in group M with non-significantly (P > 0.05) lower mean number of requests in group M compared to group C (Table 4).
Discussion
Our study showed that local anesthetic mixture (LAM) with additives, either fentanyl or midazolam, for peribulbar block (PBB) for posterior chamber surgery improved analgesic outcome manifested as significantly shorter duration till onset of motor block and longer duration of anesthesia that allowed completion of surgery despite the long operative time. Moreover, additives allowed significantly longer duration of postoperative (PO) analgesia and significantly lower consumption of rescue analgesia in comparison to control group received plain LAM.
As regards the differential effects of used additives, fentanyl in comparison to midazolam provided significantly faster anesthesia, longer duration of motor block, and is superior in terms of significantly prolonged PO analgesia with significantly lower consumption of rescue analgesia. Moreover, both additives allowed favorable hemodynamic outcomes manifested as stable blood pressure measures and heart rate comparable to the control group (Dopfmer et al. 1996).
These findings illustrated the beneficial effects of adding fentanyl or midazolam to LAM and go in hand with that reported for both additives during various forms of regional anesthesia; where various studies have demonstrated the presence of GABA receptors in peripheral nerves and the action of midazolam on GABA receptors is well established. Extra synaptic receptors for GABA are present on myelinated axons of peripheral nerves (Morris et al. 1983). Cairns et al. observed the presence of GABA receptors within the temporomandibular joint and its activation could decrease the transmission of nociceptive signals (Cairns et al. 1999).
Jarbo et al (2005) found that midazolam (50 mcg × kg−1) in combination with 30 ml of bupivacaine (0.5%) hastened the onset of sensory and motor block, and improved postoperative analgesia when used in brachial plexus block, without producing any adverse events.
Laiq et al (2008) found that bupivacaine (0.5%) in combination with midazolam (50 mcg × kg−1) quickened the onset as well as prolonged the duration of sensory and motor blockade of the brachial plexus for upper limb surgery. It improved postoperative analgesia without producing any adverse events compared to plain bupivacaine (0.5%) in equal volume. Trivedi and Patel (2010) reported a clinical study in 60 ASA I–II patients undergoing elective upper limb orthopedic surgeries. All the patients were divided into two groups (n = 30 each), group C (clonidine), and group M (midazolam) given injection bupivacaine 0.5% plain 20 ml and injection lignocaine 2% plain 10 ml in supraclavicular brachial plexus block. In group C, clonidine 150 mcg and in group M midazolam 5 mg were injected with local anesthetics. All the patients were observed for onset and duration of sensory and motor blockade, sedation score, and postoperative analgesia with visual analogue scale (VAS) score up to 12 h. They found no clinically significant difference in onset and duration of sensory and motor blockade among both the study groups. In group C more, sedation (score 2) was observed compared to group M (score 1) though the data is not statistically significant. Postoperative analgesia was more prolonged in group C (VAS score < or = 3 for 360 min) as compared to group M (VAS score < or = 3 for 300 min) Edwards et al. (1990) found that intrathecal injection of midazolam has produced segmental analgesic effect in both rats and humans. This analgesic effect is not caused by a local analgesic action of the drug and not accompanied by sedation, and this experiment suggested that the spinally mediated analgesia following intrathecal midazolam may be the result of a combination of the drug with spinal cord benzodiazepine receptors.
Moreover, Weigl et al. (2016) and Khezri et al. (2016) reported that addition of intrathecal fentanyl to spinal anesthesia is effective for intraoperative analgesia and decreases opioid consumption after cesarean section and Kumar et al. (2016) found that low-dose bupivacaine with fentanyl is superior to bupivacaine and butorphanol in terms of early PO recovery resulting in early discharge and better outcome of elderly patients with comorbidity. Also, Singh et al. (2016) found fentanyl as an adjuvant to intrathecal ropivacaine prolongs PO pain relief without increasing duration of motor blockade.
The effect of fentanyl could be mediated through a direct action on the peripheral opioid receptors, in the primary afferent tissues (dorsal roots) (Laduron 1984), or through centrally mediated opioid receptor analgesia after its uptake into the systemic circulation.
Karakaya et al (2001) found that addition of fentanyl to bupivacaine in brachial plexus axillary block approach enhanced and prolonged anesthesia and analgesia, increased duration of sensory and motor block and increased the duration of postoperative analgesia.
A synergism between opioids and local anesthetics has been reported (Nishikawa et al. 2000). It appears that opioids and local anesthetics act their action independently via different mechanisms. Local anesthetics block propagation and generation of neural action potentials by a selective effect on Na channels, whereas opioids act on the opioid receptors creating an increase in a K channel conductance. This action results in hyperpolarization of the nerve cell membrane and a decrease in excitability. Although Na channel block is proposed to be the primary mode of action, local anesthetic also has an effect on synaptic transmission. Li et al. (1995) showed that lidocaine inhibits both substance P binding and substance P-evoked increase in intracellular Ca.
Unfortunately, few studies evaluated these LAM additives for ophthalmic surgeries. However, the obtained results go hand in hand with multiple studies that used varied additives to LAM for peribulbar block (PBB) where Reah et al. (1998), Küçükyavuz and Arici ( 2002), Godarzi et al. (2011), Aissaoui et al. (2010), and Messeha and Elhesy (2015) found the addition of vecuronium (Reah et al. 1998), atracurium (Küçükyavuz and Arici 2002; Godarzi et al. 2011), rocuronium (Aissaoui et al. 2010; Messeha and Elhesy 2015) to LAM decreased the onset time of akinesia, improved the quality of globe and lid akinesia, reduced the need for supplementary injections, and provided better surgical conditions. Also, Madan et al. (2001) and Bharti et al. (2002) found that the addition of clonidine to LAM significantly increases the duration of anesthesia and analgesia after PBB with limited side effects. Moreover, Abo El Enin et al. (2009) found addition of fentanyl to LAM decrees the onset and increases the duration of akinesia and improves quality of PO pain in peribulbar block (PBB) and Channabasappa et al. (2013) found addition of dexmedetomidine to lidocaine and bupivacaine in PBB shortens the onset time and prolongs the duration of the block and postoperative analgesia. Also, Sinha et al. (2016) found addition of 50 mg of magnesium sulfate to the lidocaine-bupivacaine mixture for peribulbar block decreases the onset of akinesia without any obvious side effect.
Conclusion
The use of additives to the local anesthetic mixture during peribulbar block for posterior segment surgery provided satisfactory anesthetic outcome than local anesthetic mixture alone. Fentanyl was superior to midazolam as additive to local anesthetic mixture in terms of significantly faster onset and longer duration of block with significantly longer postoperative analgesia and lesser consumption of rescue analgesia. Moreover, both additives provided adjusted hemodynamic variables comparable to control group.
Study limitations
A limitation to our study was the limited sample size in each group. Further investigations are needed on a wider population sample in order to concur our results, to confirm their safety, and to support the absence of systemic complications. More researches may be suggested using different local anesthetics and different doses of midazolam and fentanyl to confirm our data.
Availability of data and materials
The data that support the findings of this study are available from Cairo University Hospitals but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Cairo University Hospitals.
Abbreviations
- ANOVA:
-
Analysis of variance
- ASA:
-
American Society of anesthesiologists
- ECG:
-
Electrocardiograph
- HR:
-
Heart rate
- Hs:
-
Hours
- IOP:
-
Intra ocular pressure
- IV:
-
Intravenous
- LAM:
-
Local anesthetic mixture
- M:
-
Mean
- MAP:
-
Mean arterial pressure
- O2:
-
Oxygen
- OMS:
-
Ocular movement score
- PBB:
-
Peribulbar block
- PO:
-
Postoperative
- SD:
-
Standard deviation
- SpO2 :
-
Pulse oximeter and oxygen saturation
- VAS:
-
Visual analogue score
References
Abo El Enin MA, Amin IE, Abd El Aziz AS, Mahdy MM, Abo El Enin MA, Mostafa MM (2009) Effect of fentanyl addition to local anaesthetic in peribulbar block. Indian J Anaesth 53(1):57–63
Aissaoui Y, Belyamani L, Kamili ND (2010) Effect of the addition of rocuronium to local anesthetics for peribulbar block. ActaAnaesthesiol Belg 61(2):51–54
Baburin I, Khom S, Timin E, Hohaus A, Sieghart W, Hering S (2008) Estimating the efficiency of benzodiazepines on GABA(A) receptors comprising gamma1 or gamma2 subunits. Br J Pharmacol 155(3):424–433
Beltrán González AN, Pomata PE, Goutman JD, Gasulla J, Chebib M, Calvo DJ (2014) Benzodiazepine modulation of homomeric GABAAρ1 receptors: differential effects of diazepam and 4′-chlorodiazepam. Eur J Pharmacol 743:24–30
Bharti N, Madan R, Kaul HL, Khokhar SK, Mishra S (2002) Effect of addition of clonidine to local anaesthetic mixture for peribulbar block. Anaesth Intensive Care 30(4):438–441
Cairns BE, Sessle BJ, Hu JW (1999) Activation of peripheral GABAA receptors inhibits temporomandibular joint-evoked jaw muscle activity. J Neurophysiol 81(4):1966–1969
Celver J, Xu M, Jin W, Lowe J, Chavkin C (2004) Distinct domains of the mu-opioid receptor control uncoupling and internalization. MolPharmacol. 65(3):528–537
Channabasappa SM, Shetty VR, Dharmappa SK, Sarma J (2013) Efficacy and safety of dexmedetomidine as an additive to local anesthetics in peribulbar block for cataract surgery. Anesth Essays Res 7(1):39–43
Chen C, Gupta A (2014) Clinical and pharmacokinetic considerations of novel formulations of fentanyl for breakthrough cancer pain. Pain Manag 4(5):339–350
Dopfmer UR, Maloney DG, Gaynor PA, Ratcliffe RM, Döpfmer S (1996) Prilocaine 3% is superior to a mixture of bupivacaine and lidocaine for peribulbar anaesthesia. Br J Anaesth 76:77–80
Edwards M, Serrao JM, Gent JP, Goodchild CS (1990) On the mechanism by which midazolam causes spinally mediated analgesia. Anesthesiology. 73(2):273–277 [PubMed]
Gentili ME, Ligier JN, Dermer J, Sleth JC (2016) Spinal bupivacaine and clonidine for orthopaedic and general paediatric surgery in remote location. AnaesthCrit Care Pain Med 16:30154
Godarzi M, Beyranvand S, Arbabi S, Sharoughi M, Mohtaram R, Soltani AE (2011) Comparing the effect of using atracourium and cis-atracourium as adjuvant agents to the local anesthetic substance on peribulbar-induced akinesia. Acta Med Iran 49(8):509–512
Griffin CE 3rd, Kaye AM, Bueno FR, Kaye AD (2013) Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J 13(2):214–223
Jarbo K, Batra YK, Panda NB (2005) Brachial plexus block with midazolam and bupivacaine improves analgesia. Can J Anaesth 52:822–826
Karakaya D, Büyükgöz F, Baré S et al (2001) Addition of fentanyl to bupivacaine prolongs anesthesia and analgesia in axillary brachial plexus block. Canadian J Anesth 26:434–438 [PubMed]
Khezri MB, Tahaei E, Atlasbaf AH (2016) Comparison of postoperative analgesic effect of intrathecal ketamine and fentanyl added to bupivacaine in patients undergoing cesarean section: a prospective randomized double-blind study. Middle East J Anaesthesiol 23(4):427–436
Khom S, Baburin I, Timin EN, Hohaus A, Sieghart W, Hering S (2006) Pharmacological properties of GABAA receptors containing gamma1 subunits. MolPharmacol. 69(2):640–649
Kraemer HC, Theimann S (1987) How many subjects? Statistical power analysis in research. Sage, Newbury Park
Küçükyavuz Z, Arici MK (2002) Effects of atracurium added to local anesthetics on akinesia in peribulbar block. RegAnesth Pain Med 27(5):487–490
Kuhar JR, Bedini A, Melief EJ, Chiu YC, Striegel HN, Chavkin C (2015) Mu opioid receptor stimulation activates c-Jun N-terminal kinase 2 by distinct arrestin-dependent and independent mechanisms. Cell Signal 27(9):1799–1806
Kumar A, Kumar R, Verma VK, Prasad C, Kumar R, Kant S, Kumar G, Singh N, Kumari R (2016) A randomized controlled study between fentanyl and Butorphanol with low dose intrathecal bupivacaine to facilitate early postoperative ambulation in urological procedures. Anesth Essays Res 10(3):508–511
Laduron PM (1984) Axonal transport of opiate receptors in capsaicin sensitive neurones. BrainRes 294:157–160
Laiq N, Khan MN, Arif M, Khan S (2008) Midazolam with bupivacaine for improving analgesia quality in brachial plexus block for upper limb surgeries. J Coll Physicians Surg Pak 18:674–678
Li M, Jin S, Zhao X, Xu Z, Ni X, Zhang L, Liu Z (2016) Does magnesium sulfate as an adjuvant of local anesthetics facilitate better effect of perineural nerve blocks? A meta-analysis of randomized controlled trials. Clin J Pain 32(12):1053–1061
Li YM, Wingrove DE, Too P et al (1995) Local anaesthetics inhibit substance P binding and evoked increases in intracellular Ca2+. Anesthesiology 82:166–173 [PubMed]
Lötsch J, Walter C, Parnham MJ, Oertel BG, Geisslinger G (2013) Pharmacokinetics of non-intravenous formulations of fentanyl. ClinPharmacokinet. 52(1):23–36
Madan R, Bharti N, Shende D, Khokhar SK, Kaul HL (2001) A dose response study of clonidine with local anesthetic mixture for peribulbar block: a comparison of three doses. Anesth Analg. 93(6):1593–1597
Messeha MM, Elhesy AE (2015) Comparison of orbital muscle akinesia caused by rocuronium versus hyaluronidase mixed to the local anesthetic in single injection peribulbar block for cataract surgery. Anesth Essays Res 9(3):374–378
Morris ME, Di Costanzo GA, Fox S, Werman R (1983) Depolarizing action of GABA (γ-aminobutyric acid) on myelinated fibers of peripheral nerves. Brain Res 278(1–2):117–126. https://doi.org/10.1016/0006-8993(83)90230-5
Murphy KR, Myors B (2004) Statistical Power Analysis: A Simple and General Model for Traditional and Modern Hypothesis Tests, (2nd edn). Mahwah: Lawrence Erlbaum Associates, Inc
Naguib M, El Gammer M, Elhattab YS et al (1995) Midazolam for caudal analgesia in children: comparison with caudal bupivacaine. Can J Anaesth 42:758–764
Newman EL, Smith KS, Takahashi A, Chu A, Hwa LS, Chen Y, DeBold JF, Rudolph U, Miczek KA (2015) α2-containing GABA(A) receptors: a requirement for midazolam-escalated aggression and social approach in mice. Psychopharmacology 232(23):4359–4369
Nishikawa K, Kanaya N, Nakayama M et al (2000) Fentanyl improves analgesia but prolongs the onset of axillary brachial plexus block by peripheral mechanism. Anesth Analg. 91:384–387 [PubMed]
Pierce S, Bonanno L, Sanvi S (2016) Effectiveness of intrathecal dexmedetomidine as an adjuvant to bupivacaine spinal anesthesia in adult patients undergoing elective surgery: a systematic review protocol. JBI Database System Rev Implement Rep 14(10):15–21
Reah G, Bodenham AR, Braithwaite P, Esmond J, Menage MJ (1998) Peribulbar anaesthesia using a mixture of local anaesthetic and vecuronium. Anaesthesia. 53(6):551–554
Rodrigues AR, Castro MS, Francischi JN, Perez AC, Duarte ID (2005) Participation of ATP-sensitive K+ channels in the peripheral antinociceptive effect of fentanyl in rats. Braz J Med Biol Res 38(1):91–97
Sarvela J, Nikki P, Paloheimo M (1993) Orbicularis muscle akinesia in regional ophalmic anaesthesia with pH- adjusted bupivacaine; effect of hyaluronidase and epinephrine. Can J Anaesth 40:1028–1033
Singh AP, Kaur R, Gupta R, Kumari A (2016) Intrathecal buprenorphine versus fentanyl as adjuvant to 0.75% ropivacaine in lower limb surgeries. J Anaesthesiol Clin Pharmacol 32(2):229–233
Sinha R, Sharma A, Ray BR, Chandiran R, Chandralekha C, Sinha R (2016) Effect of addition of magnesium to local anesthetics for peribulbar block: a prospective randomized double-blind study. Saudi J Anaesth 10(1):64–67
Trivedi V, Patel N (2010) A comparative clinical study of injection clonidine versus midazolam in supraclavicular brachial plexus block for sedation and postoperative analgesia: a study of 60 cases. J Indian Med Assoc 108:563–567 [PubMed]
Weigl W, Bierylo A, Wielgus M, Krzemień-Wiczyńska S, Szymusik I, Kolacz M, Dabrowski MJ (2016) Analgesic efficacy of intrathecal fentanyl during the period of highest analgesic demand after cesarean section: a randomized controlled study. Medicine (Baltimore) 95(24):e3827
Acknowledgements
The authors provide thanks to all members of Ophthalmology Department, Cairo University Hospital, for assistance for completion of the current work.
Declarations
Registration: Pan African Clinical Trials Registry (PACTR201708002496243) Registered 03/08/2017 retrospectively.
Funding
This research was done in Cairo University Hospitals using the equipment and resources available.
Author information
Authors and Affiliations
Contributions
AAM contributed to the article idea, study design and data analysis, patient recruitment, data collection, and writing up of the first draft of the paper. AAM made a substantial contribution to the conception and design, acquisition of data and analysis, and interpretation of data. AAM also drafted the article, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work thereby ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. TFS made a substantial contribution to the conception and design, acquisition of data and analysis, and interpretation of data. TFS drafted the article, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work thereby ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Research Committee of Anaesthesia Department. Date of Approval: 6/12/2016. Reference number: Committee manuscript Code: N-33-2017/ RS. The patients signed written fully informed consent for study participation.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Abdalla Mohamed, A., Safan, T.F. Fentanyl versus midazolam as additive to local anesthetic mixture for peribulbar block during posterior segment surgery in adult patients a prospective randomized double-blind study. Ain-Shams J Anesthesiol 11, 21 (2019). https://doi.org/10.1186/s42077-019-0036-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s42077-019-0036-8