Abstract
Background
We aim to evaluate the efficacy and safety of a new marine oligosaccharide drug, sodium oligomannate (GV-971), developed in China to treat Alzheimer’s disease (AD).
Methods
We researched the following databases: Embase, Web of Science, PubMed, Cochrane library, and Scopus until Sep 2022. We used the Cochrane risk of bias tool to assess the risk of bias and the GRADE scale to assess the quality of the evidence. The meta-analysis was performed using review manager 5.4.
Results
We included three randomized controlled trials with 1108 patients. Sodium oligomannate improved the AD assessment scale-cognitive subscale compared to the placebo at 12, 24, and 36 weeks (mean difference (MD) = − 0.69, 95% confidence interval (CI) [− 1.23 to − 0.14], p = 0.01), (MD = − 0.68, 95% CI [− 1.26 to − 0.10], P = 0.02), and (MD = − 3.84, 95% CI [− 6.40 to − 1.27], and P = 0.003), respectively. On the other hand, results showed no significance in terms of adverse events and other assessed scales (Clinician’s Interview-Based Impression of Change with caregiver input, AD Cooperative Study-Activities of Daily Living, and Neuropsychiatric Inventory) (p > 0.05).
Conclusions
Sodium oligomannate is a well-tolerated and promising drug for Alzheimer’s patients. However, to better evaluate sodium oligomannate’s efficacy in the clinical setting, we need more randomized controlled trials with larger samples and higher quality.
Similar content being viewed by others
Introduction
Alzheimer’s disease (AD) is a neurodegenerative disease that affects older adults. At the beginning of the disease, it is characterized by the inability to store or encode new information and memories. After that, the behaviors and cognitions abilities decline progressively [1]. According to World Health Organization, in 2012, about 36 million people lived with dementia. Moreover, dementia will affect about 115 million people by 2050. AD is the most common cause of dementia [2]. On the other hand, in the United States, AD was estimated to cost $ 305 billion in 2020 [3]. Moreover, caregivers of AD patients suffer from many psychological problems, such as anxiety, sleep problems, and depression [4].
The pathophysiological mechanism of AD is not precise. But the amyloid hypothesis explained the mechanism by the imbalance between Aβ production and clearance, which leads to sequential cleavage of the amyloid protein by the γ and β [5]. Unfortunately, in the last decade, the drug targeting the main problem, such as amyloid deposition and tau protein, showed minimal relevant clinical efficacy [6,7,8]. On the other hand, several medications were investigated to treat AD, but only four were approved [9,10,11,12]. According to Cui et al., donepezil improved cognitive dysfunction, and it was better than all other AD medications, but it failed to stop the progression of the disease. Therefore, there has been no effective treatment till now.
A new marine oligosaccharide drug, sodium oligomannate (SO) (GV-971), was developed at the Shanghai Institute of Materia Medica, China [13]. It inhibits the neurotoxicity of Aβ aggregation by inhibiting Aβ accumulation and declines the polymerization of the Aβ [14]. In addition, a small amount of SO can enter the brain and directly block the development of Aβ fibrils while destabilizing already-produced fibrils into nontoxic molecules [15, 16].
This systematic review and meta-analysis aim to collect all information available about SO efficacy and safety. Furthermore, as the SO was only used in China, our work may have a role in reconsidering the drug for trials from other countries and increasing the available evidence.
Methods
We performed this systematic review and meta-analysis depending on the recent updates of the PRISMA statement and Cochrane guidelines [17, 18].
Literature search and data collection
Our research was conducted until 18/9/2022 using the following databases: Embase, PubMed, Cochrane library, Scopus, and Web of Science. Our research term was (“Sodium oligomannate” OR “Sodium oligo-mannurarate” OR “GV-971”) AND (“Alzheimer Syndrome” OR “Alzheimer-Type” OR ATD OR “Alzheimer Type” OR “Alzheimer Dementia” OR “Alzheimer's Disease" OR "Alzheimer Sclerosis" OR "Alzheimer Disease" OR "Alzheimers Disease" OR "Familial Alzheimer Disease" OR FAD).
Studies selection and eligibility criteria
Randomized control trials (RCTs) were only included in our research with the following criteria (1) population: patients with AD; (2) intervention: sodium oligomannate; (3) comparator: placebo; (4) outcomes: safety and efficacy outcomes as listed below. After the research was done, first, we removed the duplicates using EndNote. After that, we performed title and abstract screening, followed by the full-text screening based on our eligibility criteria. Moreover, the references of the included studies were reviewed by two independent reviewers for any missed relevant articles. Two authors have done these steps, and a third reviewer resolved any conflicts between both authors.
Quality assessment
The Cochrane risk of bias tool (version 1) has evaluated our included studies [19]. The following domains make up this tool: (1) detection selection bias and other biases; (2) allocation of arms; (3) participant and investigator blinding; (4) assessment of outcomes and their blinding; and (5) randomization of the population. The possibility of bias in judgment can be a high, low, or ambiguous risk of bias. We used the grading of recommendations assessment, development and evaluation (GRADE) methodology (GRADEpro, version 20. McMaster University, 2013) to assess the quality of evidence of the analyzed outcomes [20].
Data extraction
We retrieved the data in an Excel sheet. The extraction sheets contained the following: (1) summary data: study arms, trial registration, inclusion criteria, main outcomes. (2) baseline characteristics of the selected population: age, gender, education, disease time, Alzheimer’s Disease Assessment Scale-cognitive subscale (ADAS cog), the Clinician’s Interview-Based Impression of Change with caregiver input (CIBIC+), Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL), and Neuropsychiatric Inventory (NPI). (3) outcomes: adverse events, ADAS cog, CIBIC+, ADCS-ADL, and NPI.
Data synthesis
Data synthesis was done using Review Manager software version 5.4. We reported risk ratios (RR) and 95% confidence intervals (95% CI) in case the data were dichotomous. If the data were continuous, we mentioned mean differences (MD) and 95% CI. Heterogeneity was tested using the I-square test (I2) and the Chi-square test. The difference was reported as significant if the p-value < 0.5. According to heterogeneity, the studies were considered heterogeneous if the p-value of Chi-square was < 0.1 and the I2 value was above 50%. If the data were heterogeneous, we pooled them in the random effects model, while for homogeneous data pooled them in the fixed effect model. Finally, we performed a subgroup analysis for ADAS cog based on the follow-up periods.
Results
Literature search and study selection
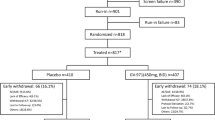
Relied on our strategy for strategy, we found 46 articles after removing duplicates. After title and abstract screening, four studies were available for full-text screening. Then, three studies met our inclusion criteria and were suitable for the quantitative analysis (Fig. 1).
Study characteristics
We included three RCT articles [21,22,23] with a total sample size of 1108 participants, 603 (54.4%) individuals received SO, whereas 505 (45.6%) received placebo or donepezil. All studies were conducted in China. The SO dose differed between studies from 450 mg to 600 and 900 mg twice daily. Most of the patients were females, and the mean age was between 60 and 70 in all groups (Table 1).
The quality of the included studies
Two studies had a low risk of bias [22, 23]. However, Zhang et al. [21] were unclear regarding performance and detection biases with a high risk of other biases (Fig. 2). The GRADE tool revealed very low to moderate overall evidence quality (Additional file 1).
Outcomes
Adverse events
All studies [21,22,23] were pooled in this outcome, and the total sample size was 1108. The results showed no significant difference in adverse events between the SO group and the placebo group (RR = 0.79, 95% CI [0.61–1.03], p-value = 0.08). the analyzed studies were homogenous with the fixed effect model (p-value = 0.85, I2 = 0%) (Fig. 3).
Change in ADAS cog score
At 12 weeks
Two studies [21, 22] reported this outcome with a total population of 840 participants. The pooled estimate suggested SO group had a significantly lower ADAS cog score than the placebo group (MD = − 0.69, 95% CI [− 1.23 to − 0.14], p-value = 0.01). The results were homogeneous (p-value = 0.43, I2 = 0%) (Fig. 4A).
At 24 weeks
The pooled studies (sample size = 1014) showed that there was no significant difference in ADAS cog score between the intervention and control group (MD = − 2.01, 95% CI [− 5.09 to 1.07], p-value = 0.20). The results were heterogeneous (p-value < 0.00001, I2 = 98%) with random effect model (Fig. 4A).
After removing Zhang et al. [21], the heterogeneity was resolved (p-value = 0.85, I2 = 0%). Moreover, the data significantly favored the SO group over the placebo group (MD = − 0.68, 95% CI [− 1.26 to − 0.10], p-value = 0.02) (Fig. 4B).
At 36 weeks
The studies measured this outcome at 36 weeks with a total sample size of 732. The ADAS cog score declined significantly in the SO group compared to the placebo group with a (MD = − 3.84, 95% CI [− 6.40 to − 1.27], and p-value = 0.003). The results were heterogeneous (p-value < 0.00001, I2 = 96%) (Fig. 4A).
Change in ADL score (from baseline to the last follow-up period)
The pooled results (sample size = 968) showed that there was no difference between the placebo and SO group (MD = 0.34, 95% CI [− 0.65 to 1.32], p-value = 0.50). The results were homogeneous (p-value = 0.78, I2 = 0%) (Fig. 5).
Change in CIBIC scale (from baseline to the last follow-up period)
Two studies [21, 22] reported this outcome with a sample size of 728. The analysis suggested no significant difference between the intervention and placebo group (MD = − 2.44, 95% CI [− 7.21 to 2.34], p-value = 0.32). The results were heterogeneous (p-value < 0.00001, I2 = 95%) (Fig. 6).
Change of NPI score (from baseline to the last follow-up period)
The results (sample size = 968) concluded no variation in NPI score between the SO and placebo groups (MD = − 2.19, 95% CI [− 8.59 to 4.21], p-value = 0.50) (Fig. 7A). The heterogeneity (p-value < 0.00001, I2 = 98%) was resolved after removing Zhang et al. [21] (p-value = 0.28, I2 = 15%). The results remained insignificant (MD = 0.48, 95% CI [− 0.78 to 1.73], p-value = 0.45) (Fig. 7B).
Discussion
Our analysis concluded a significant difference between the SO group and placebo group in the ADAS cog score at 12, 24, and 36 weeks. Moreover, there was no difference between the SO and placebo groups in adverse events. On the other hand, the results were insignificant according to the following outcomes: ADL, CIBIC, and NPI at the last follow-up.
The SO proposed has two mechanisms of action. First, it prevents Aβ from aggregating and breaks down Aβ aggregates into harmless conformers [14, 15]. Second, it inhibits neuroinflammation by reconstituting gut microbiota and decreasing metabolite-driven peripheral immune cell influx into the nervous system [24].
Compared to the placebo and donepezil, the SO improved the ADAS cog scale, which means that the SO may have a role in enhancing the AD patients' condition daily [25]. This scale measured 12 essential tasks: word recall, commands, orientation, remembering test instructions, and word-finding difficulty. These tasks are the most critical in AD patients' life. Therefore, improving the ADAS cog may improve the quality of life of patients and their caregivers.
A previous meta-analysis concluded that galantamine, donepezil, and rivastigmine improved the ADAS cog scale [26]. Our results remarkably resemble the effects of these cholinesterase inhibitors. Moreover, the mean difference and effect size increased with the drug's period. Therefore, our drug is a good candidate for treating AD patients.
SO was a safe and well-tolerated drug. Most adverse events were infections, gastrointestinal tract disorders, or nervous system problems. Furthermore, none of the adverse events were more common in the SO group compared to the placebo group and donepezil group. Finally, according to safety and efficacy, SO is similar to the already approved drug and is a good candidate for further research.
Our results were insignificant in three scales (CIBIC, ADL, and NPI). The insignificance might be attributed to the size of the sample being relatively small, culture variation, and behavioral evaluation may depend on the different interpretations of cultures [27].
Our study is the first meta-analysis of SO medication, collecting all available evidence. Moreover, all studies are randomized controlled studies. On the other hand, it has limitations. First, the number of patients is relatively small. Second, all studies conducted in the same country may be liable to selection and racial bias. Third, the absence of a diagnostic amyloid biomarker leads to including some patients with non-amyloid-related dementia. Finally, the study period was slightly short, which may mask some delayed side effects.
Three other studies with a sample size of 3450 patients are being conducted now [28, 29]. The results of these studies may give more evidence about the SO medication and may affect the outcomes measured in this study. Therefore, for further research, we recommend more RCTs on the SO with larger sample sizes and different doses in different places and situations. Moreover, we need to evaluate the drug with various comorbidities to know how it will affect elderly patients if used for a long time.
Conclusions
Sodium oligomannate is a promising drug for Alzheimer's patients and does not cause significant adverse events. However, to better evaluate the efficacy of SO in the clinical setting, we need more randomized controlled trials with larger samples and higher quality.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AD:
-
Alzheimer’s disease
- SO:
-
Sodium oligomannate
- RCTs:
-
Randomized controlled trials
- GRADE:
-
The Grading of Recommendations Assessment, Development and Evaluation
- ADAS cog:
-
Alzheimer’s Disease Assessment Scale-cognitive subscale
- CIBIC+:
-
The Clinician’s Interview-Based Impression of Change with caregiver input
- ADCS-ADL:
-
Alzheimer’s Disease Cooperative Study-Activities of Daily Living
- NPI:
-
Neuropsychiatric inventory
- RR:
-
Risk ratio
- MD:
-
Mean difference
- CI:
-
Confidence interval
References
Soria Lopez JA, González HM, Léger GC. Alzheimer’s disease. Handb Clin Neurol. 2019;167:231–55.
WHO. Dementia cases set to triple by 2050 but still largely ignored: WHO 2012. Available from: https://www.who.int/news/item/11-04-2012-dementia-cases-set-to-triple-by-2050-but-still-largely-ignored.
Wong W. Economic burden of Alzheimer disease and managed care considerations. Am J Manag Care. 2020;26(8 Suppl):S177–83.
Liu S, Li C, Shi Z, Wang X, Zhou Y, Liu S, et al. Caregiver burden and prevalence of depression, anxiety and sleep disturbances in Alzheimer’s disease caregivers in China. J Clin Nurs. 2017;26(9–10):1291–300.
Serý O, Povová J, Míšek I, Pešák L, Janout V. Molecular mechanisms of neuropathological changes in Alzheimer’s disease: a review. Folia Neuropathol. 2013;51(1):1–9.
Salloway S, Sperling R, Fox NC, Blennow K, Klunk W, Raskind M, et al. Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer’s disease. N Engl J Med. 2014;370(4):322–33.
Castello MA, Jeppson JD, Soriano S. Moving beyond anti-amyloid therapy for the prevention and treatment of Alzheimer’s disease. BMC Neurol. 2014;14:169.
Panza F, Solfrizzi V, Imbimbo BP, Logroscino G. Amyloid-directed monoclonal antibodies for the treatment of Alzheimer’s disease: the point of no return? Expert Opin Biol Ther. 2014;14(10):1465–76.
Rogers SL, Farlow MR, Doody RS, Mohs R, Friedhoff LT. A 24-week, double-blind, placebo-controlled trial of donepezil in patients with Alzheimer’s disease. Donepezil Study Group. Neurology. 1998;50(1):136–45.
Rösler M, Anand R, Cicin-Sain A, Gauthier S, Agid Y, Dal-Bianco P, et al. Efficacy and safety of rivastigmine in patients with Alzheimer’s disease: international randomised controlled trial. BMJ. 1999;318(7184):633–8.
van Dyck CH, Tariot PN, Meyers B, Malca RE. A 24-week randomized, controlled trial of memantine in patients with moderate-to-severe Alzheimer disease. Alzheimer Dis Assoc Disord. 2007;21(2):136–43.
Wilcock GK, Lilienfeld S, Gaens E. Efficacy and safety of galantamine in patients with mild to moderate Alzheimer’s disease: multicentre randomised controlled trial. Galantamine International-1 Study Group. BMJ. 2000;321(7274):1445–9.
Wang T. New drug research and development for Alzheimer’s pathology: present and prospect. Shanghai Arch Psychiatry. 2017;29(4):237–9.
Kong LN, Geng MY, Mu L, Xin XL, Yang N, Zuo PP. Effects of acidic oligose on differentially expressed genes in the mice model of Alzheimer’s disease by microarray. Yao Xue Xue Bao. 2005;40(12):1105–9.
Fan Y, Hu J, Li J, Yang Z, Xin X, Wang J, et al. Effect of acidic oligosaccharide sugar chain on scopolamine-induced memory impairment in rats and its related mechanisms. Neurosci Lett. 2005;374(3):222–6.
Hu J, Geng M, Li J, Xin X, Wang J, Tang M, et al. Acidic oligosaccharide sugar chain, a marine-derived acidic oligosaccharide, inhibits the cytotoxicity and aggregation of amyloid beta protein. J Pharmacol Sci. 2004;95(2):248–55.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264–9 (w64).
Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions. USA: Wiley; 2019.
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:5928.
Mustafa RA, Santesso N, Brozek J, Akl EA, Walter SD, Norman G, et al. The GRADE approach is reproducible in assessing the quality of evidence of quantitative evidence syntheses. J Clin Epidemiol. 2013;66(7):736–42 (quiz 42.e1-5).
Zhang LF, Zhang YP, Lin PX, Xue LH. Efficacy and safety of sodium oligomannate in the treatment of Alzheimer’s disease. Pak J Pharm Sci. 2022;35(3):741–5.
Xiao S, Chan P, Wang T, Hong Z, Wang S, Kuang W, et al. A 36-week multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 3 clinical trial of sodium oligomannate for mild-to-moderate Alzheimer’s dementia. Alzheimers Res Ther. 2021;13(1):62.
Wang T, Kuang W, Chen W, Xu W, Zhang L, Li Y, et al. A phase II randomized trial of sodium oligomannate in Alzheimer’s dementia. Alzheimers Res Ther. 2020;12(1):110.
Wang X, Sun G, Feng T, Zhang J, Huang X, Wang T, et al. Sodium oligomannate therapeutically remodels gut microbiota and suppresses gut bacterial amino acids-shaped neuroinflammation to inhibit Alzheimer’s disease progression. Cell Res. 2019;29(10):787–803.
Kueper JK, Speechley M, Montero-Odasso M. The Alzheimer’s disease assessment scale-cognitive subscale (ADAS-Cog): modifications and responsiveness in pre-dementia populations. A narrative review. J Alzheimers Dis. 2018;63(2):423–44.
Hansen RA, Gartlehner G, Webb AP, Morgan LC, Moore CG, Jonas DE. Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer’s disease: a systematic review and meta-analysis. Clin Interv Aging. 2008;3(2):211–25.
Robert P, Ferris S, Gauthier S, Ihl R, Winblad B, Tennigkeit F. Review of Alzheimer’s disease scales: is there a need for a new multi-domain scale for therapy evaluation in medical practice? Alzheimers Res Ther. 2010;2(4):24.
Long-term Efficacy and Safety Study of GV-971. https://ClinicalTrials.gov/show/NCT05181475.
Efficacy and Safety of Donepezil and Sodium Oligomannate in Patients With Mild to Moderate Alzheimer's Disease. https://ClinicalTrials.gov/show/NCT05114499.
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
AZN and IK: conceptualization, methodology, screening and formal analysis; AGT: screening, quality assessment and writing—original draft; AIH and AFH: data collection, data extraction, and writing—original draft; MSZ: quality assessment and supervision. All authors reviewed the manuscript and approved it for publication.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
The quality of evidence of the analyzed outcomes using the GRADE tool.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nourelden, A.Z., Kamal, I., Tawfik, A.G. et al. Safety and efficacy of sodium oligomannate in patients with Alzheimer’s disease: a systematic review and meta-analysis. Egypt J Neurol Psychiatry Neurosurg 59, 82 (2023). https://doi.org/10.1186/s41983-023-00682-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41983-023-00682-y