Abstract
Background
Attention Deficit Hyperactivity Disorder (ADHD) is a prevalent neurological disorder. ADHD has been linked to epilepsy.
Main body
ADHD was found to be present in 30–40% of epileptic children. Researchers have developed numerous theories to explain how and why ADHD and epilepsy coexist. Whether ADHD and epilepsy symptoms are caused by co-occurring psychiatric disorders or by the temporary effects of epileptic discharges or by antiepileptic medicines is critical to consider. Diagnosis and treatment of individuals with ADHD and epilepsy are complicated and challenging from the clinical base.
Conclusions
Comorbidity between ADHD and epilepsy is still challenging to understand. The two diseases have a bidirectional link, so the association may not be coincidental. A disputable point is whether co-occurring ADHD and epilepsy symptoms represent a comorbid psychiatric disorder or are the epileptic discharges’ temporary effects, and are they related to antiepileptic drugs (AEDs). It is recommended to follow up with children with epilepsy or ADHD as they may develop comorbidity after a while.
Similar content being viewed by others
Introduction
Attention Deficit Hyperactivity Disorder (ADHD) is a prevalent neurological disorder that begins during childhood and is associated with structural and functional abnormalities in various brain areas [1]. Since the 1950s, ADHD has been linked to epilepsy [2]. ADHD is frequently observed in pediatric epilepsy. Pellock suggested that approximately 30% of epileptic children may have ADHD, while others have suggested that the prevalence may reach as high as 80% [3]. Many researchers have suggested that the two diseases have a bidirectional link, so the association may not be just coincidental. ADD/ADHD has been shown to increase the risk of epilepsy developing and vice versa [4].
As ADHD is one of the neurodevelopmental disorders, a possible link between ADHD and epilepsy could present. This link could be due to the involvement of inflammation, anxiety, and stress in development of ADHD, which are common findings in the pathogenesis of psychiatric and neurological disorders. Saccaro and colleagues reported in their narrative review an interplay between stress, anxiety, and immune dysregulation in ADHD, which had the implications of a potential disrupted neuroendocrine stress response in ADHD [5]. Theoretically, ADHD and its symptoms (such as emotion dysregulation) may expose a patient to a higher stress risk, which, in turn, reduces the individual’s threshold for experiencing subjective stress. Abnormal stress reactivity ensues, which is associated with inflammatory dysregulation. On the other hand, stress reactivity might be the predisposing factor to ADHD through facilitation of inflammation process. Moreover, inflammation can lead to disruption of brain structures, leading to neurodevelopmental disorders, such as ADHD [5].
Moreover, chronic low-grade inflammation (LGI) has been linked to the prodromal stage of a wide range of chronic illnesses, such as cardiovascular, metabolic, immunologic, neurodegenerative, and psychiatric diseases. The tryptophan (TRP)–kynurenine (KYN) metabolic pathway is a main player of TRP metabolism through which more than 95% of TRP is catabolized. The pathway is activated by acute and chronic immune responses leading to a wide range of illnesses, including neurodegenerative diseases and psychiatric disorders. The immunomodulators are known to facilitate the immune system towards a tolerogenic state, resulting in chronic low-grade inflammation (LGI). Changes in KYNA concentrations have been described in several neurological disorders, including epilepsy [6].
Furthermore, KYN aminotransferases (KATs) catalyze the conversion L-KYN to KYN and its activity is implicated in neurological and cognitive symptoms and the elderly. A higher local KYN concentration is necessary for higher activity of KATs due to its low affinity. A cofactor, pyridoxal 5ʹ-phosphate (PLP), the active form of vitamin B6, and a cosubstrate, α-keto acid, are required for KATs. Genetic dysfunction of the salvage pathway enzymes and drug interactions of PLP or pyridoxal kinase will lead to convulsions and epileptic encephalopathy. A lower level of PLP due to genetic dysfunction of the salvage pathway enzymes or drug interactions of PLP or pyridoxal kinase leads to convulsions and epileptic encephalopathy and is associated with neurological disorders, including epilepsy [7]. Furthermore, TRP–KYN pathway enzymes are known to be activated by the stress hormone cortisol and inflammatory cytokines, and genotypic variants were observed to contribute to inflammation and thus various diseases [6]. All these data are confirming the involvement of inflammation and stress as underlying possible link between ADHD and epilepsy development.
A disputable point is whether co-occurring ADHD and epilepsy symptoms represent a comorbid psychiatric disorder or are the epileptic discharges temporary effects and are they related to antiepileptic drugs (AEDs). This leads to speculation: If some children with epilepsy also have ADHD symptoms, then the clinical patterns of psychiatric comorbid conditions in these children could be very similar to those in children who do not have epilepsy but do have ADHD [8].
Main text
Epidemiology of association of ADHD and epilepsy
ADHD was found to be present in 30–40% of epileptic children in a study conducted by the International League against epilepsy. These children had an average prevalence of ADHD, of 2.5 to 5.5 times that of control children [3, 9]. When epileptic children were categorized by the age at which they first experienced clinical seizures, 15.8% of those who had seizures with the onset prior to age 10 years showed ADHD symptoms, while 8.1% of those newly diagnosed had ADHD symptoms. These findings show that seizures or subclinical epileptiform discharges likely have a role in the onset of attention problems [10]. This also shows that the two conditions have a bidirectional connection, which substantiates the overlap [4].
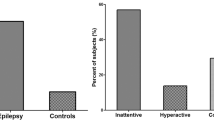
An observational study that enrolled children and adolescents (mean age, 11 years 10 months) with chronic epilepsy was conducted by Dunn and colleagues to characterize epilepsy-associated ADHD in children [11]. Dunn found that 58% of these children and 42% of the adolescents were at risk for having ADHD, while 37% of the children and 25% of the adolescents had clinically overt ADHD. Also, 24% of the children had ADHD that was mostly inattentive, 11% had ADHD that was predominantly mixed, and 2% had ADHD that was predominantly hyperactive–impulsive [2, 11, 12].
Many studies have discovered significant differences between ADHD in epileptic patients and ADHD in children without epilepsy. The inattentive type of ADHD is more prevalent in children with epilepsy than the mixed type, whereas the mixed type is the most prevalent in the general population [13]. In a study of children with severe epilepsy, patients with the mixed type of ADHD experienced earlier start of seizures and more refractory epilepsy than children with the inattentive type [14]. In the general population, boys are 3–7 times more likely than girls to have ADHD [15], whereas in epilepsy patients, both genders are equally represented [16]. Children with epilepsy and ADHD, like children with ADHD without epilepsy, are prone to other neuropsychiatric comorbidities, such as anxiety and oppositional defiant disorder [17].
Theories explaining ADHD and epilepsy comorbidity
Numerous theories have been developed by researchers to explain how and why ADHD and epilepsy coexist, including the following:
-
1.
Independent circumstantial relationships: Because both ADHD and epilepsy are prevalent in children, it is reasonable to expect that some children will have both [18].
-
2.
Dependent circumstantial relationships: ADHD and epilepsy may share the following causative factors:
-
a.
Genetic factors: The two diseases could be linked by shared genetic tendencies [11].
-
b.
Adrenergic system dysfunction: Low urine catecholamine secretion during a cognition test in ADHD children led to the theory that both epilepsy and ADHD may be caused by adrenergic system failures or significant deficiencies [15].
-
c.
Multi-factorial events: When specific genetic and environmental variables interact, they may initiate a chain reaction of transcriptional changes in the brain, changing plasticity, apoptosis, and neurogenesis, and modifying behavior or cognition before seizures begin [13].
-
a.
-
3.
Direct causative relationships: Epileptic seizures may cause or exacerbate ADHD. ADHD is more prevalent in epileptic children than in their normal siblings, suggesting a common neurological impairment [11].
-
4.
Direct effect of antiepileptic drugs: Some antiepileptic drugs exacerbate hyperactivity and impulsivity. Drugs, like phenobarbital and topiramate, are known to cause inattention and hyperactivity. Benzodiazepines also are known to cause inattention and disinhibition [19].
Pathophysiology of relation between ADHD and epilepsy
The question of whether ADHD and epilepsy symptoms are caused by co-occurring psychiatric disorders or by the temporary effects of epileptic discharges or by antiepileptic medicines is critical to consider [8, 20].
Effect of the type of epilepsy on occurrence of ADHD
Generalized epilepsy
Attentional difficulties are more common among patients with generalized epilepsies than those with partial seizures [13, 21].
In children with uncontrollable symptomatic epilepsy, as well as in idiopathic epilepsy, attention problems are common [22]. ADHD-like behavior may also be predisposed by certain epilepsy syndromes [23]. Impaired brain development in children with epilepsy has been linked to the development of ADHD, which in turn has been linked to a range of cognitive and behavioral issues [13]. The presence of symptoms of ADHD during the beginning of epilepsy is consistent with impaired cognitive development [21].
Frontal lobe epilepsy (FLE)
The manifestations of FLE depend on the actual localization of seizure activity within the frontal lobes and can lead to psychiatric, neurologic (both motor and cognitive), and medical (as nausea, vomiting, and vertigo) symptoms, and thus, psychiatrists should be aware of presentations of FLE [24]. The psychiatric features that could be manifested in FLE include impulsivity, disinhibition, and excitement/irritability, similar to those with ADHD without epilepsy. Executive functions, such as inhibition control and set shifting, depend largely on the prefrontal cortex [25]. As ADHD may mimic FLE and ADHD, however, has a high prevalence in FLE likely because of dysregulation of the frontal lobe networks. It is difficult to discern whether ADHD is a misdiagnosis, a comorbid diagnosis, or a diagnosis owing to interictal changes in a developing brain [24].
Action control represents a complex mechanism that includes numerous various processes, such as inhibition, selection, competition, and switching of actions with a composite network named as action inhibition network (AIN) in the prefrontal areas that have a role in coordinating such processes. A recent review by Borgomaneri and colleagues in 2020 explored the neural circuits of action inhibition in humans by using non-invasive brain stimulation, the critical role of prefrontal areas, including the pre-supplementary motor area (pre-SMA) and the inferior frontal gyrus (IFG), in inhibitory control and the neural and behavioral evidence of proactive and reactive action inhibition. They reported the crucial role of pre-SMA and IFG in action inhibition [26].
Successful motor inhibition to threat signals increased activation in limbic regions (amygdala), SMA, in a region in the lateral orbitofrontal cortex, distinct from areas in the IFG typically associated with voluntary inhibition. The ability to voluntarily inhibit unnecessary actions is an important aspect in psychiatric disorders [27]. Deficits in performance in action control have been observed in various psychiatric disorders, which are characterized by serious impulsivity problems that can determine significant impairment or distress. These psychopathological conditions included schizophrenia, bipolar disorder, and ADHD [26, 27]. Some patients with FLE display a pattern of behavioral and cognitive impairment that may be linked to an epilepsy-related deterioration of these networks [28].
Epileptiform frontal lobe (Electroencephalogram) EEG discharges, according to Ben-Ari, disrupt the development of the brain system which leads to attention and hyperactivity disorders at a crucial early stage of brain maturation [29].
Frontal cortex abnormalities and executive dysfunctions were associated with impairments in memory and learning. Evidence from a recent study conducted by Battaglia and colleagues in 2020 on patients with lesion in the ventromedial portion of prefrontal cortex (vmPFC) revealed that this brain structure is involved in the acquisition of emotional conditioning (learning) and authors provided evidence that naturally occurring bilateral lesion centered on the vmPFC compromises the generation of fear conditioning as measured via skin conductance responses. From a clinical perspective, part of the cognitive and behavioral difficulties observed in patients with a vmPFC lesion may result from such an acquisition deficit [30].
Accordingly, in a recent theoretical review conducted by Battaglia and colleagues in 2021 focused on neurobiology of fear conditioning, the anterior/posterior subregions of the vmPFC were detected to have distinct contributions of in the processing of safety threat information. Authors provided evidence for the fundamental role of the vmPFC in the evaluation and representation of stimulus–outcome’s value needed to produce sustained physiological responses [31]. These findings highlighted prefrontal cortex’s key role in the acquisition of new learning, and how its disrupted function may contribute to irregular behavioral responses and therefore to consequent development of neuropsychiatric disorders characterized by altered value attribution, reward anticipation, awareness and control of action, or fear extinction. This was highlighted by many studies that monitor the effect of frontal lobe damage [32,33,34].
Co-occurring pediatric ADHD with epilepsy and frontal lobe lesions was also found to be common in tuberous sclerosis patients [35]. The side of the seizure focus in people with epilepsy may also play a role in executive dysfunction; in particular, a left frontal focus is more likely to impair the inhibitory processes than right frontal foci [36].
Another recent evidence for the possibility of shared underlying pathological mechanisms of altered control inhibition in patient with ADHD and FLE was stemmed from report by Battaglia in 2022. The author demonstrated the importance of the human prefrontal cortex (PFC) as a crucial component of the neural network governing fear conditioning. On one hand, the dorsomedial PFC (dmPFC) activation enables the long-term storage and retrieval of old memories. On the other hand, the ventromedial prefrontal cortex (vmPFC) forms significant reciprocal connections with the amygdala and other subcortical structures as well as the lateral cortex. Furthermore, the lateral PFC (LPFC) is responsible for response selection and control. So, human PFC modulates the activity of the amygdala and hippocampus after fear conditioning (extinction learning). Fear extinction appears to be mediated by the dorsolateral PFC (dlPFC) as a result of its ability to shift attention from the stimulus to the context, as well as its role as a site of explicit short-term memory processes in humans [37]. So, frontal lobe dysfunction will impair fear conditioning and extinction, which may be a characteristic of FLE and ADHD. In FLE, fearful emotional expression mixed with non-stereotyped gestural motor behavior generating a defensive or offensive reaction was common hallmarks of FLE coming from vmPFC. This fearful ictal behavior was primarily caused by aberrant activation of proven prefrontal circuits that are involved in the formation of emotional responses, like fear [38]. In ADHD, Spencer and colleagues showed in 2017 that people with ADHD have problems with emotion management due to aberrant activation in brain areas that mediate fear extinction [39].
Childhood absence epilepsy (CAE)
Nearly 10–15% of all childhood-onset seizures are determined to be absence seizures. Some studies have linked absence seizures and attentional disorders to similar cortical–subcortical neural networks [40]. Visual sustained attention, verbal and non-verbal attention, and memory are all recognized to be problems in children with CAE [36] and are at times, successfully treated with antiepileptic drugs. Children with normal intelligence are also not as severely affected. In children with CAE, ADHD is the most frequent psychiatric diagnosis, with the inattentive subtype being the most common [11].
In 61% of children with CAE, a psychiatric comorbidity, such as ADHD, was detected, with just a small proportion of these children obtaining adequate therapy for this comorbidity [23].
Williams and colleagues conducted a clever study in which they aimed to discover behavioral characteristics that could be linked to either absence seizures or ADHD: Observers filled out a 40-item questionnaire about their own behavior. “Glassy eyes” and “change of breathing” were the strongest indicators of an absence seizure. Patients with absence seizures were less likely to indicate “fidgeting,” which is a strong predictor of ADHD [40].
Rolandic epilepsy
Benign epilepsy with centrotemporal spikes (BECTS) or Rolandic epilepsy is the most common form of idiopathic focal epilepsy in childhood and typically begins in the first decade of life between 7 and 10 years of age. It is characterized by simple focal motor seizures that consist of brief movements localized in the orofacial region, often preceded by sensory phenomena. Seizures occur mainly during sleep or upon awakening with or without a secondary generalization [41]. Regarding BECTS, the prevalence of ADHD has been reported to be between 30 and 50% [42, 43]. Both BECTS and ADHD share some clinical features: (i) they both start during childhood, and they seem to be limited by puberty in some cases; (ii) the prevalence in boys is higher than that in girls; and (iii) both diseases show a deficit in executive function, impulsivity, and externalizing concerns [44].
Rolandic spikes are more prevalent in children with ADHD than in the general population of children, although no clear explanation has been determined yet. According to a neuropsychological study, Rolandic epileptiform discharges exacerbate the symptoms of ADHD in children and increase their impulsivity [45]. Children with centrotemporal spikes are more vulnerable to visual distracters than healthy children or children with idiopathic generalized epilepsy [46].
Miriam and colleagues, in their systematic review [47], concluded that despite BECTS being considered to have a relative benign course, many studies have documented cognitive and/or behavioral problems in patients diagnosed with this type of epilepsy. In particular, children affected by Rolandic epilepsy should receive a complete neuropsychological evaluation at seizure onset considering the high rate of comorbidity with ADHD.
Sleep-related epilepsies
About a third of all epileptic patients experience both focal and generalized spikes while they sleep, which is known as sleep-related epilepsies. As assessed by the maintenance of wakefulness test, nocturnal seizures lower sleep efficiency, lengthen the time between first REM cycles, and increase sleepiness [48]. This has negative consequences, such as poor sleeping habits and serious psychiatric issues [49]. Sleep fragmentation/disruption as a result of sleep-related epileptic illness progression could be a cause of both inattention and hyperactivity.
Local slow wave activity could be disrupted by long-term focal epileptic activity during sleep, such as that seen in electrical status epilepticus during slow wave sleep. This would have an impact on neural processes as well as possible local plastic alterations in learning and other cognitive functions [50]. Attentional deficits and hyperactivity have been linked to Electrical status epilepticus in sleep (ESES) [51].
Nocturnal FLE, which is defined by seizures that occur during sleep and originate in the orbitofrontal or mesial frontal areas, has been linked to symptoms of ADHD [52]. Frontal EEG discharges decrease executive skills, and the progressive deterioration of sleep-related cognitive abilities all have a topographic link [51]. This proved by evidence from previous studies. The first was an electrophysiological study which highlighted how altered EEG activity recorded from medio-frontal areas reflects impaired frontal cortex functions, that impact on cognitive abilities (like prediction error learning). These findings suggest a critical role of timing and salience information in prediction error signaling [53]. Another study explored the dysfunctions in frontal lobe and its contribution to cross-modal prediction error. They reported that aberrant frontal theta-band oscillations reflect cross-modal prediction error processing deficits in schizophrenia [54].
Neuropsychological studies
Before initiating treatment for a child with ADHD symptoms, it is important to conduct a thorough evaluation. Seizures that go unreported can be detected using EEG monitoring, especially if inattention is a key sign of the condition. It is possible that epileptiform EEG discharges affect brain function and attention in subtle way [55].
EEG abnormalities, such as interictal epileptiform discharges (IEDs) or a specific location of IED foci, have been reported in several studies of children with ADHD [56, 57]. Patients with ADHD who have epileptiform discharges and patients with ADHD who have a normal EEG have been compared to determine how epileptiform discharges affect ADHD [58]. According to Socanski and colleagues 44.4% of children with ADHD and EEG abnormalities fulfilled criteria for the inattentive ADHD subtype [59]. Moreover, this EEG paroxysm abnormalities and ADHD scores were observed to respond to valproate therapy [60]. This confirms the shared underlying pathology.
To measure cognitive, behavioral, socioeconomic level, and quality of life, research was completed in five groups of children: Those with ADHD and EEG changes, those with ADHD and without EEG changes, those with epilepsy (without ADHD), those with ADHD and epilepsy, and the control group, who had neither ADHD nor epilepsy nor any EEG changes. In terms of psychiatric comorbidity, children with ADHD and EEG changes were found to be similar to children with epilepsy (but no ADHD) in terms of anxiety, depression, somatic problems, oppositional defiance problems, and conduct problems [61]. Furthermore, ADHD combined with epilepsy was associated with low performance IQ, a low socioeconomic level, and a low quality of life [62].
Video polysomnography can also aid in the detection and diagnosis of all forms of suspected sleep problems, which are often linked to epilepsy and ADHD. In individuals who are experiencing cognitive decline, nocturnal sleep-EEG investigations are indicated as they may lead to therapies otherwise not being considered [56].
Attention difficulties in epilepsy
In children with benign childhood epilepsy with centro-temporal spikes and complex partial seizures (CPSs), a review of neuropsychological studies of attention problems in epilepsy discovered that sustained attention is more often impaired than divided or selective attention, and complex attention is more affected than simple attention [63].
CPS and ADHD children performed worse in terms of attention than children with ADHD who did not have epilepsy [64]. Simple inattention has been reported in children who have recently developed epilepsy and are not receiving antiepileptic drugs (AEDs) [65].
In epileptic patients, sustained attention is shown to be affected more in children with generalized seizures, than in children with focal seizures [66].
In a population-based examination of cognitive capacity in children with epilepsy compared to controls, more than half of the patients showed a relative impairment in at least one of the four working memory subtests administered [67].
Regardless of seizure type or location, most investigations have found no substantial deficits in attention or executive functioning in children with frontal lobe seizures [68].
The onset of seizures at an age younger than 5 years was linked to a slower motor speed. Furthermore, children with epilepsy scored considerably worse on complicated attention tasks than controls, with more than half of the epilepsy group scoring at least one standard deviation below the normative sample mean for these tasks [69].
The surge in ADHD prevalence in epileptic children has been attributed to a number of factors including AEDs, prolonged seizures, and/or non-convulsive epileptiform discharges which all have an impact on cognition, memory, and processing speed [20, 70, 71].
In line with previous evidence, another report by Battaglia and colleagues in 2022 demonstrated how alterations in social cognition and social functioning in neurologic and psychiatric patients are assumed to rely on altered activity within the amygdala and superior temporal sulcus (STS). In their report, authors used neuroimaging findings and explored the neural bases of gaze cueing and perception of gaze direction, as well as how contextual variables interact with the gaze shift of attention. According to neuroimaging research, fearful faces with an averted gaze affected amygdala and STS activity more than fearful faces with a direct gaze [72].
Assessment and diagnosis of ADHD with epilepsy comorbidity in children
History taking
Before beginning treatment, the patient’s history should be thoroughly reviewed both by extensive interview with the parent or guardian and by review of records from prior healthcare providers. ADHD is often diagnosed as an “exclusion” diagnosis based on the patient’s medical history. Typical ADHD symptoms manifest themselves in a variety of settings, including with the family and at school [51].
Epilepsy can also be diagnosed based on a patient’s medical history because clinicians rarely see seizures, and there are typically no physical exam findings to support the diagnosis. Epilepsy symptoms may be more noticeable in peri-ictal temporal durations, especially if prodromes and postictal phases are substantially extended across several hours or days. The key differences are demonstrated in Table 1 [40].
Neurophysiological studies
Neurophysiological studies can measure cognitive capacity and detect learning impairments and may consist of task performance, EEG evaluations, and sleep studies [40, 51].
Task performance
Some researchers have looked for specific symptoms or task performance tests that could help them distinguish between the two conditions. Based on performance on timed cognitive decision-making tasks and reaction times, prospective research could help separate epilepsy from ADHD [40].
On simple response time tasks, ADHD patients were shown to be characterized by impulsive errors and disinhibition, whereas epilepsy patients showed a generalized slower performance on challenging decision-making tasks. This shows that ADHD patients suffer from inconsistency in cognitive processing, whereas epilepsy patients suffer from a more widespread slowing of function [40].
Electroencephalography (EEG) monitoring
-
1.
Inattention is a common indication of undetected seizures; therefore, EEG monitoring can also help detect them. It also aids in the detection of epileptogenic activity.
-
2.
Video polysomnography can detect and diagnose all forms of suspected sleep abnormalities, including those linked to epilepsy and ADHD.
-
3.
Overnight sleep-EEG studies are recommended in case of cognitive decline [51].
Rating scales
Rating scales may be non-specific, particularly when diagnostic confounding variables associated with epilepsy are present and are best utilized as a screening tool to aid in the clinical evaluation of psychiatric comorbidity [40].
Management of ADHD with epilepsy comorbidity
Aim of treatment
According to Parise, the treatment aims of the treatment to the following:
-
1.
Determine whether lowering AED polytherapy or changing to an AED with fewer psychological and cognitive effects is more appropriate than an ADHD medication in the context of worsening ADHD symptoms.
-
2.
Investigate methods for improving seizure management to alleviate sleep issues and ADHD symptoms.
-
3.
Conduct periodic reassessments to determine whether ADHD medication is either no longer necessary or can undergo a dosage reduction.
-
4.
Several AEDs have been linked to increased behavioral problems, which could exacerbate underlying ADHD symptoms, while psychiatric medicines used to treat ADHD can also lower the seizure threshold but seldom do so [51].
Choosing the best antiepileptic drug with ADHD comorbidity
Valproate is used as a mood stabilizer. It has been demonstrated to be useful in lowering impulsivity in ADHD and reducing concomitant oppositional defiant disorder [73, 74].
Topiramate has the potential to cause significant behavioral changes in pediatric epilepsy with ADHD [75], it may be used with caution in these children.
Levetiracetam might be used to treat ADHD with nocturnal focal epileptic discharges [76]. Although 38% of children will experience non-psychotic behavioral changes including aggression and agitation.
AEDs, like carbamazepine or oxcarbazepine, may be beneficial for some patients with partial epilepsy because of their positive effects on ADHD-related behavioral and mood problems [2, 12, 22] and improvement in attention [40]. Previous research found that the long-term use of both clonazepam and carbamazepine might increase the incidence of behavioral problems especially increased severity of anxiety and depression and decreased performance IQ compared with either clonazepam or carbamazepine alone. Moreover, patients with long-term use of carbamazepine treatment might have more personality changes and lowered verbal IQ than others [77].
Lamotrigine is very effective at lowering epileptic discharges and improving behavior and attention in epilepsy patients [22].
Recommendations for ADHD treatment in epilepsy
The most effective drugs for treating ADHD symptoms are methylphenidate and atomoxetine, which both considerably increase activity in important cortical and subcortical areas involved in attention and executive function [78, 79]. So, in patients with epilepsy, the risk of seizures worsening during ADHD treatment should be considered although it rarely occurs [40].
-
A.
Methylphenidate (MPH): Patients treated with MPH have had case reports warn about the development of new-onset seizures; however, MPH has been shown in controlled trials to alleviate ADHD symptoms without exacerbating seizures in patients with epilepsy and ADHD. Seizure exacerbation occurs in only a small percentage of people with active epilepsy [75]. A year of MPH treatment could help restore the neural correlates of attention to their normal state [40].
-
B.
Atomoxetine: During the first 2 weeks of treatment with atomoxetine, only one out of 17 individuals with ADHD and epilepsy saw an increase in the incidence of epileptic seizures [80]. Seizures were not substantially different between methylphenidate, atomoxetine, and placebo, according to a meta-analysis [81]. If seizures occur less frequently than once a month and there is at least some impairment from ADHD, either MPH or atomoxetine should be started, with MPH having the best evidence for effectiveness in this group [51].
-
C.
Alternative medications: If MPH and atomoxetine fail, a trial with guanfacine or clonidine, depending on the severity of the ADHD, may be explored [82]. Modafinil and tricyclic antidepressants should be used with caution in children who have significant cognitive impairment [75] largely because of undesirable side effects.
-
D.
Sleep difficulties should be detected and addressed. Melatonin therapy may help with epilepsy-related sleep disorders because it improves sleep organization [83], and this helps improve executive skills in both REM and NREM-related disorders [84]. Lack of sleep is also a major contributor to ADHD-related symptoms. So, restoring sleep quality stands to improve the child’s overall function.
Drug interactions
AEDs and ADHD medications should be used with caution in this patient population because of the potential for negative pharmacokinetic interactions [51] and untoward side effects of the medications.
Atomoxetine has been shown to have no interactions with any AEDs [85], while MPH can raise serum phenytoin concentrations [86]. Additionally, concurrent administration of carbamazepine may reduce MPH serum concentrations, resulting in reduced efficacy [51].
Limitation and future recommendations
The limitation for the current research includes limited number of investigated studies. We recommend conducting more studies with large sample size to help in determining the possible link between ADHD and epilepsy which could help in proper treatment of patients with such comorbidity which will have good impact on family and community.
Conclusions
ADHD and epilepsy are frequent comorbid conditions and are associated with important psychosocial and academic consequences. Despite the evidence of epidemiological connection between them is well established, the mechanisms of the comorbidity remain poorly understood [54]. The two diseases have a bidirectional link, so the association may not be coincidental. A disputable point is whether co-occurring ADHD and epilepsy symptoms represent a comorbid psychiatric disorder or are the epileptic discharges temporary effects, and are they related to antiepileptic drugs (AEDs). It is recommended to follow up with children with epilepsy or ADHD as they may develop comorbidity after a while.
Availability of data and materials
Data sharing is not applicable to this article as no data sets were generated or analyzed during the current study.
Abbreviations
- ADHD:
-
Attention Deficit Hyperactivity Disorder
- AEDs:
-
Antiepileptic drugs
- BECTS:
-
Benign childhood epilepsy with centrotemporal spikes
- ESES:
-
Electrical status epilepticus in sleep
- FLE:
-
Frontal lobe epilepsy
- IEDs:
-
Interictal epileptiform discharges
- CAE:
-
Childhood Absence Epilepsy
- CPSs:
-
Complex partial seizures
- MPH:
-
Methylphenidate
- EEG:
-
Electroencephalography
- REM:
-
Rapid eye movement
References
Denckla MB. ADHD: topic update. Brain Dev. 2003;25(6):383–9.
Pellock JM. Defining the problem: psychiatric and behavioral comorbidity in children and adolescents with epilepsy. Epilepsy Behav. 2004;5:3–9.
Besag F, Gobbi G, Caplan R, Sillanpää M, Aldenkamp A, Dunn DW. Psychiatric and behavioural disorders in children with epilepsy (ILAE Task Force Report): epilepsy and ADHD. Epileptic Disord. 2016;18(s1):S8–15.
Chou IC, Chang YT, Chin ZN, Muo CH, Sung FC, Kuo HT, et al. Correlation between epilepsy and attention deficit hyperactivity disorder: a population-based cohort study. PLoS ONE. 2013;8(3):e57926.
Saccaro LF, Schilliger Z, Perroud N, Piguet C. Inflammation, anxiety, and stress in attention-deficit/hyperactivity disorder. Biomedicines. 2021;9(10):1313.
Tanaka M, Tóth F, Polyák H, Szabó Á, Mándi Y, Vécsei L. Immune influencers in action: metabolites and enzymes of the tryptophan–kynurenine metabolic pathway. Biomedicines. 2021;9(7):734.
Tanaka M, Toldi J, Vécsei L. Exploring the etiological links behind neurodegenerative diseases: inflammatory cytokines and bioactive kynurenines. Int J Mol Sci. 2020;21(7):2431.
Gonzalez-Heydrich J, Dodds A, Whitney J, MacMillan C, Waber D, Faraone SV, et al. Psychiatric disorders and behavioral characteristics of pediatric patients with both epilepsy and attention-deficit hyperactivity disorder. Epilepsy Behav. 2007;10(3):384–8.
Auvin S, Wirrell E, Donald KA, Berl M, Hartmann H, Valente KD, et al. Systematic review of the screening, diagnosis, and management of ADHD in children with epilepsy. Consensus paper of the Task Force on Comorbidities of the ILAE Pediatric Commission. Epilepsia. 2018;59(10):1867–80.
Dunn DW, Austin JK, Harezlak J. ADHD and epilepsy in childhood. Dev Med Child Neurol. 2003;45(1):50–4.
Dunn DWAJ, Harezlak J, et al. ADHD and epilepsy in childhood. Dev Med Child Neurol. 2003;45:50–4.
Pellock JM. Understanding co-morbidities affecting children with epilepsy. Neurology. 2004;62(5 suppl 2):S17–23.
Hermann B, Jones J, Dabbs K, Allen CA, Sheth R, Fine J, et al. The frequency, complications and aetiology of ADHD in new onset paediatric epilepsy. Brain. 2007;130(12):3135–48.
Sherman EM, Slick DJ, Connolly MB, Eyrl KL. ADHD, neurological correlates and health-related quality of life in severe pediatric epilepsy. Epilepsia. 2007;48(6):1083–91.
Hesdorffer DC, Ludvigsson P, Olafsson E, et al. ADHD as a risk factor for incident unprovoked seizures and epilepsy in children. Arch Gen Psychiatry. 2004;61:731–6.
Barkley RA. Attention-deficit/hyperactivity disorder. New York: Guilford Publications; 2006.
Parisi P, Moavero R, Verrotti A, Curatolo P. Attention deficit hyperactivity disorder in children with epilepsy. Brain Dev. 2009;32:10–6.
Kaufmann R, Goldberg-Stern H, Shuper A. Attention-deficit disorders and epilepsy in childhood: incidence, causative relations and treatment possibilities. J Child Neurol. 2009;24(6):727–33.
Loring DWMK. Cognitive side effects of antiepileptic drugs in children. Neurology. 2004;62:872–7.
Holtmann M, Becker K, Kentner-Figura B, Schmidt MH. Increased frequency of rolandic spikes in ADHD children. Epilepsia. 2003;44(9):1241–4.
Hermann BP, Jones JE, Sheth R, Koehn M, Becker T, Fine J, et al. Growing up with epilepsy: a two-year investigation of cognitive development in children with new onset epilepsy. Epilepsia. 2008;49(11):1847–58.
Schubert R. Attention deficit disorder and epilepsy. Pediatr Neurol. 2005;32(1):1–10.
Caplan R, Siddarth P, Stahl L, Lanphier E, Vona P, Gurbani S, et al. Childhood absence epilepsy: behavioral, cognitive, and linguistic comorbidities. Epilepsia. 2008;49(11):1838–46.
Gold JA, Sher Y, Maldonado JR. Frontal lobe epilepsy: a primer for psychiatrists and a systematic review of psychiatric manifestations. Psychosomatics. 2016;57(5):445–64.
Pasini A, Paloscia C, Alessandrelli R, Porfirio MC, Curatolo P. Attention and executive functions profile in drug naive ADHD subtypes. Brain Dev. 2007;29(7):400–8.
Borgomaneri S, Serio G, Battaglia S. Please, don’t do it! Fifteen years of progress of non-invasive brain stimulation in action inhibition. Cortex. 2020;132:404–22.
Battaglia S, Serio G, Scarpazza C, D’Ausilio A, Borgomaneri S. Frozen in (e)motion: how reactive motor inhibition is influenced by the emotional content of stimuli in healthy and psychiatric populations. Behav Res Ther. 2021;146:103963.
Holmes GL, Lenck-Santini P-P. Role of interictal epileptiform abnormalities in cognitive impairment. Epilepsy Behav. 2006;8(3):504–15.
Ben-Ari Y, Holmes GL. Effects of seizures on developmental processes in the immature brain. Lancet Neurol. 2006;5(12):1055–63.
Battaglia S, Garofalo S, di Pellegrino G, Starita F. Revaluing the role of vmPFC in the acquisition of pavlovian threat conditioning in humans. J Neurosci. 2020;40(44):8491–500.
Battaglia S, Harrison BJ, Fullana MA. Does the human ventromedial prefrontal cortex support fear learning, fear extinction or both? A commentary on subregional contributions. Mol Psychiatry. 2021. https://doi.org/10.1038/s41380-021-01326-4.
Schall JD, Stuphorn V, Brown JW. Monitoring and control of action by the frontal lobes. Neuron. 2002;36(2):309–22. https://doi.org/10.1016/s0896-6273(02)00964-9.
Dimitrov M, Nakic M, Elpern-Waxman J, Granetz J, O’Grady J, Phipps M, et al. Inhibitory attentional control in patients with frontal lobe damage. Brain Cogn. 2003;52(2):258–70.
Wolpe N, Moore JW, Rae CL, Rittman T, Altena E, Haggard P, et al. The medial frontal-prefrontal network for altered awareness and control of action in corticobasal syndrome. Brain. 2014;137(Pt 1):208–20.
D’Agati E, Moavero R, Cerminara C, Curatolo P. Attention-deficit hyperactivity disorder (ADHD) and tuberous sclerosis complex. J Child Neurol. 2009;24(10):1282–7.
McDonald CR, Delis DC, Norman MA, Wetter SR, Tecoma ES, Iragui VJ. Response inhibition and set shifting in patients with frontal lobe epilepsy or temporal lobe epilepsy. Epilepsy Behav. 2005;7(3):438–46.
Battaglia S. Neurobiological advances of learned fear in humans. Adv Clin Exp Med. 2022;31:217–21.
Bonini F, McGonigal A, Trébuchon A, Gavaret M, Bartolomei F, Giusiano B, et al. Frontal lobe seizures: from clinical semiology to localization. Epilepsia. 2014;55(2):264–77.
Spencer AE, Marin MF, Milad MR, Spencer TJ, Bogucki OE, Pope AL, et al. Abnormal fear circuitry in Attention Deficit Hyperactivity Disorder: a controlled magnetic resonance imaging study. Psychiatry Res Neuroimaging. 2017;262:55–62.
Salpekar JA, Mishra G. Key issues in addressing the comorbidity of attention deficit hyperactivity disorder and pediatric epilepsy. Epilepsy Behav. 2014;37:310–5.
Guerrini R, Pellacani S. Benign childhood focal epilepsies. Epilepsia. 2012;53(Suppl 4):9–18.
Tovia E, Goldberg-Stern H, Ben Zeev B, Heyman E, Watemberg N, Fattal-Valevski A, et al. The prevalence of atypical presentations and comorbidities of benign childhood epilepsy with centrotemporal spikes. Epilepsia. 2011;52(8):1483–8.
Holtmann M, Matei A, Hellmann U, Becker K, Poustka F, Schmidt MH. Rolandic spikes increase impulsivity in ADHD—a neuropsychological pilot study. Brain Dev. 2006;28(10):633–40.
Xiao F, Li L, An D, Lei D, Tang Y, Yang T, et al. Altered attention networks in benign childhood epilepsy with centrotemporal spikes (BECTS): a resting-state fMRI study. Epilepsy Behav. 2015;45:234–41.
Holtmann M, Becker K, Kentner-Figura B, Schmidt MH. Response: increased frequency of rolandic spikes in ADHD children. Epilepsia. 2004;45(5):565–6.
Deltour L, Barathon M, Quaglino V, Vernier M-P, Despretz P, Boucart M, et al. Children with benign epilepsy with centrotemporal spikes (BECTS) show impaired attentional control: evidence from an attentional capture paradigm. Epileptic Disord. 2007;9(1):32–8.
Aricò M, Arigliani E, Giannotti F, Romani M. ADHD and ADHD-related neural networks in benign epilepsy with centrotemporal spikes: a systematic review. Epilepsy Behav. 2020;112:107448.
Foldvary-Schaefer N, Grigg-Damberger M. Sleep and epilepsy: what we know, do not know, and need to know. J Clin Neurophysiol. 2006;23(1):4–20.
Batista BHB, Nunes ML. Evaluation of sleep habits in children with epilepsy. Epilepsy Behav. 2007;11(1):60–4.
Nickels K, Wirrell E. Electrical status epilepticus in sleep. In: Seminars in pediatric neurology. Elsevier; 2008. p. 50–60.
Parisi P, Moavero R, Verrotti A, Curatolo P. Attention deficit hyperactivity disorder in children with epilepsy. Brain Dev. 2010;32(1):10–6.
Ryvlin P, Rheims S, Risse G. Nocturnal frontal lobe epilepsy. Epilepsia. 2006;47:83–6.
Garofalo S, Timmermann C, Battaglia S, Maier ME, di Pellegrino G. Mediofrontal negativity signals unexpected timing of salient outcomes. J Cogn Neurosci. 2017;29(4):718–27.
Roa Romero Y, Keil J, Balz J, Gallinat J, Senkowski D. Reduced frontal theta oscillations indicate altered crossmodal prediction error processing in schizophrenia. J Neurophysiol. 2016;116(3):1396–407.
Aldenkamp AAJ. The relative influence of epileptic EEG discharges, short nonconvulsive seizures and type of epilepsy on cognitive function. Epilepsia. 2004;45:54–63.
Silvestri R, Gagliano A, Calarese T, Arico I, Cedro C, Condurso R, et al. Ictal and interictal EEG abnormalities in ADHD children recorded over night by video-polysomnography. Epilepsy Res. 2007;75(2–3):130–7.
Fonseca LC, Tedrus GMA, Moraes Cd, Machado AdV, Almeida MPd, Oliveira DOFd. Epileptiform abnormalities and quantitative EEG in children with attention-deficit/hyperactivity disorder. Arq Neuropsiquiatr. 2008;66(3):462–7.
Lee EH, Choi YS, Yoon HS, Bahn GH. Clinical impact of epileptiform discharge in children with attention-deficit/hyperactivity disorder (ADHD). J Child Neurol. 2015;31(5):584–8.
Socanski D, Herigstad A, Thomsen PH, Dag A, Larsen TK. Epileptiform abnormalities in children diagnosed with attention deficit/hyperactivity disorder. Epilepsy Behav. 2010;19(3):483–6.
Kanemura H, Sano F, Tando T, Hosaka H, Sugita K, Aihara M. EEG improvements with antiepileptic drug treatment can show a high correlation with behavioral recovery in children with ADHD. Epilepsy Behav. 2013;27(3):443–8.
Ahmed GK, Darwish AM, Khalifa H, Khashbah MA. Evaluation of psychiatric comorbidity in attention-deficit hyperactivity disorder with epilepsy: a case-control study. Epilepsy Res. 2021;169:106505.
Ahmed GK, Darwish AM, Khalifa H, Khashbah MA. Comparison of cognitive function, socioeconomic level, and the health-related quality of life between epileptic patients with attention deficit hyperactivity disorder and without. Middle East Curr. 2020;27(1):45.
Yoong M. Quantifying the deficit—imaging neurobehavioural impairment in childhood epilepsy. Quant Imaging Med Surg. 2015;5(2):225.
Sánchez-Carpintero R, Neville BG. Attentional ability in children with epilepsy. Epilepsia. 2003;44(10):1340–9.
Triplett RL, Asato MR. Brief cognitive and behavioral screening in children with new-onset epilepsy: a pilot feasibility trial. Pediatr Neurol. 2015;52(1):49–55.
Kang S-H, Yum M-S, Kim E-H, Kim H-W, Ko T-S. Cognitive function in childhood epilepsy: importance of attention deficit hyperactivity disorder. J Clin Neurol. 2015;11(1):20–5.
Berl MM, Terwilliger V, Scheller A, Sepeta L, Walkowiak J, Gaillard WD. Speed and complexity characterize attention problems in children with localization-related epilepsy. Epilepsia. 2015;56(6):833–40.
Almane D, Jones JE, Jackson DC, Seidenberg M, Hermann BP. The social competence and behavioral problem substrate of new-and recent-onset childhood epilepsy. Epilepsy Behav. 2014;31:91–6.
Williams AE, Giust JM, Kronenberger WG, Dunn DW. Epilepsy and attention-deficit hyperactivity disorder: links, risks, and challenges. Neuropsychiatr Dis Treat. 2016;12:287.
Bourgeois BF. Antiepileptic drugs, learning, and behavior in childhood epilepsy. Epilepsia. 1998;39(9):913–21.
Gilby KL. A new rat model for vulnerability to epilepsy and autism spectrum disorders. Epilepsia. 2008;49:108–10.
Battaglia S, Fabius JH, Moravkova K, Fracasso A, Borgomaneri S. The neurobiological correlates of gaze perception in healthy individuals and neurologic patients. Biomedicines. 2022;10(3):627.
Golden AS, Haut SR, Moshé SL. Nonepileptic uses of antiepileptic drugs in children and adolescents. Pediatr Neurol. 2006;34(6):421–32.
Saxena K, Howe M, Simeonova D, Steiner H, Chang K. Divalproex sodium reduces overall aggression in youth at high risk for bipolar disorder. J Child Adolesc Psychopharmacol. 2006;16(3):252–9.
Torres AR, Whitney J, Gonzalez-Heydrich J. Attention-deficit/hyperactivity disorder in pediatric patients with epilepsy: review of pharmacological treatment. Epilepsy Behav. 2008;12(2):217–33.
Gomer B, Wagner K, Frings L, Saar J, Carius A, Härle M, et al. The influence of antiepileptic drugs on cognition: a comparison of levetiracetam with topiramate. Epilepsy Behav. 2007;10(3):486–94.
Ahmed GK, Elbeh K, Elserogy Y, Mostafa S. Effect of long-term administration of clonazepam, carbamazepine, and valproate on cognitive, psychological, and personality changes in adult epilepsy: a case–control study. Middle East Curr. 2021;28(1):81.
Pliszka SR. The neuropsychopharmacology of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2005;57(11):1385–90.
Bush G, Spencer TJ, Holmes J, Shin LM, Valera EM, Seidman LJ, et al. Functional magnetic resonance imaging of methylphenidate and placebo in attention-deficit/hyperactivity disorder during the multi-source interference task. Arch Gen Psychiatry. 2008;65(1):102–14.
Hernandez A. Efficacy of atomoxetine treatment in children with ADHD and epilepsy. Epilepsia. 2005;46(6):718.
Wernicke JF, Holdridge KC, Jin L, Edison T, Zhang S, Bangs ME, et al. Seizure risk in patients with attention-deficit-hyperactivity disorder treated with atomoxetine. Dev Med Child Neurol. 2007;49(7):498–502.
Palumbo DR, Sallee FR, Pelham WE Jr, Bukstein OG, Daviss WB, McDERMOTT MP, et al. Clonidine for attention-deficit/hyperactivity disorder: I. Efficacy and tolerability outcomes. J Am Acad Child Adolesc Psychiatry. 2008;47(2):180–8.
Wasdell MB, Jan JE, Bomben MM, Freeman RD, Rietveld WJ, Tai J, et al. A randomized, placebo-controlled trial of controlled release melatonin treatment of delayed sleep phase syndrome and impaired sleep maintenance in children with neurodevelopmental disabilities. J Pineal Res. 2008;44(1):57–64.
Karpinski AC, Scullin MH, Montgomery-Downs HE. Risk for sleep-disordered breathing and executive function in preschoolers. Sleep Med. 2008;9(4):418–24.
Kanner AM, Gidal BE. Pharmacodynamic and pharmacokinetic interactions of psychotropic drugs with antiepileptic drugs. Int Rev Neurobiol. 2008;83:397–416.
Markowitz JS, Morrison SD, DeVane CL. Drug interactions with psychostimulants. Int Clin Psychopharmacol. 1999;14(1):1–18.
Acknowledgements
None.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
GA and NH searched, collected papers, and were the contributors in writing the manuscript. AA and HK revised review and manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
No ethical approval was obtained or required for the purposes of this review.
Consent for publication
Not applicable.
Competing interests
The authors declare no conflicts of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ahmed, G.K., Darwish, A.M., Khalifa, H. et al. Relationship between Attention Deficit Hyperactivity Disorder and epilepsy: a literature review. Egypt J Neurol Psychiatry Neurosurg 58, 52 (2022). https://doi.org/10.1186/s41983-022-00482-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41983-022-00482-w