Abstract
Background
Hepatitis C is a liver infection caused by the hepatitis C virus (HCV). It can cause both acute and chronic hepatitis. Sofosbuvir (sofo) is a nucleotide analog inhibitor of HCV NS5B polymerase used to treat chronic hepatitis C infection as a component of a combination of antiviral treatment regimen. Many side effects of sofo were reported in different mammalian organs including kidney. Moringa oleifera (MO) is one of the medicinal plants which have many pharmacological activities and nutritional applications due to its rich phytonutrients content. This study aimed to investigate the possible ameliorative effect of MO seed oil against nephrotoxicity induced by sofo in adult male albino rats. The experimental animals were divided equally into four groups. Group I: animals were served as control. Group II: animals were orally given MO oil (2 ml/kg/day). Group III: animals were orally administered with sofo (36 mg/kg/day). Group IV: animals were orally given sofo then after 2 h they were given MO oil (with the same previous doses). All doses were daily given to the animals for eight weeks. At the end of the experiments, animals were sacrificed and sera were collected to determine urea, creatinine and malondialdehyde levels and catalase activity. Kidneys were removed out and prepared for both the histological and immunohistochemical studies.
Results
Sofo-treated animals showed many pathological changes; damaged glomeruli and degenerated renal tubules with vacuolated lining epithelial cells contain pyknotic nuclei. In addition, leukocytic infiltration, congested blood vessels and hemorrhage were seen. Caspase-3 and PCNA were expressed in a large number of cells in the same group. Moreover, a significant increase in urea, creatinine and malondialdehyde levels was recorded as well as a significant decrease in catalase activity. Co-treatment of MO oil with sofo effectively counteracted the observed adverse effects. It attenuated the histological picture of the kidney, significantly ameliorated urea, creatinine and malondialdehyde levels and catalase activity and restored the normal expressions of caspase-3 and PCNA.
Conclusions
Moringa oleifera oil can ameliorate nephrotoxicity induced by sofo via its antioxidant, anti-inflammatory and anti-apoptotic properties.
Similar content being viewed by others
Background
Hepatitis C is a liver disease caused by hepatitis C virus (HCV) which can cause both acute and chronic hepatitis, ranging in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Globally, 58 million of people have chronic HCV infection, approximately 290,000 people died, mostly from cirrhosis and hepatocellular carcinoma (WHO, 2021).
Therapies for HCV treatment have focused on the development of direct acting antiviral (DAA) drugs which target specific HCV enzymes (non-structure protease and polymerase inhibitors) (Pockros, 2010). Sofosbuvir (sofo) is formerly known as GS-7977 or PSI-7977. Its chemical name is β-D-2ʹ-Deoxy-2ʹ-α-fluoro-2ʹ-β-C-methyluridine with molecular formula C22H29FN3O9P. It is a nucleotide prodrug approved for treatment of HCV infection by the United States Food and Drug Administration (FDA, 2013) and European Medicines Agency (2014). Sofosbuvir is one of the DAA drugs because it is a selective inhibitor of the HCV-NS5B polymerase (Sofia et al., 2010).
Although sofo treatment has a potential to reduce the HCV load, it caused many side effects. El Gharabawy et al. (2019) reported that the therapeutic dose of sofo caused renal degenerative and necrotic features including the proximal and distal convoluted tubules in rats. Also, acute kidney injury may happen in 1–15% of patients who are treated with sofo containing regimens (Dashti-Khavidaki et al., 2018). Moreover, Fayed et al. (2018) reported that sofo plus daclatasvir regimen increased risk of developing chronic kidney disease associated with increasing interstitial fibrosis, tubular atrophy and creatinine level. Toxicity after sofo treatment were reported in several organs; testis (El-Kholy et al., 2019), salivary gland (Abdeen et al., 2019), neurotoxicity (Issa & El-Sherif, 2017) and histological and immunohistochemical changes in cornea (Mekawy et al., 2019).
In the last years, medicinal plants are widely used throughout the world, due to their potent pharmacological activities, low toxicity and economic viability when compared with the synthetic drugs (Pracheta et al., 2011). Moringa oleifera (MO) is one of these plants which have many medicinal and nutritional properties associated with the presence of many functional bioactive compounds (Ma et al., 2020).
Moringa oleifera belongs to Moringaceae family and its tree is commonly known as the horseradish or drumstick tree (Morton, 1991). The root, bark, gum, leaf, pods, flowers, seed, and seed oil are used in traditional medicine for treatment of various ailments, so the plant supports the fact that it is often referred to as a wonder-tree (Stevens et al., 2013).
Moringa oleifera oil contains phenolics, flavonoids, antioxidants (Ogbunugafor et al., 2011), sterols, tocopherols and fatty acids (Anwar et al., 2005). The sterol fraction consisted mainly of campesterol, stigmasterol, β-sitosterol and Δ5-avenasterol (among which β-sitosterol was the most predominant) accompanied with trace or minute amounts of Δ7-avenasterol, 24-methylenecholesterol, campestanol, clerosterol, stigmastanol and 28-isoavenasterol while tocopherols consisted of α-, γ- and δ-tocopherols (Anwar et al., 2005). The same authors added that MO oil falls into the category of high-oleic oils and contained a high ratio of monounsaturated to saturated fatty acids.
Concerning the possible protective and curative effects of the different parts of MO, Kotb and Mostafa (2017) reported that MO oil had protective effect against potassium bromate-induced nephrotoxicity in rats through ameliorating the levels of creatinine, malondialdehyde (MDA) and the activity of catalase (CAT). Moreover, MO oil possessed a marked nephroprotective effect against gentamicin (Edeogu et al., 2019) and against 5-fluorouracil (Famurewa et al., 2019) via improving the histopathological changes and CAT activity and reducing the levels of urea, creatinine and renal MDA. Aqueous extract of MO leaves also had a protective effect against the infertility induced by mobile phone electromagnetic radiation in rats by modulating the expression of proliferating cell nuclear antigen (PCNA) and antioxidant activities (Bin-Meferij & El-Kott, 2015).
The present work was designed to evaluate the histological, immunohistochemical and biochemical impacts of sofo treatment on the renal cortex of adult male albino rats and the possible ameliorative role of MO oil.
Methods
Sofosbuvir
Sofosbuvir, its trade name is nucleobuvir, was obtained as yellow-coated tablets from Eva Pharma for Pharmaceuticals and Medical Appliances S.A.E. Company, Giza, Egypt. Each tablet contains 400 mg of sofo. The tablets were dissolved in distilled water and orally given to the experimental animals by gastric tube at a dose level of 36 mg/kg/day (equivalent to the therapeutic dose for human, Paget & Barnes, 1964) for eight weeks.
Moringa oleifera oil
Moringa oleifera oil was purchased from Pure Life Company for Investment and Agricultural Development, Giza, Egypt. It was orally given to the experimental animals at a dose level of 2 ml/kg/day (Olatosin et al., 2014) for eight weeks.
All chemicals used in the present study were of analytical grade and high purity from chemical kits companies, Egypt.
Animals
Adult male albino rats (Rattus norvegicus), 3 months old and weighing 120-140g, were obtained from Helwan farm, Cairo, Egypt. They were housed in standard rat cages under 12/12 hours dark/light cycle at 24±2 ℃. Animals were fed a standard commercial pellets diet, obtained from Egyptian Company of Oil and Soap, Kafr El-Zayat, Gharbia, Egypt, and were given water ad libitum. Rats were acclimatized for 10 days before starting the experiments.
Experimental design
Forty adult healthy male albino rats were equally divided into four experimental groups. Group I was served as control (non-treated group). Group II was orally given MO oil by gastric tube at a dose level of 2ml/kg/day. Group III was orally given sofo at a dose level of 36mg/kg/day by gastric tube. Group IV was orally given sofo, then after two hours they were orally given MO oil (with the same doses were given to groups III and II, respectively). All doses were daily given to the rats for eight weeks.
During the eight weeks of the experiment, daily health checks of the rats of all groups were observed.
Samples collection
At the end of the experiments, rats of all groups were kept fasting for 12 h, then they were anesthetized by diethyl ether and sacrificed. Blood samples were withdrawn from the portal vein in plain tubes and left to clot at room temperature, then were centrifuged at 3000 rpm for 20 min. The collected sera were stored at—20℃ for estimation of biochemical analysis. Kidneys were immediately removed after dissection and were fixed in 10% neutral formalin for 24 h.
Histological preparation
The fixed tissues were dehydrated in ascending series of ethanol, cleared in two changes of xylene, infiltrated in three changes of molten paraffin wax (melting point 58–60 ℃) and embedded in molten paraffin. Sections of 4 microns thickness were cut by using rotary microtome and stained with Ehrlich's hematoxylin and counterstained with eosin (Lillie & Fulmer, 1976).
Immunohistochemical investigations
For immunohistochemical investigations, 4 microns paraffin sections were stained immunohistochemically for visualizing cysteine-aspartic protease (caspase-3) and PCNA positive cells, using suitable antibody in each staining time (Hsu et al., 1981). The intensity of PCNA and caspase-3 expression in kidney sections was quantified by using NIH image j software.
Biochemical analysis
Urea and creatinine levels were determined according to the Quanti Chrom Assay Kit from Bioassay Systems, Hayward, CA, USA. The activity of CAT was measured according to Aebi (1984) and the level of oxidative stress marker MDA was estimated according to Ohkawa et al. (1979). The diagnostic kits for estimation of MDA level and CAT activity were purchased from Biodiagnostic Company, Egypt.
Statistical analysis
Data were expressed as mean ± standard deviation (mean ± SD). The significance of differences between the means was evaluated by using one way analysis of variance (ANOVA) test with post hoc analysis. Statistical package for social science (SPSS) program, version 20 (Inc., Chicago, USA), was used (Kirkpatrick & Feeney, 2012). The significance was classified into three categories; p < 0.0001, p < 0.001 and p < 0.05 according to the obtained p values.
Results
Clinical observations
Both the control and MO oil-treated rats were appeared to have normal activity and normal adequate food and water intake. Sofosbuvir-treated rats exhibited general weakness and loss of appetite. On the other hand, sofo- and MO oil-treated rats did not show any abnormality.
Histological observations
Examination of kidney of control rats showed that it covered externally with a thin layer of fibrous tissue, the renal capsule. The kidney tissue consists of two parts; the outer part (the renal cortex) and the inner one (the medulla). Nephron is the structural and functional unit of the kidney that consists of different parts including renal corpuscle, convoluted tubules, loop of Henle and collecting tubule. The nephron is found completely in the renal cortex except its loop of Henle and collecting tubule that found in the medulla.
The renal corpuscle is a rounded structure consists of glomerulus which is formed of a tuft of blood capillaries surrounded by a cup-like doubled layered structure named Bowman’s capsule. The external layer (parietal face) of the capsule forms its outer surface and consists of a simple squamous epithelium, while the internal layer (visceral face) consists of modified cells called podocytes and closely envelopes the glomerular capillaries. Between the two layers of the capsule there is a urinary space which is continuous with the lumen of renal tubules.
The convoluted tubules include proximal and distal ones. The proximal convoluted tubules have a relatively small lumen and lined with cuboidal cells provided with prominent brush borders and contain central nuclei. The distal convoluted tubules have a much wider lumen than proximal ones and lined with cuboidal cells with apical nuclei and do not have a brush border (Fig. 1a).
No pathological changes were observed in the kidney of MO oil-treated rats. The renal cortex showed normal structure of both glomeruli and convoluted tubules which appeared mostly similar to that of control animals (Fig. 1b).
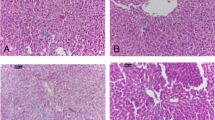
Microscopic examination of kidney of sofo-treated animals showed many pathological changes. The renal corpuscles appeared with variable sizes and Bowman’s capsules were observed with wrinkles basement membranes. Most of the glomeruli appeared either atrophied (collapsed, without identifiable capillaries) with widen urinary space or hypertrophied with narrow urinary space. Some of the renal tubules were degenerated with widen lumen filled with proteinaceous casts. Some tubular epithelial cells appeared with wrinkles membrane, vacuolated cytoplasm and pyknotic nuclei. Erosion of brush borders of some proximal tubules was also observed (Fig. 2a–c). Moreover, intratubular hemorrhage, congested blood vessels and leukocytic infiltration were observed (Fig. 2d and e).
Photomicrographs obtained from kidney sections of sofo-treated rats showing a–c collapsed glomerulus (CG) with widen urinary space, hypertrophied glomerulus (HG) with narrow urinary space, tubular epithelial cells with vacuolated cytoplasm (arrows), pyknotic nuclei (irregular arrows) and degenerated tubules with widen lumens (L) contain proteinaceous casts (stars), d and e widen lumens (L) of degenerated tubules, hemorrhage (H), leukocytic infiltration (LI) and congested blood vessel (CBV), (H&E, X 400)
Histological examination of kidney sections of rats treated with sofo and MO oil showed an obvious degree of recovery. The majority of Bowman’s capsules appeared with regular basement membranes and normal urinary spaces. Most of the glomeruli appeared healthy with normal capillary tufts. The lining epithelium of both proximal and distal tubules appeared mostly normal except few degenerated cells (Fig. 3a and b).
Immunohistochemical investigations
PCNA expression
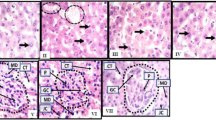
In kidneys of both control and MO oil-treated animals, PCNA was expressed as brown color in few nuclei of glomerular and tubular cells (Fig. 4a and b). There was nonsignificant difference (1.56 ± 0.18 & 1.81 ± 0.10) in the percentage areas of PCNA expression between control and MO oil groups, respectively (Fig. 4e). Sofosbuvir-treated animals showed positive PCNA expression in a large number of the nuclei of glomerular and tubular cells (Fig. 4c). This group showed significant increase (11.49 ± 1.01) in the percentage area at p˂0.0001 when compared with the control group (Fig. 4e). On the other hand, sofo- and MO oil-treated animals showed decrease in the number of PCNA positive nuclei (Fig. 4d). In this group, a significant decrease (3.59 ± 0.54) at p ˂ 0.0001 was recorded in the percentage area of PCNA expression comparing with sofo group, but still being recorded a significant increase (p ˂ 0.001) comparing with the control one (Fig. 4e).
Photomicrographs obtained from kidney sections of a control rat showing PCNA expression as brown color in few nuclei of glomerular and tubular cells (arrows), b MO oil-treated rat showing normal PCNA expression (arrows), c sofo-treated rat showing large number of nuclei with positive PCNA expression (arrows), d sofo- and MO oil-treated rat showing PCNA expression in some nuclei of glomerular and tubular cells (arrows), (PCNA immunostain, counterstained with hematoxylin, X 400). e The percentage area of PCNA expression in the different experimental groups. (⁎⁎) p < 0.001 and (⁎⁎⁎) p < 0.0001significant comparing with control group. (a) p < 0.0001 significant comparing with sofo group
Caspase-3 expression
In both the control and MO oil groups, caspase-3 was expressed as brown color in the cytoplasm of few glomerular and tubular cells (Fig. 5a and b). Nonsignificant difference (1.45 ± 0.23 & 1.48 ± 0.29) in the percentage areas of caspase-3 expression was recorded between the two groups (Fig. 5e). Sections obtained from sofo-treated animals showed positive caspase-3 expression in the cytoplasm of large number of tubular and glomerular cells (Fig. 5c). A significant increase (11.84 ± 0.67) at p ˂ 0.0001 was recorded in the percentage area of caspase-3 expression of this group comparing with the control one (Fig. 5e). On the other hand, kidney of sofo- and MO oil-treated rats showed positive caspase-3 expression in the cytoplasm of few cells (Fig. 5d). This group recorded significant decrease (3.41 ± 0.42) at p ˂ 0.0001 in the percentage area of caspase-3 expression comparing with sofo group, but still being recorded significant increase (p˂0.05) comparing with the control one (Fig. 5e).
Photomicrographs obtained from kidney sections of a control rat showing normal caspase-3 expression as brown color in the cytoplasm of few glomerular and tubular cells (arrows), b MO oil-treated rat showing normal expression of caspase-3 in the cytoplasm of few cells (arrows), c sofo-treated rat showing caspase-3 expression in the cytoplasm of large number of glomerular and tubular cells (arrows), d sofo- and MO oil-treated rat showing caspase-3 expression in the cytoplasm of few numbers of glomerular and tubular cells (arrows), (caspase-3 immunostain, counterstained with hematoxylin, X 400). e The percentage area of caspase-3 expression in the different experimental groups. (⁎⁎) p < 0.001 and (⁎⁎⁎) p < 0.0001significant comparing with control group. (a) p < 0.0001 significant comparing with sofo group
Biochemical results
Kidney function markers (urea and creatinine) in serum
Figure 6a and b shows the changes in urea and creatinine levels in sera of the different experimental groups. In both control and MO oil-treated animals, nonsignificant differences were recorded in the levels of both urea (16.95 ± 2.43 and 17.19 ± 2.51, respectively) and creatinine (0.16 ± 0.03 and 0.18 ± 0.05, respectively). When animals were treated with sofo, a significant increase (59.70 ± 5.13 and 1.09 ± 0.18) at p < 0.0001 was recorded in urea and creatinine levels, respectively, comparing with the control group, whereas sofo- and MO oil-treated rats showed significant decrease (22.91 ± 1.42 and 0.36 ± 0.06) at p < 0.0001 in urea and creatinine levels, respectively, comparing with sofo group, but still being showed a significant increase (p < 0.001) comparing with the control group.
Oxidative stress marker, MDA, in serum
Nonsignificant difference (5.90 ± 1.17 and 5.72 ± 0.91) in MDA levels was recorded in sera of control and MO oil-treated animals, respectively. Moreover, a significant increase (28.70 ± 4.39) at p < 0.0001 in MDA level was recorded in sofo-treated rats when compared with the control group. On the other hand, when rats were treated with sofo and MO oil, a significant decrease (9.69 ± 0.44) at p < 0.0001 in MDA level was recorded comparing with sofo group, but still being recorded significant increase (p < 0.05) comparing with the control group (Fig. 6c).
Catalase activity
Figure 6d shows CAT activity in sera of the different experimental groups. There was non-statistical difference (42.60 ± 4.84 and 42.50 ± 4.45) between the control and MO oil-treated groups, respectively. In sofo-treated animals, a significant decrease (13.47 ± 2.73) at p < 0.0001 in CAT activity was recorded, comparing with the control group. On the other hand, sofo- and MO oil-treated rats showed significant increase (35.90 ± 1.98) at p < 0.0001 in CAT activity comparing with sofo group, but still being recorded significant decrease (p < 0.001) comparing with the control group.
Discussion
In the present study, sofo-treated rats showed many degenerative changes in the renal cortex. These changes may be attributed to the toxic effect of sofo as a nucleotide inhibitor (ribonucleotide analog). Feng et al. (2016) reported that all nucleotide inhibitors of HCV are ribonucleotide analogs that inhibit mitochondrial protein synthesis leading to a corresponding decrease in mitochondrial oxygen consumption in cells which consider as a valuable evidence to the mechanism for the clinical toxicity observed with some ribonucleotide analogs.
The cytoplasmic vacuolization that observed in the present study may be attributed to impairment of mitochondria. This result is concord with explanation of Miller and Zachary (2017) who reported that the impairment of mitochondria may be led to reduction of ATP production which causes depletion in Na–K pump leading to accumulation of water inside the cells in order to regulate the osmotic pressure. Abdeen et al. (2019) also attributed the cytoplasmic vacuolization induced by sofo to the mitochondrial toxicity. Elwan et al. (2018) attributed this pathological feature to the disturbance of cell membrane function of the renal tubular epithelium that leads to massive influx of water and Na+ ions inside the cells.
Sofosbuvir treatment in the present study led to congestion of blood vessels and leukocytic infiltration, which may be related to a prominent immune response of the body tissues by movement of fluids and leukocytes from the blood vessels to the intravascular tissue. This consequently causes concentration of erythrocytes leading to increase blood viscosity and slow the circulation which microscopically translated by numerous dilated small vessels packed with erythrocytes (Mitchell & Cotran, 2003). In agreement with our results, Salem et al. (2017) and El Gharabawy et al. (2019) reported that sofo treatment induced leukocytic inflammatory cells in the connective tissue stroma and congested dilated blood vessels in different organs in rats.
Moreover, the interstitial hemorrhage which observed in our results may be due to severe interstitial inflammation induced by sofo. The renal interstitial hemorrhage was explained by Asim et al. (2021) who reported that, in the severe interstitial inflammation, a significant breach in the integrity of the walls of the interstitial blood vessels leads to extravasation of red blood cells from the peri-tubular capillaries resulting in widespread interstitial hemorrhages. Hemolysis of extravasated erythrocytes released hemoglobin and led to tissue accumulation of ferric ions which promotes production of iron-derived hydroxyl radicals leading to promotion of acute kidney injury and generation of a compensatory protective response (Cleary et al., 2010). Sofosbuvir treatment also caused hypercellularity, vascular dilation and extravasation of blood in cerebral cortex of rats that result in impairment or loss of neural function (Issa & El-Sherif, 2017).
Concerning the immunohistochemical observation, in the current study, sofo treatment increased PCNA and caspase-3 expressions which may be attributed to the mitochondrial damage resulted from the oxidative stress induced by sofo. This suggestion is concord with Sara et al. (2016) who found that the increased ROS induced mitochondrial disturbance which translated by their swelling and increasing permeability as well as elevation of caspase-3 expression causing apoptosis. Moreover, ROS increase pro-inflammatory cytokines leading to activation of caspase-3 which moderates inflammatory response and apoptosis (Refaie et al., 2017). Increased caspase activity was reported to be associated with cellular damage which occurs in a number of diseases (Lavrik et al., 2005). Reactive oxygen species are considered the important signaling molecules which may influence cell proliferation, cell death (either apoptosis or necrosis) and gene expression (Genestra, 2007). In agreement with the present results, Elghazouly and Yassien (2021) reported that sofo caused marked increase in PCNA expression in rats. Expression of PCNA may be transiently accumulated at site of DNA damage (Essers et al., 2005).
In the current study, sofo treatment increased urea, creatinine and MDA levels and decreased CAT activity. These results may be attributed to the oxidative stress mediated by sofo. Excessive ROS increased urea and creatinine levels and reduced the renal antioxidant capacity, so the kidney became more susceptible to oxidative damage (Hussein et al., 2020; Elmansy et al., 2021). Increased urea and creatinine levels in serum are recognized as biomarkers for chronic kidney damage (Nisha et al., 2017). Similarly, Mohamed et al. (2021) found that sofo-induced acute kidney injury was confirmed by elevation in urea and creatinine levels in sera of rats.
In the present study, when MO oil was given only to the rats, it did not cause any histopathological, immunohistochemical and biochemical changes. Similar results were confirmed by Abarikwu et al. (2017) and Edeogu et al. (2019) in kidney and by Mahran et al. (2021) in liver of rats.
When MO oil was co-administered with sofo, in the current study, an obvious degree of recovery was observed. Both glomeruli and renal tubules appeared nearly similar to the control ones. This improvement may be attributed to the bioactive components of MO oil. Mohamed et al. (2020) attributed the protective effect of MO to its antioxidants, free radical scavenging abilities and anti-inflammatory effects. The anti-inflammatory effect of MO oil was attributed to tocopherols and sterols contents which is effective against pro-inflammatory cytokines production (Adeyi et al., 2021; Edeogu et al., 2019). The improving role of MO oil may be also attributed to its natural antioxidant components; phenolics and carotenoids or to vitamins E and C contents which are actively boosting the immunity of cells in order to prevent or fight against oxidative stress and increase renal repair and regeneration potential (Gasmalbari et al., 2020; Omotoso et al., 2019).
In the present study, when MO oil was co-administered with sofo, it ameliorated caspase-3 and PCNA expressions. This ameliorative effect may be attributed to tocopherols and fatty acids (oleic and linoleic) contents in MO oil. Famurewa et al. (2019) reported that MO oil blocked caspase-3 activation signaling by its antioxidant effect. Reducing the high activity of caspase-3 may be due to tocopherols and sterols contents of MO oil which have anti-apoptotic effects (Edeogu et al., 2019). Abdel-Daim et al. (2020) found that MO reduced the elevated caspase-3 expression and amended the oxidative status by restoring the activity of antioxidant molecules and lowering the levels of pro-oxidant molecules in the kidney tissue. Moringa oleifera also ameliorated PCNA expression in rats after monosodium glutamate-induced hepatic toxicity (Albrahim & Binobead, 2018) and after electromagnetic radiation-induced testicular damage (Bin-Meferij & El-Kott, 2015).
In the current study, when MO oil was co-administered with sofo, it ameliorated urea, creatinine and MDA levels and CAT activity. These biochemical improvements were confirmed by the observed improvement of the kidney structure, which could be attributed to the antioxidant potential of the essential components of MO oil. Moringa oleifera oil's tocopherols and unsaturated fatty acids (oleic and linoleic) helped to reduce MDA level by scavenging free radicals and inhibiting the chain reaction of lipid peroxidation reducing oxidative stress damage (Al-Said et al., 2012; Nafiu, et al., 2019). Recently, Adeyi et al. (2021) found that MO ameliorated MDA level and CAT activity and attributed these results to the presence of electron-donating phytochemicals (phenolic acids, flavonoids, eugenol and tannins) which have anti-ROS effects.
Conclusions
Moringa oleifera oil improved the nephrotoxicity induced by sofo via improving the histopathological, immunohistochemical and biochemical changes due to its richness with different antioxidant, anti-inflammatory and anti-apoptotic components.
Availability of data and materials
Data generated during this study are included in this published article. Please contact authors for data.
Abbreviations
- Caspase-3:
-
Cysteine-aspartic protease
- CAT:
-
Catalase
- DAA:
-
Direct acting antiviral
- HCV:
-
Hepatitis C virus
- MDA:
-
Malondialdehyde
- MO:
-
Moringa oleifera
- PCNA:
-
Proliferating cell nuclear antigen
- ROS:
-
Reactive oxygen species
- sofo:
-
Sofosbuvir
References
Abarikwu, S. O., Benjamin, S., Ebah, S. G., Obilor, G., & Agbam, G. (2017). Protective effect of Moringa oleifera oil against HgCl2-induced hepato- and nephro-toxicity in rats. Journal of Basic and Clinical Physiology and Pharmacology, 28(4), 337–345.
Abdeen, A. M., Essawy, T., & Mohammed, S. S. (2019). Effect of sofosbuvir administration and its withdrawal on the submandibular salivary gland of adult male albino rats: A histological and ultra-structural study. Open Access Macedonian Journal of Medical Sciences, 7(23), 4101–4109.
Abdel-Daim, M. M., Alkahtani, S., Almeer, R., & Albasher, G. (2020). Alleviation of lead acetate-induced nephrotoxicity by Moringa oleifera extract in rats: Highlighting the antioxidant, anti-inflammatory and anti-apoptotic activities. Environmental Science and Pollution Research, 27, 33723–33731.
Adeyi, A. O., Ajisebiola, B. S., Adeyi, O. E., Adekunle, O., Akande, O. B., James, A. S., Ajayi, B. O., Yusuf, P. O., & Idowu, B. A. (2021). Moringa oleifera leaf fractions attenuated Naje haje venom-induced cellular dysfunctions via modulation of Nrf2 and inflammatory signaling pathways in rats. Biochemistry and Biophysics Reports, 25, 100890. https://doi.org/10.1016/j.bbrep.2020.100890
Aebi, H. (1984). Catalase in vitro. Methods in Enzymology, 105, 121–126.
Albrahim, T., & Binobead, M. A. (2018). Roles of Moringa oleifera leaf extract in improving the impact of high dietary intake of monosodium glutamate-induced liver toxicity, oxidative stress, genotoxicity, DNA damage, and PCNA alterations in male rats. Oxidative Medicine and Cellular Longevity, 2018, 4501097. https://doi.org/10.1155/2018/4501097
Al-Said, M. S., Mothana, R. A., Al-Yahya, M. A., Al-Blowi, A. S., Al-Sohaibani, M., Ahmed, A. F., & Rafatullah, S. (2012). Edible oils for liver protection: Hepatoprotective potentiality of Moringa oleifera seed oil against chemical-induced hepatitis in rats. Journal of Food Science, 77(7), T124–T130.
Anwar, F., Ashraf, M., & Bhanger, M. I. (2005). Interprovenance variation in the composition of Moringa oleifera oilseeds from Pakistan. Journal of the American Oil Chemists’ Society, 82(1), 45–51.
Asim, M., Ahmad, F., & Akhtar, M. (2021). Florid interstitial hemorrhages: A novel feature of amoxicillin-clavulanate-induced acute tubulointerstitial nephritis. The American Journal of Case Reports, 22, e928989. https://doi.org/10.12659/AJCR.928989
Bin-Meferij, M. M., & El-Kott, A. F. (2015). The radioprotective effects of Moringa oleifera against mobile phone electromagnetic radiation-induced infertility in rats. International Journal of Clinical and Experimental Medicine, 8(8), 12487–12497.
Cleary, C. M., Moreno, J. A., Fernández, B., Ortiz, A., Parra, E. G., Gracia, C., Blanco-Colio, L. M., Barat, A., & Egido, J. (2010). Glomerular haematuria, renal interstitial haemorrhage and acute kidney injury. Nephrology Dialysis Transplantation, 25(12), 4103–4106.
Dashti-Khavidaki, S., Khalili, H., & Nasiri-Toosi, M. (2018). Potential nephrotoxicity of sofosbuvir-based treatment in patients infected with hepatitis C virus: A review on incidence, type and risk factors. Expert Review of Clinical Pharmacology, 11(5), 525–529.
Edeogu, C. O., Kalu, M. E., Famurewa, A. C., Asogwa, N. T., Onyeji, G. N., & Ikpemo, K. O. (2019). Nephroprotective effect of Moringa oleifera seed oil on gentamicin-induced nephrotoxicity in rats: Biochemical evaluation of antioxidant, anti-inflammatory, and antiapoptotic pathways. Journal of the American College of Nutrition, 39(4), 307–315.
El Gharabawy, G. S. H., Abdallah, E. E. A., Kaabo, H. F., & Abdel Aleem, A. M. A. (2019). Histological effects of sofosbuvir on the kidney of male albino rats and the possible protective role of vitamin C. The Egyptian Journal of Hospital Medicine, 76(1), 3335–3354.
Elghazouly, D. E., & Yassien, R. I. (2021). Effect of sofosbuvir (sovaldi) on the fundic mucosa of adult male albino rats and the possible protective role of fucoidan: Histological, histochemical, and immunohistochemical study. Egyptian Journal of Histology. https://doi.org/10.21608/ejh.2021.51729.1392
El-Kholy, W. B., Faried, M. A., Salama, R. M., El-Fiky, M. M., & El-Garawani, I. (2019). Evaluation of testicular cytotoxicity and genotoxicity of sofosbuvir and sofosbuvir-ribavirin in the adult male albino rats. European Journal of Anatomy, 23(6), 393–403.
Elmansy, R. A., Seleem, H. S., Mahmoud, A. R., Hassanein, E., & Ali, F. (2021). Rebamipide potentially mitigates methotrexate-induced nephrotoxicity via inhibition of oxidative stress and inflammation: A molecular and histochemical study. Anatomical Record, 304(3), 647–661.
Elwan, W. M., Ragab, A. M. H., & Ragab, M. H. (2018). Histological and immunohistochemical evaluation of the dose dependent effect of gold nanoparticles on the renal cortex of adult female albino rat. Egyptian Journal of Histology, 41(2), 167–181.
Essers, J., Theil, A. F., Baldeyron, C., van Cappellen, W. A., Houtsmuller, A. B., Kanaar, R., & Vermeulen, W. (2005). Nuclear dynamics of PCNA in DNA replication and repair. Molecular and Cellular Biology, 25(21), 9350–9359.
European Medicine Agency. (2014). https://www.ema.europa.eu/en/medicines/human/EPAR/sovaldi
Famurewa, A. C., Asogwa, N. T., Aja, P. M., Akunna, G. G., Awoke, J. N., Ekeleme-Egedigwe, C. A., Maduagwuna, E. K., Folawiyo, A. M., Besong, E. E., Ekponoc, E. U., & Nwoha, P. A. (2019). Moringa oleifera seed oil modulates redox imbalance and iNOS/NF-κB/caspase-3 signaling pathway to exert antioxidant, anti-inflammatory and antiapoptotic mechanisms against anticancer drug 5-fluorouracil-induced nephrotoxicity in rats. South African Journal of Botany, 127, 96–103.
Fayed, A., El Nokeety, M. M., Samy Abdelaziz, T., Samir, H. H., Hamza, W. M., & El Shabony, T. (2018). Incidence and characteristics of de novo renal cryoglobulinemia after direct-acting antivirals treatment in an Egyptian hepatitis C cohort. Nephron, 140(4), 275–281.
Feng, J. Y., Xu, Y., Barauskas, O., Perry, J. K., Ahmadyar, S., Stepan, G., Yu, H., Babusis, D., Park, Y., McCutcheon, K., Perron, M., Schultz, B. E., Sakowicz, R., & Ray, A. S. (2016). Role of mitochondrial RNA polymerase in the toxicity of nucleotide inhibitors of hepatitis C virus. Antimicrobial Agents and Chemotherapy, 60(2), 806–817.
Gasmalbari, E., & EL-Kamali, H. H., & Abbadi, O. S. (2020). Biochemical and haematological effects and histopathological changes caused by Moringa oleifera on albino rats. Chinese Journal of Medical Research, 3(3), 84–88.
Genestra, M. (2007). Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cellular Signalling, 19(9), 1807–1819.
Hsu, S. M., Raine, L., & Fanger, H. (1981). Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: A comparison between ABC and unlabeled antibody (PAP) procedures. Journal of Histochemistry and Cytochemistry, 29(4), 577–580.
Hussein, S. A., El-Senosi, Y. A., Esmael, T. E. A., Amin, A., & Sarhan, E. A. M. (2020). Thymoquinone suppressed cyclosporine A-induced nephrotoxicity in rats via antioxidant activation and inhibition of inflammatory and apoptotic signaling pathway. Benha Veterinary Medical Journal, 39(1), 40–46.
Issa, N. M., & El-Sherif, N. M. (2017). Light and electronic histological studies to the effect of sofosbuvir on the visual cerebral cortex of adult male albino rat. Journal of American Science, 13(4), 79–87.
Kirkpatrick, L. A., & Feeney, B. C. A. (2012). A Simple guide to IBM SPSS statistics: For version 20 (12th ed.). Wadsworth.
Kotb, M. A., & Mostafa, M. Y. (2017). Nephroprotective effect of Moringa oleifera oil against KBr O3 toxicity in adult rats. Journal of Studies and Searches of Specific Education, 3(1), 181–198.
Lavrik, I. N., Golks, A., & Krammer, P. H. (2005). Caspases: Pharmacological manipulation of cell death. The Journal of Clinical Investigation, 115(10), 2665–2672.
Lillie, R. D., & Fulmer, H. M. (1976). Histopathological techniques and practical histochemistry (4th ed.). McGraw-Hill.
Ma, Z. F., Ahmad, J., Zhang, H., Khan, I., & Muhammad, S. (2020). Evaluation of phytochemical and medicinal properties of moringa (Moringa oleifera) as a potential functional food. South African Journal of Botany, 129, 40–46.
Mahran, H. A., Gawaan, Y. M., & El-Gerbed, M. S. A. (2021). Ameliorative effect of moringa seed oil against chlorambucil-induced DNA damage and hepatotoxicity in adult male albino rats. Research Journal of Pharmaceutical, Biological and Chemical Sciences, 12(1), 182–196.
Mekawy, N. H., Abdel-aziz, H. M., & Ibrahem, N. E. (2019). Protective effect of chrysin on corneal structural alterations induced by sofosbuvir in male albino rats (histological and immunohistochemical study). Journal of Histology & Histopathology. https://doi.org/10.7243/2055-091X-6-5
Miller, M. A., & Zachary, J. F. (2017). Mechanisms and morphology ofcellular injury, adaptation, and death. In J. F. Zachary & M. D. McGavin (Eds.), Pathologic basis of veterinary disease (6th ed.). Missouri: St Louis.
Mitchell, R. N. & Cotran, R. S. (2003). Acute and chronic inflammation. In Kumar, V., Cotran, R. S., & Robbins D. S. L. (7th Ed), Robbins basic pathology (p. 35). Philadelphia, Elsevier Saunders.
Mohamed, A. F., Abo-Ouf, A. M., & Arafa, M. (2021). Histological and biochemical studies on effect of Sofosbuvir (Sovaldi) on adult male albino rat kidney. Ultrastructural Pathology, 16, 1–11. https://doi.org/10.1080/01913123.2021.1966148
Mohamed, N. B., Mohamed, A. H., Abu-Aita, N. A., Nasr, S. M., Nassar, S. A., & Ahmed, K. A. (2020). Moringa oleifera leaf ethanol extract ameliorates lead-induced hepato-nephrotoxicity in rabbits. Asian Pacific Journal of Tropical Biomedicine, 10(6), 263–272.
Morton, J. F. (1991). The horseradish tree, Moringa pterygosperma (Moringaceae): A boon to arid lands? Economic Botany, 45(3), 318–333.
Nafiu, A. O., Akomolafe, R. O., Alabi, Q. K., Idowu, C. O., & Odujoko, O. O. (2019). Effect of fatty acids from ethanol extract of Moringa oleifera seeds on kidney function impairment and oxidative stress induced by gentamicin in rats. Biomedicine and Pharmacotherapy, 117, 109154. https://doi.org/10.1016/j.biopha.2019.109154
Nisha, R., Kannan, S. R. S., Mariappan, K. T., & Jagatha, P. (2017). Biochemical evaluation of creatinine and urea in patients with renal failure undergoing hemodialysis. Journal of Clinical Pathology and Laboratory Medicine, 1(2), 1–5.
Ogbunugafor, H. A., Eneh, F. U., Ozumba, A. N., Igwo-Ezikpe, M. N., Okpuzor, J., Igwilo, I. O., Adenekan, S. O., & Onyekwelu, O. A. (2011). Physico-chemical and antioxidant properties of Moringa oleifera seed oil. Pakistan Journal of Nutrition, 10(5), 409–414.
Ohkawa, H., Ohishi, N., & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95(2), 351–358.
Olatosin, T. M., Akinduko, D. S., & Uche, C. Z. (2014). Antioxidant capacity of Moringa oleifera seed oil against CCl4-induced hepatocellular lipid peroxidation in wistar albino rats. European Journal of Experimental Biology, 4(1), 514–518.
Omotoso, O. D., Adelakun, S. A., Idomeh, I. J., & Ogbonna, E. (2019). Histomorphological characterization of Moringa oleifera oil and walnut oil on cadmium-induced lateral geniculate body damage in adult wistar rats (Rattus norvegicus). Journal of Biology and Medicine, 3(1), 001–007.
Paget, C. E., & Barnes, J. M. (1964). Toxicity tests. In Laurence, D. R., & Bacharach, A. L., Evaluation of drug activity pharmacometries (pp. 135). London: Academic Press.
Pockros, P. J. (2010). New direct-acting antivirals in the development for hepatitis C virus infection. Therapeutic Advances in Gastroenterology, 3(3), 191–202.
Pracheta, S. S., Sharma, V., Paliwal, R., Sharma, S., Singh, L., Janmeda, B. S., Yadav, S., & Sharma, S. H. (2011). Chemoprotective activity of hydro-ethanolic extract of Euphorbia nerrifolia Linn. leaves against DENA-induced liver carcinogenesis in mice. Biology and Medicine, 3(2), 36–44.
Refaie, M. M., Ibrahim, S. A., Sadek, S. A., & Abdelrahman, A. M. (2017). Role of ketotifen in methotrexate-induced nephrotoxicity in rats. Egyptian Journal of Basic and Clinical Pharmacology, 7(2), 70–80.
Salem, Z. A., Elbaz, D. A., & Farag, D. B. (2017). Effect of an anti-hepatitis C viral drug on rat submandibular salivary gland. Egyptian Dental Journal, 63(1), 657–665.
Sara, H., Rachid, R., Salim, G., Aml, A., Amna, A., Aya, S., Nadjiba, T., Chahinez, T., Zina, B., Hajer, C., Samira, B., Mohamed, K., Mariem, A., & Belgacem, D. (2016). Oxidative stress status, caspase-3, stromal enzymes and mitochondrial respiration and swelling of Paramecium caudatum in responding to the toxicity of Fe3O4 nanoparticles. Toxicology and Environmental Health Sciences, 8(2), 161–167.
Sofia, M. J., Bao, D., Chang, W., Du, J., Nagarathnam, D., Rachakonda, S., Reddy, P. G., Ross, B. S., Wang, P., Zhang, H. R., Bansal, S., Espiritu, C., Keilman, M., Lam, A. M., Steuer, H. M. M., Niu, C., Otto, M. J., & Furman, P. A. (2010). Discovery of a β-d-2’-deoxy-2’-α-fluoro-2’-β-C-methyluridine nucleotide prodrug (PSI-7977) for the treatment of hepatitis C virus. Journal of Medicinal Chemistry, 53(19), 7202–7218.
Stevens, G. C., Baiyeri, K. P., & Akinnnagbe, O. (2013). Ethno-medicinal and culinary uses of Moringa oleifera Lam Nigeria. Journal of Medicinal Plants Research, 7(13), 799–804.
United States Food and Drug Administration (FDA) (2013). https://www.gilead.com/news-and-press/press-room/press releases/2013/12/us-food-and-drug-administration-approves-gileads-sovaldi-sofosbuvir-for-the-treatment-of-chronic-hepatitis-c.
WHO (2021). https://www.who.int/news-room/fact-sheets/detail/hepatitis-c.
Acknowledgements
Not applicable.
Funding
The authors received no funding for this study.
Author information
Authors and Affiliations
Contributions
MH and AS contributed to the design, prepared materials and figures and supervised the practical experiments, wrote and revised the manuscript. OY read and approved the final manuscript. ZA carried out the practical experiments, prepared data analysis, materials and figures and wrote the first draft of the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All the experiments were done in compliance with the guide for care and the use of laboratory animals approved by Faculty of Science, Menoufia University, Egypt (Approval No. MUFS/F/HI/5/18), and according to the National Institutes of Health guide for the care and use of laboratory animals (NIH publications No. 8023, received 1978).
Consent for publication
All authors consent to submission of the manuscript for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mahran, H.A., Okdah, Y.A., Zaky, A.A. et al. The possible ameliorative role of Moringa oleifera seed oil on sofosbuvir-induced nephrotoxicity in albino rats; histopathological, immunohistochemical and biochemical studies. JoBAZ 83, 16 (2022). https://doi.org/10.1186/s41936-022-00281-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41936-022-00281-y