Abstract
Background
Tardive dyskinesia (TD) is a persistent and potentially disabling movement disorder associated with antipsychotic use. Data from RE-KINECT, a real-world study of antipsychotic-treated outpatients, were analyzed to assess the effects of possible TD on patient health and social functioning.
Methods
Analyses were conducted in Cohort 1 (patients with no abnormal involuntary movements) and Cohort 2 (patients with possible TD per clinician judgment). Assessments included: EuroQoL’s EQ-5D-5L utility (health); Sheehan Disability Scale (SDS) total score (social functioning); patient- and clinician-rated severity of possible TD (“none”, “some”, “a lot”); and patient-rated impact of possible TD (“none”, “some”, “a lot”). Regression models were used to analyze the following: associations between higher (worse) severity/impact scores and lower (worse) EQ-5D-5L utility (indicated by negative regression coefficients); and associations between higher (worse) severity/impact scores and higher (worse) SDS total score (indicated by positive regression coefficients).
Results
In Cohort 2 patients who were aware of their abnormal movements, patient-rated TD impact was highly and significantly associated with EQ-5D-5L utility (regression coefficient: − 0.023, P < 0.001) and SDS total score (1.027, P < 0.001). Patient-rated severity was also significantly associated with EQ-5D-5L utility (− 0.028, P < 0.05). Clinician-rated severity was moderately associated with both EQ-5D-5L and SDS, but these associations were not statistically significant.
Conclusions
Patients were consistent in evaluating the impacts of possible TD on their lives, whether based on subjective ratings (“none”, “some”, “a lot”) or standardized instruments (EQ-5D-5L, SDS). Clinician-rated severity of TD may not always correlate with patient perceptions of the significance of TD.
Similar content being viewed by others
Background
Tardive dyskinesia (TD) is a persistent and potentially disabling movement disorder that is associated with exposure to antipsychotics and other dopamine receptor blocking agents [1, 2]. While second-generation (“atypical”) antipsychotics have been associated with a lower incidence of TD, prevalence studies have shown the continued importance of TD as a clinical consideration with these therapies due to their expanding use, especially in non-psychotic conditions [3, 4]. Therefore, it is important that all patients taking any antipsychotic medication be screened regularly for potential TD symptoms, including abnormal involuntary movements in the face/mouth, neck/trunk, upper extremities, and lower extremities [5].
Early case reports of TD suggested that it may be reversible if diagnosed early in some patients who could be safely withdrawn from antipsychotics and followed over time; therefore, when appropriate, tapering off antipsychotic medications could be tried when TD first emerges [6]. However, antipsychotic discontinuation is not a practical option for patients with chronic psychotic disorders, and the overall evidence for discontinuation leading to resolution of TD is insufficient, especially in established cases [7]. In these cases, use of an approved TD medication (i.e., vesicular monoamine transporter 2 inhibitor) is appropriate. Moreover, current treatment guidelines indicate that factors such as patient preference, impairment, or psychosocial functioning should be considered when planning treatment [8]. Consistent with these recommendations, results from a modified Delphi consensus study indicated that improvements in patient-reported distress, functional impairments, and health-related quality of life (HRQoL) should be considered when determining treatment success in patients with TD [9].
The impact of TD can be wide-ranging. The cumulative effects of TD on patients with a serious mental illness may include worsened psychopathology, higher rates of comorbidities, increased risk of mortality, and poorer treatment outcomes [10,11,12]. TD can negatively affect motor functions such as speech, gait, and respiration, as well as cognitive functions such as verbal memory and processing [11, 13, 14]. TD can also lead to feelings of stigmatization, social withdrawal, loss of employment, and higher healthcare resource utilization [12, 15, 16].
Although many clinicians, patients, and caregivers are aware of these negative impacts, quantitative assessments of HRQoL in TD are very limited. RE-KINECT was a real-world study that included psychiatric outpatients who had been treated with antipsychotics. Previously published results from this study indicated that 27.6% of these patients had abnormal involuntary movements that were consistent with possible TD, and that the impact of these movements on both patients and caregivers was considerable [17, 18]. Relevant to the current analyses, RE-KINECT included two established patient-reported HRQoL measures: EuroQoL’s 5-dimension 5-level questionnaire (EQ-5D-5L) and the Sheehan Disability Scale (SDS). Analyses based on these outcomes were used to further explore the impact of TD on patients’ overall health, physical wellness, and social functioning. The findings from these analyses are intended to provide the type of information needed for appropriate health technology assessments, as needed for the evaluation of medications used to treat TD. In a broader sense, the quantitative analyses in this report are useful for understanding the impact of TD beyond subjective impressions. Moreover, they allow for comparisons among different types of patients (e.g., those with no abnormal involuntary movements versus those with possible TD).
Methods
Study design
RE-KINECT was a prospective, real-world, observational, multicenter study conducted at 37 outpatient psychiatry clinics (e.g., research institutions, community health centers, private practices) in the United States from April 2017 to January 2018 [17]. In brief, this study included adults with ≥ 3 months of lifetime exposure to antipsychotic medication and ≥ 1 clinician-confirmed psychiatric disorder according to Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria [19]. The 3 month lifetime antipsychotic exposure requirement was based on the Schooler-Kane criteria for TD research and DSM-IV-TR [20, 21], and it is consistent with current American Psychiatric Association guidelines (DSM-5-TR) [22]. All patients provided written informed consent prior to participation; Institutional Review Board approval was obtained at each site.
Patient cohorts
Patients were assigned to 1 of 2 cohorts based on clinician assessment (Fig. 1). Training for TD screening (via videos) was provided to all clinical site personnel to promote inter-rater reliability. Cohort 1 was defined in the study protocol as patients who had no abnormal involuntary movements or whose movements were not consistent with possible TD based on clinician assessment. To avoid any potential overlap with patients with possible TD (as described below), this analysis focused on a modified Cohort 1 (no abnormal involuntary movements), which excluded patients with non-TD involuntary movements such as tremor.
Cohort 2 was defined in the study protocol as patients who had abnormal involuntary movements that were confirmed by their clinician as possible TD. All patients in Cohort 2 had a clinician-rated severity of possible TD as “some” or “a lot” in at least 1 of the following body regions: head/face, neck/trunk, upper extremities, and/or lower extremities. For this analysis, Cohort 2A (“aware”) was defined as patients with clinician-confirmed possible TD who also self-reported having abnormal involuntary movements within the past 4 weeks and had a self-rated severity of “some” or “a lot” in at least 1 of the 4 body regions. Data from Cohort 2NA (“not aware”), defined as patients with possible TD who self-reported having no abnormal involuntary movements in the past 4 weeks, were analyzed to provide supplementary information about the potential effects of patient awareness on HRQoL.
Assessments
Prior to clinician assessment and cohort assignment, all eligible and consenting patients were asked to complete the EQ-5D-5L [23], which assesses problems in 5-dimensions (mobility, self-care, usual activities, pain/discomfort, anxiety/depression) that can be used to calculate a utility score, which ranges from 0 (health state equivalent to death) to 1 (perfect health). The EQ-5D-5L also includes a visual analog scale (VAS) that measures current health state, which ranges from 0 to 100 (best possible health). Patients were also asked to complete the SDS [24], which assesses disruption in 3 social functioning domains (work/school, social life, family life/home responsibilities) and includes a total score (sum of domain scores) which ranges from 0 (no disruption) to 30 (extreme disruption).
For severity of possible TD, clinicians and patients were asked to “rate the severity of visible, uncontrollable movements” for each of 4 body regions (head/face, neck/trunk, upper extremities, and/or lower extremities) using simple descriptors of “none”, “some”, or “a lot”. For impact of possible TD, patients who were aware of their abnormal involuntary movements (Cohort 2A) were asked to rate how much “over the past 4 weeks” did these movements “impact your ability” to perform each of 7 different activities/functions (usual activities, talking, eating, breathing, being productive, self-care, socializing), also using the descriptors of “none”, “some”, or “a lot”. For regression analyses, these descriptors were assigned values of 0, 1, and 2, respectively, with the summary score for severity ranging from 0 (“none” in all 4 regions) to 8 (“a lot” in all 4 regions) and the summary score for impact ranging from 0 (“none” in all 7 activities) to 14 (“a lot” in all 7 activities).
Analyses
All analyses were intended to be exploratory, and no adjustments were made for multiple comparisons. Patients’ demographics, clinical characteristics, EQ-5D-5L scores, and SDS scores were analyzed for Cohorts 2 and 2A versus modified Cohort 1, as well as for Cohort 2A versus Cohort 2NA. Chi-squared tests were used for categorical variables and t-tests were used for continuous variables. Analyses were adjusted for potentially confounding factors as follows: age (18–95 years); sex (male or female); patient-reported overall health status (0 = “no health problems” to 10 = “health as bad as you can imagine”); clinician-rated severity of psychiatric condition (0 = “normal, not ill” to 6 = “among the most severely ill”); clinician-rated functional status of patient (0 = working/studying/managing household independently to 2 = not working/studying/managing household in any capacity); and psychiatric diagnosis (presence or absence of schizophrenia or schizoaffective disorder, presence or absence of a mood or other disorder). These factors were based on clinically relevant characteristics that would likely affect HRQoL in patients with TD; no statistical process was used for selection.
Simple linear regressions were used to evaluate EQ-5D-5L utility and SDS total scores by clinician/patient-rated severity of possible TD and by patient-rated impact of possible TD. These analyses compared Cohorts 2 and 2A to modified Cohort 1, based on mean score differences between the cohorts. Simple linear regressions were also used to determine the associations between possible TD (clinician/patient-rated severity, patient-rated impact) and HRQoL measures—physical wellness (EQ-5D-5L utility) or social function (SDS total) in Cohort 2, Cohort 2A, and Cohort 2NA. These analyses were adjusted for age, sex, patient-reported overall health status, clinician-rated severity of psychiatric condition, clinician-rated functional status of patient, and psychiatric diagnosis.
Results
Patient cohorts and baseline characteristics
A total of 1148 patients from 37 clinical sites in 19 states were screened, including patients from research institutions, community health centers, and private psychiatric practices. Modified Cohort 1 included 450 patients who had no abnormal involuntary movements (possible TD or otherwise) (Fig. 1). The full Cohort 2 included 204 patients who had abnormal involuntary movements that were clinician-confirmed as possible TD, with a clinician-rated severity of “some” or “a lot” in ≥ 1 body region. Cohort 2A (aware) included 110 patients with self-reported abnormal involuntary movements in the past 4 weeks, with a self-rated severity of “some” or “a lot” in ≥ 1 body region. Cohort 2NA (not aware) included 94 patients who had clinician-confirmed possible TD but self-reported no abnormal involuntary movements within the past 4 weeks.
Compared to patients with no abnormal involuntary movements (modified Cohort 1), patients with possible TD (Cohort 2) were significantly older, with a higher proportion of male patients, higher incidence of schizophrenia or schizoaffective disorder, and lower incidence of mood or other psychiatric disorders (Table 1). Per clinician impression, patients with possible TD also had more severe psychiatric conditions and were less likely to be engaged in what many would consider “usual activities” such as working/studying or managing a household independently and more likely to not engage in these activities at all. Per patient self-report, those with possible TD had worse overall health status than those with no abnormal involuntary movements.
No significant differences between Cohort 2NA and Cohort 2A were found for age, sex, psychiatric condition, clinician-rated severity of psychiatric condition, or clinician impression of overall functional status. However, compared to Cohort 2NA, patients in Cohort 2A had significantly worse self-reported overall health (Additional file 1: Appendix Table S1).
EQ-5D-5L and SDS scores
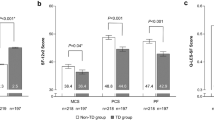
When adjusted for age, sex, health status, severity of psychiatric condition, functional status, and psychiatric diagnosis, mean baseline EQ-5D-5L scores were lower in patients with possible TD (Cohort 2) than in patients with no abnormal involuntary movements (modified Cohort 1), indicating relatively worse overall health (utility score and visual analog scale [VAS]) (Table 2). Mean SDS dimension and total scores were higher in Cohort 2 compared to modified Cohort 1, indicating relatively worse social functional status in patients with possible TD than in patients with no abnormal involuntary movements; however, none of the differences in mean scores were statistically significant. Differences between Cohorts 2A and 2NA for mean SDS total and family/home life scores indicated statistically significantly worse social functioning in patients who were aware of their possible TD than in those who were not aware (Additional file 1: Appendix Table S2).
Compared to patients with no abnormal involuntary movements (modified Cohort 1), a higher percentage of patients with clinician-confirmed possible TD (Cohorts 2 and 2A) reported having moderate problems (score = 3), severe problems (score = 4), or extreme problems (score = 5) in all EQ-5D-5L dimensions (Fig. 2). Interestingly, the dimensions of mobility and self-care were significantly worse in Cohorts 2 and 2A than in modified Cohort 1. Compared to patients who were aware of their possible TD (Cohort 2A), significantly fewer unaware patients (Cohort 2NA) had problems with mobility and self-care (Additional file 1: Appendix Fig. S1). However, moderate or severe pain/discomfort were more prevalent in unaware patients.
Analyses of EQ-5D-5L utility and SDS total scores by clinician/patient-rated severity and patient-rated impact further confirmed that patients with possible TD had worse physical wellness and social functioning than those with no abnormal involuntary movements (Table 3). When adjusted for age, sex, health status, severity of psychiatric condition, functional status, and psychiatric diagnosis, linear regressions based on mean score differences between cohorts indicated that the largest decrements in EQ-5D-5L utility were in Cohort 2A patients with “a lot” of self-reported impact (adjusted mean score difference relative to modified Cohort 1: − 0.121, P < 0.001) or “a lot” of self-reported severity (difference: − 0.089, P < 0.001). The worst SDS total scores were also found in aware patients who self-reported “a lot” of self-reported impact (adjusted mean score difference: 5.401, P < 0.001) or “a lot” of self-reported severity (difference: 3.288, P < 0.01).
Association between severity/impact of possible TD and EQ-5D-5L utility score
Linear regression analyses conducted in Cohorts 2, 2A, and 2NA indicated no statistical significance for the association between clinician-rated severity and EQ-5D-5L utility in any body region, regardless of whether patients were aware or not aware of their possible TD (Additional file 1: Appendix Table S3).
In contrast, statistical significance was found for the association between patient-rated severity of possible TD and EQ-5D-5L utility in patients who were aware of their possible TD (Cohort 2A) (Table 4). After adjustments for age, sex, health status, severity of psychiatric condition, functional status, and psychiatric diagnosis, the regression coefficient for overall patient-rated severity (summary score based on all body regions) was − 0.028 (P < 0.05). Similar coefficients were found for severity in the head/face (− 0.026) and upper extremities (− 0.032), indicating comparable magnitudes of effect on EQ-5D-5L utility; however, these results were not statistically significant. Patient-rated severity in the lower extremities had the largest association with EQ-5D-5L utility, with an adjusted regression coefficient of − 0.060 (P < 0.05).
Patient-rated impact (not severity) of possible TD had the greatest association with EQ-5D-5L utility (Table 4). After adjustments, regression coefficients (unstandardized) indicated that for Cohort 2A, EQ-5D-5L was significantly associated with the patient-rated impact of TD on 5 activities (self-care, usual activities, being productive, socializing, eating) and on overall patient-reported impact (summary score) (P < 0.05 for all coefficients). Since the EQ-5D-5L utility is scored from 0 to 1, and unstandardized coefficients reflect the underlying metric of the scale, the coefficients presented in Table 4 can be interpreted as percentage changes in overall quality of life due to the specific impacts.
Association between severity/impact of possible TD and SDS total score
Similar to EQ-5D-5L utility, no statistically significant associations were found between clinician-rated severity and SDS total score in any body region (Additional file 1: Appendix Table S3). After adjustments, the regression coefficient for overall patient-rated severity (summary score based on all body regions) was 0.725 (P > 0.05) in Cohort 2A, with severity in head/neck having the greatest effect on SDS total score (Table 4). Although coefficients based on patient-rated severity were not statistically significant, the positive coefficients for SDS indicate a positive association with patient-rated severity, suggesting that higher severity was associated with worse disability.
As with EQ-5D-5L utility, patient-rated impact of possible TD had the largest association with SDS total score (Table 4). In Cohort 2A, SDS total score was significantly associated with the patient-rated impact of TD on 4 activities (self-care, usual activities, being productive, socializing) and on overall impact (summary score) (P < 0.05 for all coefficients).
Discussion
In these analyses of RE-KINECT data, which were conducted to address the need for quantitative evaluations of HRQoL in patients with TD, patient-reported outcomes (PROs) regarding health and physical wellness (EQ-5D-5L) and social functioning (SDS) were generally worse in patients with clinician-confirmed possible TD (Cohort 2) than in those with no abnormal involuntary movements (modified Cohort 1). Cohort 2 was characterized by older age, more male patients, a higher prevalence of schizophrenia or schizoaffective disorder, greater severity of psychiatric conditions, worse overall health, and less ability to work/study than modified Cohort 1. After adjusting for these potentially confounding factors, analyses of patient-reported EQ-5D-5L and SDS mean scores indicated that wellness and functioning were most negatively affected in patients who were aware of their possible TD symptoms and rated those symptoms as having “a lot” of severity in ≥ 1 body region or “a lot” of impact on ≥ 1 daily activity. These findings were consistent with results from regression analyses, which showed that patient-reported impact had the largest (and mostly significant) effects on EQ-5D-5L utility and SDS total scores, followed by patient-reported severity. In contrast, no statistically significant associations were found between clinician-rated severity and the PRO measures (EQ-5D-5L utility and SDS total scores).
Among patients who were aware of their possible TD (Cohort 2A), self-reported severity (“some” or “a lot”) was associated with standard PROs (EQ-5D-5L utility, SDS total). These results highlight some important points about awareness and physical/social impact that should be considered when diagnosing and treating patients with TD. Severity terms like “mild” and “severe” (or “some” and “a lot”) are inherently subjective, and patients who are aware of their TD might find even “milder” symptoms to be disruptive, debilitating, or embarrassing. For optimal diagnosis and treatment, it may be important to assess patients’ awareness of their own symptoms and the impact of these symptoms on overall wellness and ability to function—whether self-reported using a formal instrument (EQ-5D-5L, SDS) or simple descriptors (“none”, “some”, or “a lot” of impact). Clinician ratings of TD severity were less likely to be associated with EQ-5D-5L and SDS scores, underscoring the importance of assessing the patient’s perspective.
Assessing the detrimental effects of TD on patients’ physical wellness and social functioning was one of the challenges in this analysis. In a cross-sectional study such as RE-KINECT, assessing causality (i.e., the additive effects of TD) can be hypothesized but not concluded. The analysis might have also been limited by potential overlapping characteristics between modified Cohort 1 (patients with no abnormal involuntary movements) and Cohort 2 (patients with possible TD) due to the fact that patients in both cohorts had been treated with an antipsychotic for ≥ 3 months, primarily for serious mental illnesses such as schizophrenia/schizoaffective disorder, bipolar disorder, and major depressive disorder. Given the functional decrements associated with these conditions [25,26,27], it was anticipated that both physical and social wellbeing would be diminished in all RE-KINECT patients regardless of cohort. Nevertheless and importantly, EQ-5D-5L and SDS scores were consistently worse in Cohort 2 than in modified Cohort 1 as expected (Tables 2 and 3). However, these differences were not all statistically significant, possibly due to some residual confounding even after adjusting for various patient characteristics (e.g., age, severity of psychiatric condition).
Another objective of this analysis was to explore whether patients who were aware of their possible TD had worse self-reported physical wellness and social functioning than those who were not aware. This appeared to have been the case. Comparisons between Cohort 2A (aware) and Cohort 2NA (not aware) demonstrated similar severity of psychiatric conditions between the cohorts. However, the patients in Cohort 2A (aware) were more likely to self-report poorer overall health (Additional file 1: Appendix Table S1). In addition, Cohort 2A patients had significantly worse SDS scores (total and family/home life), suggesting that patients who are aware of their TD felt a greater negative impact in areas that require interaction with other people. The results also indicate that in patients who are not aware of their TD, it is important to assess the impact of TD on patients’ health using caregiver feedback, including medical issues that the patient might not associate with TD (e.g., pain). Especially in patients with advanced psychotic disorders, family and caregiver accounts of TD movements and impact are valuable in assessing the overall “severity” of the condition.
The emphasis on patients’ experiences in this analysis does not discount the need for regular clinician assessments or caregiver input. Although clinician-rated severity was not significantly associated with EQ-5D-5L or SDS scores, clinician assessment of severity remains a crucial part of the treatment plan for determining the effectiveness of interventions. In patients who are not aware of their symptoms, caregivers can provide information about the location and severity of TD symptoms that are not present during the office visit or are located in areas that are not easily visible, such as the toes. In addition, while patients who reported being unaware of abnormal movements may rate the impacts of TD on functioning as less severe, it remains possible that the abnormal movements could still significantly affect social and occupational relations with others who are distracted by the patient’s movements, regardless of their lack of awareness or denial. Clinician assessments, patient perspectives, and caregiver input should all be considered when evaluating and treating patients with TD.
Limitations for this study have been previously discussed [17]. In brief, it should be noted that RE-KINECT was intended to be a screening study of possible TD with no requirement for “formal” TD diagnoses. Additionally, the cohorts were not matched for any sociodemographic factors, and statistically significant differences between patients with possible TD (Cohort 2) and patients with no abnormal involuntary movements (modified Cohort 1) were found for age, sex, psychiatric conditions, severity of psychiatric conditions, overall health status, and overall functional status (Table 1). However, the adjustments for age, sex, and psychiatric conditions in this analysis should have mitigated some of this variability. It should also be noted that although the EQ-5D-5L and SDS are well-established and standardized instruments (PROs), neither has been specifically validated for TD. However, the strong associations found between EQ-5D-5L/SDS and patient-reported impact of possible TD indicate that both scales may be appropriate for assessing HRQoL in patients with TD. Finally, the ordinal values assigned to various outcomes for the regression analyses (e.g., 0 = "none", 1 = "some", 2 = "a lot") assumes that the qualitative differences between responses are similar, which may or may not be true. The rationale for this approach was to provide a relatively simple method that can be easily reproduced. The findings based on this approach were sufficient to confirm the applicability of a simple impact assessment (“none”, “some”, or “a lot”) to real-world practice.
Conclusions
The results of this analysis indicate that physical wellness and social functioning were diminished in patients with possible TD, particularly in those who were aware of their abnormal involuntary movements and rated those movements as having “a lot” of impact on daily activities. Patient-rated severity and impact of possible TD, but not clinician-rated severity, were significantly associated with EQ-5D-5L utility or SDS total scores. These outcomes suggest that in addition to assessing the presence and severity of patients’ abnormal movements during usual care visits, clinicians or their staff may need to ask patients about how TD adversely affects their HRQoL (particularly as it relates to their daily activities) and consider these impacts when making and evaluating treatment plans. For some patients, such questions may be as important (or possibly even more important) than symptom severity. For patients not aware of their TD, discussion with caregivers may be needed to determine the impact of TD on patients’ health and daily activities. Such discussions might be facilitated by specific and descriptive questions that are specifically focused on social withdrawal (avoiding social interaction or events, isolation, embarrassment) or physical disabilities associated with TD symptoms (difficulty swallowing, breathing, walking, or writing).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the study sponsor (Neurocrine Biosciences, Inc.) upon reasonable request for research purposes.
References
Jain R, Correll CU (2018) Tardive dyskinesia: recognition, patient assessment, and differential diagnosis. J Clin Psychiatry 79(2):nu17034ah17031c
Hauser RA, Meyer JM, Factor SA, Comella CL, Tanner CM, Xavier RM, Caroff SN, Lundt L (2020) Differentiating tardive dyskinesia: a video-based review of antipsyhotic-induced movement disorders in clinical practice. CNS Spectr 27:208–217
Cloud LJ, Zutshi D, Factor SA (2014) Tardive dyskinesia: therapeutic options for an increasingly common disorder. Neurotherapeutics 11(1):166–176
Carbon M, Hsieh CH, Kane JM, Correll CU (2017) Tardive dyskinesia prevalence in the period of second-generation antipsychotic use: a meta-analysis. J Clin Psychiatry 78(3):e264–e278
Citrome L, Saklad SR (2020) Revisiting tardive dyskinesia: focusing on the basics of identification and treatment. J Clin Psychiatry 81:TV18059AH3C
Caroff SN (2020) Recent advances in the pharmacology of tardive dyskinesia. Clin Psychopharmacol Neurosci 18:493–506
Bhidayasiri R, Jitkritsadakul O, Friedman JH, Fahn S (2018) Updating the recommendations for treatment of tardive syndromes: a systematic review of new evidence and practical treatment algorithm. J Neurol Sci 389:67–75
American Psychiatric Association (2020) Practice guideline for the treatment of patients with schizophrenia. Am J Psychiatry. https://doi.org/10.1176/appi.ajp.2020.177901
Caroff SN, Citrome L, Meyer J, Sajatovic M, Goldberg JF, Jain R, Lundt L, Lindenmayer JP, Mcevoy JP, Mcintyre RS, Tohen M, Ketter TA (2020) A modified Delphi consensus study of the screening, diagnosis, and treatment of tardive dyskinesia. J Clin Psychiatry 81:19cs12983
Ascher-Svanum H, Zhu B, Faries D, Peng X, Kinon BJ, Tohen M (2008) Tardive dyskinesia and the 3-year course of schizophrenia: results from a large, prospective, naturalistic study. J Clin Psychiatry 69:1580–1588
Caroff SN, Davis VG, Miller DD, Davis SM, Rosenheck RA, Mcevoy JP, Campbell EC, Saltz BL, Riggio S, Chakos MH, Swartz MS, Keefe RS, Stroup TS, Lieberman JA, Investigators C (2011) Treatment outcomes of patients with tardive dyskinesia and chronic schizophrenia. J Clin Psychiatry 72:295–303
Caroff SN, Leong SH, Roberts C, Berkowitz RM, Campbell EC (2020) Cumulative burden of illness in veterans with tardive dyskinesia and serious mental disorders. J Clin Psychopharmacol 40:38–45
Yassa R, Jones BD (1985) Complications of tardive dyskinesia: a review. Psychosomatics 26:305–313
Strassnig M, Rosenfeld A, Harvey PD (2018) Tardive dyskinesia: motor system impairments, cognition and everyday functioning. CNS Spectr 23:370–377
Carroll B, Irwin DE (2019) Health care resource utilization and costs for patients with tardive dyskinesia. J Manag Care Spec Pharm 25:810–816
Mcevoy J, Gandhi SK, Rizio AA, Maher S, Kosinski M, Bjorner JB, Carroll B (2019) Effect of tardive dyskinesia on quality of life in patients with bipolar disorder, major depressive disorder, and schizophrenia. Qual Life Res 28:3303–3312
Caroff SN, Yeomans K, Lenderking WR, Cutler AJ, Tanner CM, Shalhoub H, Pagé V, Chen J, Franey E, Yonan C (2020) RE-KINECT: a prospective study of the presence and healthcare burden of tardive dyskinesia in clinical practice settings. J Clin Psychopharmacol 40:259–268
Cutler AJ, Caroff SN, Tanner CM, Shalhoub H, Lenderking WR, Pagé V, Franey E, Yonan C (2021) Caregiver burden in RE-KINECT: data from retrospective real-world tardive dyskinesia screening study. J Am Psychiatr Nurses Assoc. https://doi.org/10.1177/10783903211023565
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders: DSM-5. American Psychiatric Association, Arlington
Schooler NR, Kane JM (1982) Research diagnoses for tardive dyskinesia. Arch Gen Psychiatry 39:486–487
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders: DSM-IV-TR. American Psychiatric Association, Arlington
American Psychiatric Association (2022) Diagnostic and statistical manual of mental disorders: DSM-5-TR. American Psychiatric Association, Arlington
Herdman M, Gudex C, Lloyd A, Janssen M, Kind P, Parkin D, Bonsel G, Badia X (2011) Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res 20:1727–1736
Sheehan DV, Harnett-Sheehan K, Raj BA (1996) The measurement of disability. Int Clin Psychopharmacol 11(Suppl 3):89–95
Lenert LA, Sturley AP, Rapaport MH, Chavez S, Mohr PE, Rupnow M (2004) Public preferences for health states with schizophrenia and a mapping function to estimate utilities from positive and negative symptom scale scores. Schizophr Res 71:155–165
Martin-Subero M, Berk L, Dodd S, Kamalesh V, Maes M, Kulkarni J, De Castella A, Fitzgerald PB, Berk M (2014) Quality of life in bipolar and schizoaffective disorder—a naturalistic approach. Compr Psychiatry 55:1540–1545
Porcelli S, Kasper S, Zohar J, Souery D, Montgomery S, Ferentinos P, Rujescu D, Mendlewicz J, Merlo Pich E, Pollentier S, Penninx B, Serretti A (2020) Social dysfunction in mood disorders and schizophrenia: clinical modulators in four independent samples. Prog Neuropsychopharmacol Biol Psychiatry 99:109835
Acknowledgements
Medical writing assistance was provided by Mildred Bahn (Prescott Medical Communications Group; Chicago IL) with support from Neurocrine Biosciences, Inc.
Funding
This study and the data analyses were funded by Neurocrine Biosciences, Inc.
Author information
Authors and Affiliations
Author notes
Huda Shalhoub and Chuck Yonan: At time of the study
- Huda Shalhoub
Contributions
All authors met International Committee of Medical Journal Editors (ICMJE) recommendations for authorship, including critical review of all drafts and approval of the final manuscript for submission.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All patients provided written and informed consent for participation in the study. For all participating study sites, protocol and ethics approvals were provided by Quorum Review IRB (Seattle, WA).
Consent for publication
Not applicable.
Competing interests
CMT receives grants from the Michael J. Fox Foundation, the Parkinson’s Foundation, the Department of Defense, BioElectron, Roche/Genentech, Biogen, and the National Institutes of Health, compensation for serving on Data Monitoring Committees from Biotie Therapeutics, Voyager Therapeutics and Intec Pharma and personal fees for consulting from Neurocrine Biosciences, Adamas Therapeutics, Biogen, 23andMe, Alexza, Grey Matter, Acadia and CNS Ratings. SNC has served as a consultant to Neurocrine Biosciences, Inc., Teva Pharmaceuticals Industries, Ltd., Osmotica Pharmaceuticals and Dispersol Technologies, and received separate research grants from Neurocrine Biosciences, Inc., Pfizer, Inc., and Eagle pharmaceuticals. AJC has served as a consultant to, received research grants from, and is on the speaker bureau for ACADIA, Avanir, Neurocrine, Otsuka and Teva, is a consultant to MedAvante-Prophase, and is on the Board of the Neuroscience Research Institute. WRL is a current full-time employee of Evidera. HS and VP are former employees of Evidera. Evidera employees served as consultants on this work. EGF, MS, and CY are full-time employees of Neurocrine Biosciences, Inc.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1
. Appendix tables and figures.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tanner, C.M., Caroff, S.N., Cutler, A.J. et al. Impact of possible tardive dyskinesia on physical wellness and social functioning: results from the real-world RE-KINECT study. J Patient Rep Outcomes 7, 21 (2023). https://doi.org/10.1186/s41687-023-00551-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41687-023-00551-5