Abstract
Background
Human leukocyte antigen (HLA)-A*03, hemochromatosis ancestral haplotype marker, was associated with greater iron overload in hemochromatosis cohorts reported before discovery of the HFE gene. We sought to learn whether an A*03-linked locus influences phenotypes in referred HFE p.C282Y homozygotes.
Methods
We tabulated these phenotypes in probands with p.C282Y homozygosity: age, transferrin saturation (TS), serum ferritin (SF), conditions related to iron overload, fibrosis-four variables (FIB-4) index and aspartate aminotransferase-to-platelet ratio index (APRI) predictors of severe hepatic fibrosis, and iron removed to achieve depletion (QFe/age). We analyzed phenotypes of men and women separately across three A*03 subgroups.
Results
There were 104 men (57.8%) and 76 women (42.2%). Mean age (SD) was 49 ± 13 y. Mean TS was 79 ± 17%. Median SF (range) was 715 µg/L (28, 6103). Related conditions included: hemochromatosis arthropathy (21.7%); type 2 diabetes (18.9%); hypogonadotropic hypogonadism (5.8% of men); cardiomyopathy (0%); and cirrhosis (10.0%). Median QFe/age was 61 mg/y (0, 714). A*03 homozygosity, heterozygosity, and no A*03 occurred in 37 (20.6%), 104 (57.8%), and 39 probands (21.7%), respectively. In men, mean TS and median SF were significantly higher in A*03 homozygotes than heterozygotes but not A*03-negative probands. In men, median APRI was significantly lower in A*03 heterozygotes than homozygotes and A*03-negative probands. No other phenotypes, including QFe/age, differed significantly across A*03 subgroups in either men or women.
Conclusions
Our results suggest that an A*03-linked locus does not influence phenotypes in referred HFE p.C282Y homozygotes. It is unlikely that heritable factors that modify phenotypes of p.C282Y homozygotes are linked to the hemochromatosis ancestral haplotype.
Similar content being viewed by others
Introduction
Hemochromatosis in whites of western European descent is characterized by elevated transferrin saturation (TS) and serum ferritin (SF) levels and increased risk to develop iron overload and related conditions, including hemochromatosis arthropathy, diabetes mellitus, hypogonadotropic hypogonadism, cardiomyopathy, and hepatic cirrhosis [1]. Severe iron overload due to hemochromatosis occurs predominantly in men [1]. Other heritable and environmental factors modify iron loading in adults with hemochromatosis [1, 2]. Regardless, phenotype variability in hemochromatosis [1, 3] remains largely unexplained.
Human leukocyte antigen (HLA)-A*03 was among the early A series of HLA alleles discovered by Bodmer et al. in 1966 [4] and recognized by the World Health Organization Leukocyte Nomenclature Committee in 1968 [5]. Antisera to A*03 were standardized through subsequent International Histocompatibility Workshops. A*03, a cell-surface protein of 365 amino acids involved in short polypeptide presentation to T-lymphocytes, is encoded by HLA-A (chromosome 6p22.1) [6]. In whites in western European and derivative countries, A*03:01 predominates and A*03:02 is less common [7, 8]. Both alleles react with anti-A*03 serotyping antibodies.
In 1975, Simon and colleagues reported that a factor associated with hemochromatosis in French patients was linked to HLA-A*03 [9, 10]. In 1987, they reported that HLA-A*03, B*07, A*03, B*14, and other A, B haplotypes were in linkage disequilibrium with presumed hemochromatosis alleles, although A*03 alone was independently associated with hemochromatosis [11]. Simon and colleagues hypothesized that the presumed hemochromatosis allele was "a rare if not unique event that produced an ancestral HLA marking that was subsequently modified by recombinations and geographical scattering due to migrations" [11].
In a 1995 study of Australian hemochromatosis patients and their families, Jazwinska and colleagues defined the chromosome 6p hemochromatosis ancestral haplotype (AH) as D6S248(5)-D6S265(1)-HLA-A*03-HLA-F*02-D6S105(8) [12]. Among these alleles, only A*03 was an independent surrogate marker for the AH [12], consistent with the previous report of Simon and colleagues [11]. Australian hemochromatosis patients with two A*03 alleles had greater mean TS and mean hepatic iron indices than hemochromatosis patients with one or no A*03 [13]. In a 1996 study of Alabama hemochromatosis probands, mean age-adjusted units of blood removed to deplete body iron stores was significantly greater in probands with A*03 or D6S105(8) homozygosity. Intermediate values of mean units of blood removed were observed in both A*03 and D6S105(8) heterozygotes [14]. In a 1996 study of Italian men with hemochromatosis, greater amounts of iron were removed by phlebotomy, with or without adjustment for age at diagnosis, in A*03 homozygotes and heterozygotes than those without A*03 [15]. Taken together, these studies [13,14,15] supported the postulate that a locus linked to A*03 influences hemochromatosis phenotypes [16].
In 1996, Feder and colleagues discovered the HFE gene (homeostatic iron regulator, chromosome 6p22.2) and two common HFE missense alleles in patients with hemochromatosis [17]. Among 178 patients, 83% were HFE p.C282Y homozygotes, 16% were p.C282Y/p.H63D compound heterozygotes, and one was a p.H63D homozygote [17]. This discovery confirmed that there is a predominant hemochromatosis allele (p.C282Y) and that combinations of HFE alleles (p.C282Y, p.H63D) on different haplotypes account for hemochromatosis phenotypes. Subsequently, many other pathogenic HFE alleles have been identified in persons with hemochromatosis [18], although it remains unknown whether a locus linked to A*03 influences hemochromatosis phenotypes in p.C282Y homozygotes [16].
To learn more, we performed a retrospective study of 180 consecutive, referred Alabama hemochromatosis probands with HFE p.C282Y homozygosity (104 men, 76 women) who underwent HLA-A typing and achieved iron depletion with phlebotomy. We sought to determine whether A*03 homozygosity, A*03 heterozygosity, or no A*03 is significantly associated with these phenotypes: age, TS, SF, conditions related to iron overload (hemochromatosis arthropathy, diabetes, hypogonadotropic hypogonadism, and cirrhosis) at diagnosis, and age-adjusted quantity of iron removed to achieve depletion (QFe/y, mg/y). We discuss our results in the context of the three studies of patients with hemochromatosis with and without A*03 published before the discovery of HFE [13,14,15]. We review evidence for the existence of a locus linked to A*03 that influences phenotypes in p.C282Y homozygotes.
Methods
Subjects included
We compiled observations on 180 consecutive self-identified non-Hispanic white adults ≥ 18 y referred to an Alabama tertiary center (1990–2020) for evaluation of hemochromatosis who a) were HFE p.C282Y homozygotes, b) underwent HLA-A typing, c) achieved iron depletion with phlebotomy at this center, and d) were the first in their respective families to be diagnosed to have hemochromatosis (probands). Medical histories were taken from probands and records of referring physicians. All probands underwent physical examination, laboratory testing, imaging procedures, and evaluation of liver conditions, as indicated, before therapeutic phlebotomy was initiated.
Subjects excluded
We excluded HFE p.C282Y homozygotes who a) were family members of the present probands, b) presented with iron deficiency (defined as both TS < 10% and SF < 15 µg/L), c) were treated with phlebotomy or iron chelation before referral, or d) had incomplete evaluations. We also excluded probands diagnosed to have hemochromatosis because they participated in the Hemochromatosis and Iron Overload Screening (HEIRS) Study [19, 20] or other screening programs.
Laboratory methods
TS, SF, and serum levels of total testosterone, estradiol, luteinizing hormone (LH), follicle-stimulating hormone (FSH), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) were measured using morning specimens obtained without regard to fasting and standard clinical laboratory methods (Laboratory Corporation of America, Burlington, NC, USA). Platelet counts were measured using automated hematology analyzers (Cell-Dyn® Models 610 and 1700, Abbott Laboratories, Chicago, IL, USA).
HFE allele analyses were performed using standard clinical laboratory methods (Laboratory Corporation of America, Burlington, NC, USA) as previously described [16]. HLA-A phenotypes were determined using the microdroplet lymphocytotoxicity assay [14], HLA Class I SSPARMS-PCR typing kits [21, 22], or sequence-based typing (Laboratory Corporation of America, Burlington, NC, USA). Herein, we use the term A*03 whether A*03 was identified by serotyping or DNA-based typing.
Hemochromatosis ancestral haplotype
We defined HLA-A*03 as the marker of the AH, consistent with previous reports [11, 13,14,15]. We defined A*03 positivity as A*03 homozygosity or heterozygosity. We defined A*03 allele frequency as the quotient of total A*03 alleles by total HLA-A alleles.
Conditions related to iron overload
We defined conditions related to iron overload as hemochromatosis arthropathy, diabetes, hypogonadotropic hypogonadism, cardiomyopathy, and cirrhosis.
Hemochromatosis arthropathy
We defined hemochromatosis hand arthropathy as the combination of tenderness, pain, swelling, or effusion of the second and third metacarpophalangeal joints [3, 23] and radiological features consistent with those observed in hemochromatosis [1] in the absence of other cause(s). We defined hemochromatosis non-hand arthropathy as physical examination and radiological abnormalities in shoulders, elbows, hips, knees, and ankles typical of hemochromatosis [1] in the absence of other cause(s). We classified probands who had either hemochromatosis hand arthropathy, hemochromatosis non-hand arthropathy, or both as having hemochromatosis arthropathy. We did not classify autoimmune or neuropathic arthropathy as hemochromatosis arthropathy. We did not evaluate observations on spine or other arthropathy.
Diabetes
Diabetes was diagnosed by referring physicians and classified according to criteria of the American Diabetes Association [24].
Hypogonadotropic hypogonadism
Serum levels of total testosterone, LH, and FSH were measured at diagnosis of hemochromatosis in men who reported loss of libido or erectile dysfunction due to undefined cause(s) and did not take supplemental androgen(s). Serum levels of estradiol, LH, and FSH were measured at diagnosis of hemochromatosis in women who reported premature cessation of menses not due to menopause or other defined cause(s) and did not take hormone replacement therapy. Hypogonadotropic hypogonadism was defined as the combination of subnormal serum total testosterone levels < 241 ng/dL (< 8.4 nmol/L) (men) or serum estradiol levels < 30 pg/mL (<110 pmol/L) (women) and serum LH and FSH levels that did not exceed the upper reference limits (9.3 IU/L and 18.1 IU/L, respectively), in the absence of other cause(s) [25].
Cardiomyopathy
Hemochromatosis cardiomyopathy was defined as dilated cardiomyopathy with low left ventricular ejection fraction and decreased fractional shortening, with or without complete atrioventricular block or atrial and ventricular tachyarrhythmias, due to cardiac siderosis demonstrated by endomyocardial biopsy or MRI scanning [1, 26].
Cirrhosis
We recommended that probands undergo percutaneous liver biopsy (or not) in accordance with hemochromatosis diagnosis and management guidelines of the American Association for the Study of Liver Diseases [27]. Iron staining in liver specimens was graded 0–4 + as previously described [28]. Hepatic iron was measured using atomic absorption spectrometry. Pathologists interpreted liver specimens and defined cirrhosis as the histological occurrence of regenerating nodules of hepatocytes surrounded by bands of fibrous connective tissue [29]. In some probands, the presence or absence of cirrhosis was ascertained using imaging techniques (CT scanning, abdominal ultrasonography, or elastography) [30].
FIB-4 and APRI
We computed the fibrosis-four variables (FIB-4) index [31] and the AST-to-platelet ratio index (APRI) [32] in all probands. Both are moderately accurate non-invasive indices originally derived to identify patients with hepatitis virus C infection with increased cirrhosis risk. Subsequently, FIB-4 and APRI have been studied in HFE p.C282Y homozygotes [33].
Threshold (or greater) values of each index are associated with increased risk for advanced hepatic fibrosis (stage F3, severe fibrosis with architectural distortion or stage F4, cirrhosis with architectural distortion) [34]. A FIB-4 index > 1.10 identified HFE p.C282Y homozygotes with advanced fibrosis with 80% sensitivity, 80% specificity, and 81% accuracy [33]. APRI values > 0.44 identified p.C282Y homozygotes with advanced hepatic fibrosis with 79% sensitivity, 79% specificity, and 81% accuracy [33].
Iron removed by phlebotomy (QFe)
Iron depletion therapy, defined as the periodic removal of blood to eliminate storage iron, was performed in probands with elevated SF at diagnosis as previously defined (> 300 µg/L, men; SF > 200 µg/L, women) [19]. Iron depletion therapy was complete when SF was ≤ 20 μg/L [35]. QFe was estimated to be 200 mg Fe per unit of blood (450–500 mL) [35]. Herein, we defined QFe to be zero in men with SF ≤ 300 µg/L at diagnosis and women with SF ≤ 200 µg/L at diagnosis and thus such probands did not undergo therapeutic phlebotomy. We defined QFe/age at diagnosis (mg/y) as a measure of iron overload severity.
Statistical analyses
The sample size of 180 probands represented the entirety of referred Alabama probands with hemochromatosis and HFE p.C282Y homozygosity diagnosed during the study interval (1990–2020) whose medical records met all stipulations for inclusion in this study. The data we analyzed are available in an open-access database [36]. Age, TS, and SF at diagnosis and QFe/age data are displayed to the nearest integer. Evaluation of continuous data using d'Agostino’s and Shapiro–Wilk tests revealed that age and TS values were normally distributed. Results are displayed as mean ± 1 standard deviation (SD) and were compared with Student’s t tests or one-way analysis of variance (ANOVA) tests and post-hoc Scheffe’s tests, as appropriate. SF, QFe/age, FIB-4 index, and APRI data were not normally distributed. For APRI computations, we used AST 40 IU/L as the upper limit of normal. For some comparisons, we used these threshold values: FIB-4 > 1.10 and APRI > 0.44 [33]. Results are displayed as median (range) and were compared with either Mann–Whitney U tests or Kruskal–Wallis tests and post-hoc Dunn’s tests, as appropriate.
Proportions for two categories were compared using Fisher's exact test (two-tailed). Proportions for three or four categories were evaluated with Pearson’s Chi-square tests (3 × 2 or 4 × 2 tables) and compared post-hoc using 2 × 2 Fisher’s exact tests (two-tailed), as appropriate. In one analysis, we compared HLA-A*03 positivity and allele frequency in men and women, respectively, grouped by the highest quartile of QFe/age with those grouped by the lowest three quartiles of QFe/age. Values of p < 0.05 were defined as significant. Analyses were performed with Excel 2000® (Microsoft Corp., Redmond, WA, USA), GB-Stat® (2003, Dynamic Microsystems, Inc., Silver Spring, MD, USA), and GraphPad Prism 8® (2018, GraphPad Software, San Diego, CA, USA).
Results
Phenotypes of 180 probands
There were 104 men (57.8%) and 76 women (42.2%). Mean age (SD) at diagnosis of hemochromatosis was 49 ± 13 y. Mean TS was 79 ± 17%. Median SF (range) was 715 µg/L (28, 6103). SF was > 1000 µg/L (1001, 6103) in 65 probands (36.1%; 50 men, 15 women).
Hemochromatosis arthropathy was diagnosed in 39 probands (21.7%). Diabetes was diagnosed in 34 probands (18.9%), each of whom had type 2 diabetes. Hypogonadotropic hypogonadism was diagnosed in six men (6/104, 5.8%) and no women. No proband was diagnosed to have cardiomyopathy due to cardiac siderosis.
Liver specimens were obtained by biopsy in 20 of 65 probands with SF > 1000 µg/L (30.8%) and 41 of 115 probands with SF ≤ 1000 µg/dL (35.7%) (p = 0.6231). The other 45 probands with SF > 1000 µg/L were evaluated for cirrhosis with imaging studies [30]. Altogether, cirrhosis was diagnosed in 18 probands (18/180, 10.0%), 15 of whom (15/18, 83.3%) had SF > 1000 µg/L. In 17 probands with cirrhosis (17/18, 94.4%), the sole or predominant cause of cirrhosis was hepatic iron overload. The other proband with cirrhosis was a woman with SF 387 µg/L and hepatic sarcoidosis. No proband was discovered to have primary liver cancer at diagnosis of hemochromatosis.
Therapeutic phlebotomy to achieve iron depletion was indicated and performed in 178 probands (98.9%). Median QFe/age (range) was 61 mg/y (0, 714).
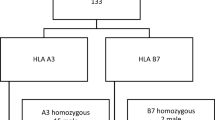
Homozygosity for A*03, heterozygosity for A*03, and no A*03 was detected in 37 probands (20.6%), 104 probands (57.8%), and 39 probands (21.7%), respectively.
Phenotypes of 104 men with hemochromatosis
These results are displayed in Table 1. In men, mean TS and median SF were significantly higher in A*03 homozygotes than heterozygotes but not A*03-negative men. Prevalences of conditions related to iron overload and median QFe/age did not differ significantly across subgroups of men with A*03 homozygosity, A*03 heterozygosity, or no A*03.
FIB-4 indices > 1.10 were observed in 47.6% of A*03 homozygous men, 47.0% of A*03 heterozygous men, and 64.7% of A*03-negative men. Percentages of men with FIB-4 > 1.10 did not differ significantly across A*03 subgroups (p = 0.4147). Median FIB-4 did not differ significantly across A*03 subgroups (Table 1).
APRI > 0.44 was observed in 47.6% of A*03 homozygous men, 22.7% of A*03 heterozygous men, and 52.9% of A*03-negative men. These percentages differed significantly across A*03 subgroups (p = 0.0160). Percentages of men with A*03 homozygosity and A*03-negative men with APRI > 0.44 were significantly greater than the percentage of men with A*03 heterozygosity with APRI > 0.44 (p = 0.0281 and 0.0143, respectively). Median APRI was significantly lower in A*03 heterozygous men than in A*03 homozygous men and A*03-negative men (Table 1).
Phenotypes of 76 women with hemochromatosis
These results are displayed in Table 2. There were no significant differences in phenotypes of women with HLA-A*03 homozygosity, A*03 heterozygosity, or no A*03.
FIB-4 indices > 1.10 were observed in 25.0% of A*03 homozygous women, 36.8% of A*03 heterozygous women, and 45.5% of A*03-negative women. These percentages did not differ significantly across A*03 subgroups (p = 0.4348). Median FIB-4 did not differ significantly across A*03 subgroups.
APRI > 0.44 was observed in 12.5% of A*03 homozygous women, 15.8% of A*03 heterozygous women, and 18.2% of A*03-negative women. These percentages did not differ significantly across A*03 subgroups (p = 0.8936). Median APRI did not differ significantly across A*03 subgroups.
QFe/age grouped by quartiles
These results are displayed in Table 3. Median QFe/age was significantly higher in men and women in the highest QFe/age quartile than men and women with QFe/age in the lowest three quartiles, respectively. Prevalence of HLA-A*03 positivity or A*03 allele frequency did not differ significantly in either men or women according to QFe/age quartile subgroup, respectively.
HLA-A*03 in present and previous hemochromatosis cohorts
HLA-A*03 positivity and allele frequency were highest in the present cohort (Table 4).
Discussion
This study revealed no significant differences in prevalences of conditions related to iron overload at diagnosis and median QFe/age in referred men or women with HFE p.C282Y homozygosity, respectively, across HLA-A*03 homozygosity, A*03 heterozygosity, and no A*03 subgroups. A*03 positivity and allele frequency did not differ significantly in men and women grouped by the highest and lowest three quartiles of QFe/age. Consistent with the present findings, A*03 was not a significant modifier of survival in Alabama hemochromatosis probands with p.C282Y homozygosity [37]. Taken together, these observations demonstrate that A*03, the only known independent marker of the hemochromatosis AH [9, 13,14,15, 38], is not linked to a putative A*03-linked locus that significantly influences iron overload or related conditions in Alabama p.C282Y homozygotes, contrary to previous postulates [13,14,15,16].
Mean TS in the present men was significantly higher in those with HLA-A*03 homozygosity than those with A*03 heterozygosity but not those with no A*03. Mean TS in the present women did not differ significantly across HLA-A*03 homozygosity, A*03 heterozygosity, and no A*03 subgroups. Mean TS did not differ significantly in hemochromatosis probands grouped by A*03 in three previous studies [13,14,15]. In a genome-wide association study of hemochromatosis patients with HFE p.C282Y homozygosity, TS was significantly associated only with the rs3811647 polymorphism in intron 11 of the transferrin gene (TF, chromosome 3q22.1) [39].
Median SF in the present men with HLA-A*03 homozygosity was significantly higher than that of men with A*03 heterozygosity but not men with no A*03. Median SF in the present women did not differ significantly across HLA-A*03 homozygosity, A*03 heterozygosity, and no A*03 subgroups. Median SF was significantly higher in men with hemochromatosis and A*03 heterozygosity in one study [15], although SF was not significantly associated with A*03 in two other studies [13, 14]. In adults unselected for hemochromatosis, numerous non-iron factors influence SF levels [40].
The prevalence of hemochromatosis arthropathy in this study did not differ significantly in either men or women grouped by HLA-A*03 homozygosity, A*03 heterozygosity, and no A*03, consistent with two previous reports [14, 15]. Greater age and SF levels at diagnosis are associated with hemochromatosis arthropathy [41,42,43]. In contrast, hemochromatosis arthropathy is more prevalent and iron overload is less severe in p.C282Y homozygotes than in persons with non-HFE hemochromatosis [44]. A man with HFE p.C282Y homozygosity without iron overload had severe, progressive knee arthropathy typical of hemochromatosis [45]. Taken together, these observations suggest that non-iron factors are also important in the pathogenesis of hemochromatosis arthropathy in p.C282Y homozygotes [44, 46].
Diabetes in the present probands was classified exclusively as type 2 and its prevalence was not significantly different in either men or women grouped by HLA-A*03 homozygosity, A*03 heterozygosity, and no A*03, consistent with two previous reports [14, 15]. In another study of Alabama referred HFE p.C282Y homozygotes, type 2 diabetes was not associated with common HLA types and haplotypes [47]. A consistent association of HLA region genes with type 2 diabetes in subjects not selected for hemochromatosis was not identified in genome-wide association studies [48, 49]. Like the HFE gene [17], loci that are risk factors for type 1 diabetes, defined as autoimmune β-cell destruction and usually leading to absolute insulin deficiency [24], occur in or are linked to the major histocompatibility complex (chromosome 6p21.33) [50, 51], although diverse autoimmune conditions in 236 referred hemochromatosis probands with p.C282Y homozygosity did not include type 1 diabetes [52].
The overall prevalence of hypogonadotropic hypogonadism in men in this study was 5.8%. The prevalence of hypogonadotropic hypogonadism did not differ significantly in men grouped by HLA-A*03 homozygosity, A*03 heterozygosity, and no A*03, consistent with a previous report [15]. In another study of referred patients with hemochromatosis (87% HFE p.C282Y homozygotes), 6.4% of men had hypogonadotropic hypogonadism [25]. Hypogonadotropic hypogonadism was not diagnosed in any of the present women. In another study, 5.2% of women with hemochromatosis had hypogonadotropic hypogonadism [25].
Cardiomyopathy due to cardiac siderosis, not diagnosed in the present probands, occurred in 2.3–14.9% of adult hemochromatosis patients but was not associated with HLA-A*03 in two previous studies [14, 15]. Hemochromatosis cardiomyopathy occurs more often in patients with juvenile-onset hemochromatosis due to homozygosity or compound heterozygosity for pathogenic alleles in the gene that encodes hemojuvelin (HJV, chromosome 1q21.1) [1, 53].
The prevalence of cirrhosis in this study did not differ significantly in either men or women grouped by HLA-A*03 homozygosity, A*03 heterozygosity, and no A*03. Proportions of the present men or women with FIB-4 and APRI values above respective thresholds for increased severe hepatic fibrosis risk and median FIB-4 and APRI values across A*03 subgroups revealed no consistent positive association with A*03. In previous studies, cirrhosis in patients with hemochromatosis was significantly associated with A*03 in one report [15] but not in another [14]. In HFE p.C282Y homozygotes, cirrhosis is positively associated with SF > 1000 µg/L at diagnosis [54, 55] and age at diagnosis, diabetes, amount of daily alcohol intake, and QFe [56].
Iron overload severity defined as QFe/age was not significantly associated with HLA-A*03 in the present study of HFE p.C282Y homozygotes. In three studies published before the discovery of HFE, age-adjusted quantities of iron removed to achieve iron depletion [14, 15] and mean hepatic iron index [13] were significantly greater in hemochromatosis patients with A*03 homozygosity.
Patients with hemochromatosis in three previous studies of HLA-A*03 [13,14,15] and some of the present probands underwent A*03 serotyping. The other present probands underwent DNA-based typing. A*03 is detected equally by serotyping and DNA-based typing. Thus, disparate typing reagents and techniques would not account for differences in A*03 frequencies across the present and three previous hemochromatosis cohorts [13,14,15].
HLA-A*03 allele frequency and positivity were higher in the present cohort than in three previous cohorts [13,14,15]. Proportions of patients with hemochromatosis who have A*03 vary across regions of Europe and derivative countries [11, 13,14,15, 22, 57]. Regardless, geographic differences would not account for the significantly greater A*03 positivity in the present Alabama cohort than in a previous Alabama hemochromatosis cohort from the same clinic [14]. In addition, ancestry reports of white Alabamians [58] and white Australians [59] are similar, although A*03 allele frequency was significantly lower in the previous Australia hemochromatosis cohort [13] than in a previous Alabama [14] and the present Alabama cohorts. Thus, geographic differences in A*03 positivity and allele frequency are an unlikely explanation for disparities observed in the present study and those reported in three previous studies [13,14,15].
We postulate that greater HLA-A*03 positivity and allele frequency in the present cohort of HFE p.C282Y homozygotes is due in part to the inclusion of patients with HFE genotypes other than p.C282Y homozygosity in the three previous hemochromatosis cohort studies published before the discovery of HFE [13,14,15]. Most adults with HFE hemochromatosis who are not p.C282Y homozygotes have HFE p.C282Y/p.H63D [17, 60], p.H63D/p.H63D [17, 61], or p.S65C [62], the third most common pathogenic HFE allele [18]. Whereas p.C282Y is often linked to A*03, p.H63D is often linked to HLA-A*29 [63,64,65]. p.S65C was linked to HLA-A*26 in Spanish subjects heterozygous for p.S65C [64] and to HLA-A*32 in an Alabama patient with hemochromatosis [66]. In addition, iron overload severity is greater, on the average, in patients with hemochromatosis and p.C282Y homozygosity than patients with hemochromatosis and p.C282Y/p.H63D [60], p.H63D/p.H63D [61], and p.S65C genotypes [62].
Proportions of patients diagnosed to have hemochromatosis who were subsequently demonstrated to be HFE p.C282Y homozygotes after the discovery of HFE in 1996 [17] were 52–96% in France (5 reports) [67], 88–100% in Australia (2 reports) [1], 59% in Alabama (1 report) [16], 33–69%% in Italy (2 reports) [1], and a global total of 78% (17 reports) [67]. These observations support our postulate that respective Australia, Alabama, and Italy hemochromatosis cohorts published during the interval 1995–1996 before the discovery of HFE [13,14,15] included patients with HFE genotypes other than p.C282Y homozygosity.
The human major histocompatibility complex (chromosome 6p21.33) is inherited as a single haplotype of closely linked polymorphic genes, comprising Class I loci (including HLA-A, telomeric), Class II loci (centromeric), and Class III loci (including TNF) [68]. TNF encodes a proinflammatory cytokine (tumor necrosis factor-alpha, TNF-α) secreted predominantly by monocytes and macrophages. TNF-α released in vitro by blood monocytes from patients with hemochromatosis was significantly less than that of control subjects in two studies [69, 70], but not in a third similar study [71]. Quantities of iron removed by phlebotomy and hepatic iron indices in referred hemochromatosis patients with p.C282Y homozygosity were significantly higher in those with TNF -308G → A than in those homozygous for TNF -308G, after adjustment for other factors [72]. In contrast, allele frequencies of seven TNF polymorphisms in HEIRS Study participants with p.C282Y homozygosity grouped by high TS/SF and low TS/SF did not differ significantly [73]. Taken together, these observations neither confirm nor deny that either TNF-α or TNF alleles influence(s) iron metabolism significantly in patients with hemochromatosis.
Portuguese hemochromatosis patients with greater iron overload and lower blood CD8 + T-lymphocyte concentrations had a highly conserved ancestral 500 kb microhaplotype on chromosome 6p designated as A-A-T, whereas other patients with milder iron overload and higher blood CD8 + T-lymphocyte concentration had a highly conserved haplotype designated as G-G-G [74]. A subsequent study of patients with hemochromatosis and HFE p.C282Y homozygosity in Portugal, Norway, and Alabama did not confirm that A-A-T is associated with greater iron overload severity [75].
Strengths of this study include a large cohort of referred, unrelated adults with hemochromatosis, HFE p.C282Y homozygosity, and HLA-A typing and a database that includes complete observations on age, TS, SF, conditions related to overload, and QFe/age [36]. The present results do not exclude the possibility that a characteristic of HFE p.C282Y homozygotes not studied herein or a locus linked to pathogenic HFE alleles other than p.C282Y influences hemochromatosis phenotypes. It is unknown which TNF polymorphism(s) is typically linked to A*03 and whether the predominant TNF polymorphism-A*03 linkage is consistent in p.C282Y homozygotes who reside in different geographic regions. It was beyond the scope of this study to classify the severity of hemochromatosis arthropathy, to measure hepcidin levels, or to perform TNF allele typing.
Conclusions
Our results suggest that an A*03-linked locus does not influence phenotypes in referred HFE p.C282Y homozygotes. It is unlikely that heritable factors that modify phenotypes of p.C282Y homozygotes are linked to the hemochromatosis ancestral haplotype.
Availability of data and materials
All supporting data of this article are included in the submitted manuscript or are available at Open Science Framework (https://osf.io/tq8x6/).
Abbreviations
- AH:
-
Ancestral haplotype
- ALT:
-
Alanine aminotransferase
- ANOVA:
-
Analysis of variance
- APRI:
-
AST-to-platelet ratio index
- AST:
-
Aspartate aminotransferase
- FIB-4:
-
Fibrosis-four variables index
- FSH:
-
Follicle-stimulating hormone
- HEIRS Study:
-
Hemochromatosis and Iron Overload Screening Study
- HLA:
-
Human leukocyte antigen
- LH:
-
Luteinizing hormone
- QFe:
-
Quantity of iron removed to achieve iron depletion
- QFE/age:
-
Age-adjusted quantity of iron removed to achieve iron depletion
- SD:
-
Standard deviation
- SF:
-
Serum ferritin
- TNF-α:
-
Tumor necrosis factor-alpha
- TS:
-
Transferrin saturation
References
Edwards CQ, Barton J.C. Hemochromatosis. In: Greer JP, Arber DA, Glader B, List AF, Means RT Jr, Paraskevas F, et al., editors. Wintrobe's Clinical Hematology. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2014. p. 662–681.
Wood MJ, Powell LW, Ramm GA. Environmental and genetic modifiers of the progression to fibrosis and cirrhosis in hemochromatosis. Blood. 2008;111:4456–62.
Allen KJ, Gurrin LC, Constantine CC, Osborne NJ, Delatycki MB, Nicoll AJ, et al. Iron-overload-related disease in HFE hereditary hemochromatosis. N Engl J Med. 2008;358:221–30.
Bodmer J, Bodmer WF, Payne R, Terasaki PI, Vredevoe D. Leucocyte antigens in man: a comparison of lymphocytotoxic and agglutination assays for their detection. Nature. 1966;210:28–31.
Walford R: First Meeting WHO Leukocyte Nomenclature Committee. New York, September, 1968. In: Terasaki PI, editor. History of HLA: Ten Recollections. Los Angeles: UCLA Tissue Typing Laboratory; 1990. p. 123–149.
GeneCards. The Human Gene Database. HLA-A Gene. Weizmann Institute of Science. https://www.genecards.org/cgi-bin/carddisp.pl?gene=HLA-A. Accessed 1 Sept 2021.
Middleton D, Menchaca L, Rood H, Komerofsky R. New allele frequency database. Tissue Antigens. 2003;61:403–7.
Creary LE, Gangavarapu S, Mallempati KC, Montero-Martin G, Caillier SJ, Santaniello A, et al. Next-generation sequencing reveals new information about HLA allele and haplotype diversity in a large European American population. Hum Immunol. 2019;80:807–22.
Simon M, Pawlotsky Y, Bourel M, Fauchet R, Genetet B. Letter: Idiopathic hemochromatosis associated with HL-A 3 tissular antigen. Nouv Presse Med. 1975;4:1432.
Simon M, Bourel M, Genetet B, Fauchet R. Idiopathic hemochromatosis. Demonstration of recessive transmission and early detection by family HLA typing. N Engl J Med. 1977;297:1017–21.
Simon M, Le ML, Fauchet R, Yaouanq J, David V, Edan G, et al. A study of 609 HLA haplotypes marking for the hemochromatosis gene: (1) mapping of the gene near the HLA-A locus and characters required to define a heterozygous population and (2) hypothesis concerning the underlying cause of hemochromatosis-HLA association. Am J Hum Genet. 1987;41:89–105.
Jazwinska EC, Pyper WR, Burt MJ, Francis JL, Goldwurm S, Webb SI, et al. Haplotype analysis in Australian hemochromatosis patients: evidence for a predominant ancestral haplotype exclusively associated with hemochromatosis. Am J Hum Genet. 1995;56:428–33.
Crawford DH, Powell LW, Leggett BA, Francis JS, Fletcher LM, Webb SI, et al. Evidence that the ancestral haplotype in Australian hemochromatosis patients may be associated with a common mutation in the gene. Am J Hum Genet. 1995;57:362–7.
Barton JC, Harmon L, Rivers C, Acton RT. Hemochromatosis: association of severity of iron overload with genetic markers. Blood Cells Mol Dis. 1996;22:195–204.
Piperno A, Arosio C, Fargion S, Roetto A, Nicoli C, Girelli D, et al. The ancestral hemochromatosis haplotype is associated with a severe phenotype expression in Italian patients. Hepatology. 1996;24:43–6.
Barton JC, Shih WW, Sawada-Hirai R, Acton RT, Harmon L, Rivers C, et al. Genetic and clinical description of hemochromatosis probands and heterozygotes: evidence that multiple genes linked to the major histocompatibility complex are responsible for hemochromatosis. Blood Cells Mol Dis. 1997;23:135–45.
Feder JN, Gnirke A, Thomas W, Tsuchihashi Z, Ruddy DA, Basava A, et al. A novel MHC class I-like gene is mutated in patients with hereditary haemochromatosis. Nat Genet. 1996;13:399–408.
Barton JC, Edwards CQ, Acton RT. HFE gene: Structure, function, mutations, and associated iron abnormalities. Gene. 2015;574:179–92.
Adams PC, Reboussin DM, Barton JC, McLaren CE, Eckfeldt JH, McLaren GD, et al. Hemochromatosis and iron-overload screening in a racially diverse population. N Engl J Med. 2005;352:1769–78.
Barton JC, Acton RT, Dawkins FW, Adams PC, Lovato L, Leiendecker-Foster C, et al. Initial screening transferrin saturation values, serum ferritin concentrations, and HFE genotypes in whites and blacks in the Hemochromatosis and Iron Overload Screening Study. Genet Test. 2005;9:231–41.
12th International Histocompabitility Workshop: HLA Class I SSP ARMS-PCR typing kit reference manual, 2nd ed. London: Tissue Antigen Laboratory and Imperial Cancer Research Fund; 1996.
Barton JC, Acton RT. HLA-A and -B alleles and haplotypes in hemochromatosis probands with HFE C282Y homozygosity in central Alabama. BMC Med Genet. 2002;3:9.
McLaren GD, McLaren CE, Adams PC, Barton JC, Reboussin DM, Gordeuk VR, et al. Clinical manifestations of hemochromatosis in HFE C282Y homozygotes identified by screening. Can J Gastroenterol. 2008;22:923–30.
American Diabetes Association: 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2020. Diabetes Care. 2020;43:S14-S31.
McDermott JH, Walsh CH. Hypogonadism in hereditary hemochromatosis. J Clin Endocrinol Metab. 2005;90:2451–5.
Aronow WS. Management of cardiac hemochromatosis. Arch Med Sci. 2018;14:560–8.
Bacon BR, Adams PC, Kowdley KV, Powell LW, Tavill AS. Diagnosis and management of hemochromatosis: 2011 Practice Guideline by the American Association for the Study of Liver Diseases. Hepatology. 2011;54:328–43.
Scheuer PJ, Williams R, Muir AR. Hepatic pathology in relatives of patients with haemochromatosis. J Pathol Bacteriol. 1962;84:53–64.
McCormick PA. Hepatic cirrhosis. In: Dooley JS, Lok ASF, Burroughs AK, Heathcote EJ, editors. Sherlock’s Diseases of the Liver and Biliary System. Chichester: Wiley-Blackwell; 2012. p. 103–20.
Procopet B, Berzigotti A. Diagnosis of cirrhosis and portal hypertension: imaging, non-invasive markers of fibrosis and liver biopsy. Gastroenterol Rep (Oxf). 2017;5:79–89.
Sterling RK, Lissen E, Clumeck N, Sola R, Correa MC, Montaner J, et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43:1317–25.
Lin ZH, Xin YN, Dong QJ, Wang Q, Jiang XJ, Zhan SH, et al. Performance of the aspartate aminotransferase-to-platelet ratio index for the staging of hepatitis C-related fibrosis: an updated meta-analysis. Hepatology. 2011;53:726–36.
Chin J, Powell LW, Ramm LE, Hartel GF, Olynyk JK, Ramm GA. Utility of serum biomarker indices for staging of hepatic fibrosis before and after venesection in patients with hemochromatosis caused by variants in HFE. Clin Gastroenterol Hepatol. 2021;19:1459–68.
Scheuer PJ. Classification of chronic viral hepatitis: a need for reassessment. J Hepatol. 1991;13:372–4.
Adams PC, Barton JC. How I treat hemochromatosis Blood. 2010;116:317–25.
Barton JC, Barton JC, Acton RT. Observations in 180 referred Alabama hemochromatosis probands with HFE p.C282Y homozygosity and HLA-A typing. 12-10-2021. 12-10-2021. https://osf.io/tq8x6/.
Barton JC, Barton JC, Acton RT. Longer survival associated with HLA-A*03, B*14 among 212 hemochromatosis probands with HFE C282Y homozygosity and HLA-A and -B typing and haplotyping. Eur J Haematol. 2010;85:439–47.
Jazwinska E: The ancestral haplotype in hemochromatosis. In: Barton JC, Edwards CQ, editors. Hemochromatosis. Genetics, pathophysiology, diagnosis and treatment. Cambridge: Cambridge University Press; 2000. p. 91–98.
de Tayrac M, Roth MP, Jouanolle AM, Coppin H, le GG, Piperno A, et al. Genome-wide association study identifies TF as a significant modifier gene of iron metabolism in HFE hemochromatosis. J Hepatol. 2015;62:664–72.
McKinnon EJ, Rossi E, Beilby JP, Trinder D, Olynyk JK. Factors that affect serum levels of ferritin in Australian adults and implications for follow-up. Clin Gastroenterol Hepatol. 2014;12:101–8.
Valenti L, Fracanzani AL, Rossi V, Rampini C, Pulixi E, Varenna M, et al. The hand arthropathy of hereditary hemochromatosis is strongly associated with iron overload. J Rheumatol. 2008;35:153–8.
Carroll GJ, Breidahl WH, Bulsara MK, Olynyk JK. Hereditary hemochromatosis is characterized by a clinically definable arthropathy that correlates with iron load. Arthritis Rheum. 2011;63:286–94.
Kolmel S, Nowak A, Krayenbuehl PA. Iron overload associated symptoms and laboratory changes in the Swiss Haemochromatosis Cohort - when a clinician should become attentive. Swiss Med Wkly. 2020;150: w20294.
Sandhu K, Flintoff K, Chatfield MD, Dixon JL, Ramm LE, Ramm GA, et al. Phenotypic analysis of hemochromatosis subtypes reveals variations in severity of iron overload and clinical disease. Blood. 2018;132:101–10.
Chehade S, Adams PC. Severe hemochromatosis arthropathy in the absence of iron overload. Hepatology. 2019;70:1064–5.
Rehman A, Carroll GJ, Powell LW, Ramm LE, Ramm GA, Olynyk JK. Arthropathy in hereditary haemochromatosis segregates with elevated erythrocyte mean corpuscular volume. Scand J Rheumatol. 2021;50:139–42.
Barton JC, Barton JC, Acton RT. Diabetes in first-degree family members: a predictor of type 2 diabetes in 159 nonscreening Alabama hemochromatosis probands with HFE C282Y homozygosity. Diabetes Care. 2014;37:259–66.
Morris AP, Voight BF, Teslovich TM, Ferreira T, Segre AV, Steinthorsdottir V, et al. Large-scale association analysis provides insights into the genetic architecture and pathophysiology of type 2 diabetes. Nat Genet. 2012;44:981–90.
Saxena R, Elbers CC, Guo Y, Peter I, Gaunt TR, Mega JL, et al. Large-scale gene-centric meta-analysis across 39 studies identifies type 2 diabetes loci. Am J Hum Genet. 2012;90:410–25.
Acton RT, Barger BO, Boshell BR, Go RC, Murphy CC, Reitnauer PJ, et al. Epidemiology and genetics of insulin dependent diabetes mellitus in the white and black population from the southeastern United States. In: Hilderman WH, editor., et al., Frontiers in Immunogenetics. New York: Elsevier-North Holland; 1981. p. 239–55.
Barrett JC, Clayton DG, Concannon P, Akolkar B, Cooper JD, Erlich HA, et al. Genome-wide association study and meta-analysis find that over 40 loci affect risk of type 1 diabetes. Nat Genet. 2009;41:703–7.
Barton JC, Barton JC. Autoimmune conditions in 235 hemochromatosis probands with HFE C282Y homozygosity and their first-degree relatives. J Immunol Res. 2015;2015: 453046.
Lee PL, Beutler E, Rao SV, Barton JC. Genetic abnormalities and juvenile hemochromatosis: mutations of the HJV gene encoding hemojuvelin. Blood. 2004;103:4669–71.
Morrison ED, Brandhagen DJ, Phatak PD, Barton JC, Krawitt EL, El-Serag HB, et al. Serum ferritin level predicts advanced hepatic fibrosis among U.S. patients with phenotypic hemochromatosis. Ann Intern Med. 2003;138:627–33.
Barton JC, Barton JC, Acton RT, So J, Chan S, Adams PC. Increased risk of death from iron overload among 422 treated probands with HFE hemochromatosis and serum levels of ferritin greater than 1000 mug/L at diagnosis. Clin Gastroenterol Hepatol. 2012;10:412–6.
Barton JC, McLaren CE, Chen WP, Ramm GA, Anderson GJ, Powell LW, et al. Cirrhosis in hemochromatosis: independent risk factors in 368 HFE p.C282Y homozygotes. Ann Hepatol. 2018;17:871–9.
Yaouanq J: Human leukocyte antigen (HLA) association and typing in hemochromatosis. In: Barton JC, Edwards CQ, editors. Hemochromatosis. Genetics, pathophysiology, diagnosis and treatment. Cambridge: Cambridge University Press; 2000. p. 63–74.
Barton EH, Barton JC, Hollowell WW, Acton RT. Countries of ancestry reported by hemochromatosis probands and control subjects in central Alabama. Ethn Dis. 2004;14:73–81.
1301.0 - Year Book Australia, 1995. Ethnic and cultural diversity in Australia. Australian Bureau of Statistics. https://www.abs.gov.au/Ausstats/abs@.nsf/94713ad445ff1425ca25682000192af2/49f609c83cf34d69ca2569de0025c182!OpenDocument. Accessed 26 Aug 2021.
Gurrin LC, Bertalli NA, Dalton GW, Osborne NJ, Constantine CC, McLaren CE, et al. HFE C282Y/H63D compound heterozygotes are at low risk of hemochromatosis-related morbidity. Hepatology. 2009;50:94–101.
Aguilar-Martinez P, Bismuth M, Picot MC, Thelcide C, Pageaux GP, Blanc F, et al. Variable phenotypic presentation of iron overload in H63D homozygotes: are genetic modifiers the cause? Gut. 2001;48:836–42.
Holmström P, Marmur J, Eggertsen G, Gafvels M, Stal P. Mild iron overload in patients carrying the HFE S65C gene mutation: a retrospective study in patients with suspected iron overload and healthy controls. Gut. 2002;51:723–30.
Porto G, Alves H, Rodrigues P, Cabeda JM, Portal C, Ruivo A, et al. Major histocompatibility complex class I associations in iron overload: evidence for a new link between the HFE H63D mutation, HLA-A29, and non-classical forms of hemochromatosis. Immunogenetics. 1998;47:404–10.
Pacho A, Mancebo E, del Rey MJ, Castro MJ, Oliver D, Garcia-Berciano M, et al. HLA haplotypes associated with hemochromatosis mutations in the Spanish population. BMC Med Genet. 2004;5:25.
Porto G, de Sousa M. Variation of hemochromatosis prevalence and genotype in national groups. In: Barton JC, Edwards CQ, editors. Hemochromatosis. Genetics, pathophysiology, diagnosis and treatment. Cambridge: Cambridge University Press; 2000. p. 51–62.
Barton JC, Sawada-Hirai R, Rothenberg BE, Acton RT. Two novel missense mutations of the HFE gene (I105T and G93R) and identification of the S65C mutation in Alabama hemochromatosis probands. Blood Cells Mol Dis. 1999;25:147–55.
Hanson EH, Imperatore G, Burke W. HFE gene and hereditary hemochromatosis: a HuGE review. Human Genome Epidemiology Am J Epidemiol. 2001;154:193–206.
Choo SY. The HLA system: genetics, immunology, clinical testing, and clinical implications. Yonsei Med J. 2007;48:11–23.
Gordeuk VR, Ballou S, Lozanski G, Brittenham GM. Decreased concentrations of tumor necrosis factor-alpha in supernatants of monocytes from homozygotes for hereditary hemochromatosis. Blood. 1992;79:1855–60.
Fargion S, Valenti L, Dongiovanni P, Scaccabarozzi A, Fracanzani AL, Taioli E, et al. Tumor necrosis factor alpha promoter polymorphisms influence the phenotypic expression of hereditary hemochromatosis. Blood. 2001;97:3707–12.
Distante S, Elmberg M, Foss Haug KB, Ovstebo R, Berg JP, Kierulf P, et al. Tumour necrosis factor alpha and its promoter polymorphisms’ role in the phenotypic expression of hemochromatosis. Scand J Gastroenterol. 2003;38:871–7.
Krayenbuehl PA, Maly FE, Hersberger M, Wiesli P, Himmelmann A, Eid K, et al. Tumor necrosis factor-alpha -308G>A allelic variant modulates iron accumulation in patients with hereditary hemochromatosis. Clin Chem. 2006;52:1552–8.
Acton RT, Barton JC, Leiendecker-Foster C, Zaun C, McLaren CE, Eckfeldt JH. Tumor necrosis factor-alpha promoter variants and iron phenotypes in 785 Hemochromatosis and Iron Overload Screening (HEIRS) Study participants. Blood Cells Mol Dis. 2010;44:252–6.
Cruz E, Whittington C, Krikler SH, Mascarenhas C, Lacerda R, Vieira J, et al. A new 500 kb haplotype associated with high CD8+ T-lymphocyte numbers predicts a less severe expression of hereditary hemochromatosis. BMC Med Genet. 2008;9:97.
Costa M, Cruz E, Barton JC, Thorstensen K, Morais S, da Silva BM, et al. Effects of highly conserved major histocompatibility complex (MHC) extended haplotypes on iron and low CD8+ T lymphocyte phenotypes in HFE C282Y homozygous hemochromatosis patients from three geographically distant areas. PLoS ONE. 2013;8: e79990.
Acknowledgements
Not applicable.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to this work. JaCB and RTA conceived the study. JaCB diagnosed and treated probands. JClB and JaCB compiled proband data and performed statistical analyses. JaCB and RTA performed literature reviews. JaCB, JClB, and RTA wrote the first draft of the manuscript and all authors commented on versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This work was performed according to the principles of the Declaration of Helsinki [43]. Western Institutional Review Board granted an exemption for performance of this study under 45 CFR 46.101(b)(4) (submission 2539985–44189619). Obtaining informed consent was not required because this study involves retrospective chart review and analyses of observations recorded in routine medical care. Data reported in proband-related observations were compiled in a manner that maintained anonymity in the present database and results displayed in this study. This study did not involve the direct analysis of biological specimens, images, or questionnaires.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Barton, J.C., Barton, J.C. & Acton, R.T. HLA-A*03, the hemochromatosis ancestral haplotype, and phenotypes of referred hemochromatosis probands with HFE p.C282Y homozygosity. Hereditas 159, 25 (2022). https://doi.org/10.1186/s41065-022-00237-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41065-022-00237-w