Abstract
Introduction
Exercise therapy is usually prescribed as first-line treatment for lower limb tendinopathies. The multitude of exercise- and non-exercise-based management options can be overwhelming for the treating sports professional and patient alike. We chose to investigate the comparative effectiveness of exercise therapy with or without adjuncts for managing the commonest lower limb tendinopathies.
Methods
Through an extensive systematic literature search using multiple databases, we aimed to identify eligible randomised controlled trials (RCTs) on Achilles tendinopathy, patellar tendinopathy or greater trochanteric pain syndrome (GTPS) that included at least one exercise intervention in their treatment arms. Our primary outcomes were patient-reported pain and function (Victorian Institute of Sport Assessment; VISA). Follow-up was defined as short-term (≤ 12 weeks), mid-term (> 12 weeks to < 12 months) and long-term (≥ 12 months). The risk of bias and strength of evidence were assessed with the Cochrane Collaboration and GRADE-NMA tools, respectively. Analyses were performed separately for each one of the three tendinopathies.
Results
A total of 68 RCTs were included in the systematic review. All pairwise comparisons that demonstrated statistically and clinically significant differences between interventions were based on low or very low strength of evidence. Based on evidence of moderate strength, the addition of extracorporeal shockwave therapy to eccentric exercise in patellar tendinopathy was associated with no short-term benefit in pain or VISA-P. From the network meta-analyses, promising interventions such as slow resistance exercise and therapies administered alongside eccentric exercise, such as topical glyceryl trinitrate for patellar tendinopathy and high-volume injection with corticosteroid for Achilles tendinopathy were based on low/very low strength of evidence.
Conclusion
In this network meta-analysis, we found no convincing evidence that any adjuncts administered on their own or alongside exercise are more effective than exercise alone. Therefore, we recommend that exercise monotherapy continues to be offered as first-line treatment for patients with Achilles and patellar tendinopathies and GTPS for at least 3 months before an adjunct is considered. We provide treatment recommendations for each tendinopathy.
PROSPERO registration number CRD42021289534.
Similar content being viewed by others
Key Points
-
Several treatment modalities are used early on in managing lower limb tendinopathies without sufficient evidence to support their use.
-
Extracorporeal shockwave therapy does not appear to add any pain-relieving or functional benefits when added to eccentric exercise in patellar tendinopathy.
-
Exercise interventions other than eccentric exercise and adjuncts to eccentric exercise appear promising but need further high-quality evidence.
Introduction
Tendinopathy is a pathological state classically characterised by persistent pain and functional impairment due to damaged tendons. The tendon damage is secondary to abnormalities in its microstructure, composition and cellularity, usually as a result of repetitive mechanical overload [1, 2]. The primary aetiology of tendinopathy is linked to an impaired healing response secondary to repetitive tendon stress and overload [1,2,3]. Equally, it has been demonstrated that physical activity alone does not account for the extent of histopathological changes, suggesting that other factors are at the root cause of its aetiology [4]. Psychosocial factors are increasingly recognised to contribute to tendinopathy, and a biopsychosocial approach has been recommended for its treatment, which addresses the biological, psychological and social contributions to chronic pain and disability [5]. The tendons most commonly affected in the lower limb are the Achilles, patellar and gluteal (greater trochanteric pain syndrome; GTPS).
Whilst exercise has been implicated in inducing this condition, over the past 3 decades, strong evidence has demonstrated that rehabilitation with progressive loading improves pain and functional outcomes in tendinopathy [6]. The adaptive potential of tendons secondary to exercise has been demonstrated in several studies and there are multiple variables that may play a role and need to be considered for exercise interventions, such as intensity, load, frequency, muscle contraction type and repetitions [7,8,9]. Recent evidence suggests that loading magnitude and muscle contraction intensity may be more important than muscle contraction type [9].
The management of lower limb tendinopathies is complex. The need for long and often successive treatment modalities poses significant health and economic burden, which in conjunction with the often-debilitating effects of these conditions, make identification of the most effective treatments imperative [10, 11]. A significant proportion of patients do not regain full function after treatment, and chronic symptoms persist in approximately one-quarter of patients [12]. Over recent years, numerous therapeutic modalities have been assessed for treating lower limb tendinopathies, usually in conjunction with exercise therapy. These include, but are not limited to, extracorporeal shockwave therapy (ESWT), orthotics, ultrasound therapy (UST), laser therapy (LT), topical glyceryl trinitrate (GTN) therapy, corticosteroid (CS), platelet-rich plasma (PRP) and high-volume (HVI) injections. Surgery is reserved for treatment-resistant tendinopathy; however, the efficacy of surgical treatment is far from certain [12]. With the plethora of available interventions applied alone or in combination regimes, there is an increasing need for high-quality comparative studies and an overall assessment of the combined evidence that will inform management decisions and protocols.
The aim of the present study was to expand on our previous systematic review of patellar tendinopathy [13] and provide a more comprehensive living systematic review and network meta-analysis comparing the effectiveness of exercise interventions with or without adjunct treatments to other treatments or no treatment in patients with the commonest lower limb tendinopathies (Achilles, patellar tendinopathy and GTPS) with regard to patient-reported pain and function.
Methods
This living systematic review and network meta-analysis (NMA) was prospectively registered on PROSPERO (CRD42021289534), conducted and authored as per PRISMA-NMA” and “PERSiST” guidance [14, 15]. This review was administered at the Institute of Infection, Immunity and Inflammation, University of Glasgow, Scotland, and we plan to update the NMA annually for a minimum of 5 years. We intend to screen the literature annually to identify new eligible data and re-perform analyses where necessary. When new data become available, we will update the analysis and present the updated findings on the website of Glasgow University (https://www.gla.ac.uk/researchinstitutes/iii/staff/nealmillar/). A plain-language summary for patients and clinicians dealing with lower limb tendinopathies will also be provided.
Eligibility Criteria
Types of Studies
Randomised controlled trials (RCTs) of any type that investigated patellar and Achilles tendinopathies and GTPS were eligible for inclusion. Only studies published in English were screened for inclusion.
Types of Participants
Studies of patients with mid-portion Achilles tendinopathy, patellar tendinopathy or GTPS were eligible for inclusion. A clinical diagnosis of Achilles tendinopathy, patellar tendinopathy or GTPS made by a medical professional was required for inclusion with or without radiographic confirmation. Diagnostic criteria for each type of tendinopathy were not controlled for. Duration of symptoms or level of physical activity were not exclusion criteria for the trials. The present study excluded trials with (1) patients with insertional Achilles tendinopathy, (2) trials with participants < 18 years old, (3) partial/complete tendon rupture, (4) previous tendon surgery, and (5) animal or in vitro studies.
Types of Interventions
Studies assessing the effectiveness of any form and duration of exercise therapy with or without adjunct treatments were included. Both supervised and non-supervised exercise programmes were eligible. Any type of intervention that was administered alongside exercise therapy was considered an adjunct treatment.
Types of Comparators
Any type of treatment (exercise or non-exercise-based), placebo/sham treatment or no treatment that was compared to an exercise therapy with or without adjunct treatments was considered as a comparator.
Types of Outcome Measures
The primary outcomes were patient-reported pain and function, measured by the visual analogue scale (VAS) or equivalent (0–10 or 0–100), and the Victorian Institute of Sport Assessment (VISA) scale (0–100), respectively. The VISA questionnaire has been developed into distinct questionnaires to assess pain and functional outcomes in Achilles (VISA-A) and patellar (VISA-P) tendinopathies and GTPS (VISA-G) [16,17,18]. For the purpose of analysis and pooling of results, outcome measures were divided into three distinct intervals, short-term (≤ 12 weeks), mid-term (> 12 weeks to < 12 months) and long-term (≥ 12 months). No secondary outcomes were assessed. Where studies reported results at more than one time point within our pre-specified intervals, those closest to the longer end of the interval were used.
When trials used different types of patient-reported pain, the following hierarchy was used: (a) pain at rest, (b) pain with (any) activity, (c) pain during sports, (d) pain during the day, (e) pain at night, (f) current pain.
Literature Search
Search strategies were developed for each lower limb tendinopathy in “all fields” with the following Boolean operators: (a) ‘(patellar tendin* OR jumper’s knee), (b) ‘(Achilles tendin*), (c) ‘(gluteal tendin* OR greater trochanteric pain syndrome OR GTPS) AND (treatment OR management OR therapy OR intervention OR shockwave OR exercise OR physiotherapy OR loading OR eccentric OR concentric OR platelet-rich plasma OR PRP OR glyceryl trinitrate OR GTN OR *steroid OR glucocorticoid OR injection OR laser OR acupuncture OR orthotics)’.
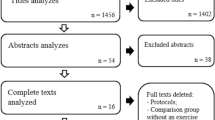
The following databases were screened for published and unpublished trials from inception to 15/03/22 by a single author: Medline, Embase, PubMed, Cochrane Central, Scopus, CINAHL SPORTDiscus, OpenGrey.eu and WorldCat.org. For unpublished or ongoing studies, we searched the WHO International Clinical Trials Registry Platform (http://apps.who.int/trialsearch/) Clinical Trials.gov, The European Union Clinical Trials Register and the ISRCTN registry. All eligible studies' reference and citation lists were screened for further eligible trials. The PRISMA flow-chart is illustrated in Fig. 1.
Data Extraction
Patient characteristics, duration of symptoms, nature of the therapeutic interventions, exercise programme description, outcome measures, and follow-up time points were extracted from individual trials and recorded in Microsoft Word version 16.43 (Microsoft Corporation) by two authors (GC and DC) in a previously constructed data extraction table. Additional file 1 was utilised where available for descriptive statistics. For missing data, attempts were made to contact the corresponding authors of studies published in the last 5 years, and if these were unsuccessful, the papers were excluded.
Data Handling: Synthesis of Results
Comparisons of interventions reported by two or more studies at similar follow-up time points were pooled quantitatively by pairwise meta-analyses in the absence of significant clinical heterogeneity (similar populations, follow-up time points and interventions). Only studies of patients with the same type of tendinopathy were pooled. Raw mean differences (MD) with their accompanying 95% confidence interval (CI) were calculated and used in the tests as the tools used across studies were the same. Finally, a network meta-analysis was conducted for both outcome measures (pain VAS and VISA) at each follow-up period where adequate data existed.
For the present article, “Achilles tendinopathy” refers to non-insertional Achilles tendinopathy only.
Risk of Bias and Strength of Evidence Assessment
The Cochrane Internal Validity Tool was used to assess the risk of bias for each RCT [19]. Studies were assessed by two authors separately (GC and DC), and disagreements were resolved with the involvement of the senior author (NLM). The overall risk of bias for each RCT was labelled as low or high based on how likely the assessor thought that the presence of high risk of bias in the individual domains was to affect the true outcomes of the assessed interventions. We avoided using pre-specified criteria (e.g. overall “high risk” if specific domains of the tool or a certain number of them were of high risk of bias for each study) as we believe that the assessor’s judgement was more fair and accurate for determining the overall risk of bias.
The Grading of Recommendations Assessment, Development and Evaluation for network meta-analysis (GRADE-NMA) was used to appraise the strength (certainty) of evidence [20]. For pairwise meta-analyses, the strength of evidence was assessed based on four domains: overall risk of bias, imprecision, inconsistency and other confounding factors (including publication bias). The result of each comparison of interventions arising from the pooling of similar studies was assigned one of high, moderate, low or very low strength of evidence. This process was completed independently by two authors (GC and DC) for each outcome measure, and disagreements were resolved by the involvement of the senior author (NLM). For network meta-analyses, the strength of both the direct and the indirect evidence was taken into account as per GRADE-NMA, along with intransitivity (clinical heterogeneity of studies participating in direct versus those participating in indirect evidence) and similarly to pairwise meta-analyses, the results were assigned one of the aforementioned four strengths of evidence. Recommendations for clinical practice were given only based on results of high or moderate strength of evidence. An intervention was thought to be more effective than another intervention when its superiority was based on both statistical and clinical significance.
For network meta-analyses, the strength of evidence of the direct estimate was rated first using the overall risk of bias, inconsistency (statistical heterogeneity), indirectness (clinical heterogeneity) and publication bias (GRADE-NMA tool). Subsequently, the indirect estimate was rated using the lowest of the ratings of the two direct comparisons forming the most dominant first-order loops and intransitivity (differences in study characteristics of studies used in indirect comparisons). Finally, the network estimate was rated using the highest certainty of evidence between direct and indirect estimates, the incoherence (difference between direct and indirect comparisons—assessed using the “node splitting” approach) and imprecision.
Statistical Analysis
The Review Manager V.5 (RevMan) software was used to calculate the pooled mean difference and generate forest plots for pairwise meta-analyses and their accompanying heterogeneity tests (Chi2 and I2). STATA 16.1 with Ian White’s extension (multivariate random-effects meta-regression) was used for network meta-analyses (frequentist approach) [21]. When exact numerical mean, mean difference or standard deviation (SD) values were not recorded in the individual papers, an estimated value was extrapolated from available graphs. When the results were recorded as mean difference and interquartile range (IQRs), the SD value was derived as IQR divided by 1.35. Where median values were reported, the mean was assumed to be the same. The RevMan software was used to generate SD values when only confidence intervals were recorded in trials. In studies in which only mean values were presented without SDs, the prognostic method described by Ma et al. [22] was used to generate an SD by calculating the mean of all the other SDs in that comparison.
Pooled SDs were calculated with the following formula:
where SD = standard deviation, n = sample size, k = number of samples.
The following formula was used for the sample size calculation as part of GRADE’s assessment for imprecision:
where N = the sample size required in each of the groups (optimal information size), x = minimal clinically relevant difference (MCRD); defined as 1.5 points for pain VAS and 13 points for VISA, SD2 = population variance (calculated using pooled SD from included treatment groups), a = 1.96 (for 5% type I error), b = 0.842 (for 80% power).
The optimal information size (minimum number of overall patients combined in each meta-analysis for sufficient “precision” in the GRADE assessment) with the use of the above formula was calculated as N = 34 patients for pain and N = 20 for VISA.
Potential publication bias was not assessed as no pairwise meta-analyses included more than 10 studies. Expecting wide-range variability in studies’ settings, a random-effects model was employed in all meta-analyses. Where heterogeneity was found to be high (I2 = 50–75%), the strength of evidence was downgraded by one level; where it was found to be substantial (I2 > 75%), it was downgraded by two levels.
Subgroup and sensitivity analyses were not performed as all pairwise meta-analyses had a maximum of only three RCTs.
Protocol Deviations
There were no deviations from the pre-defined protocol.
Definition of Loading Types
Isotonic—eccentric exercise types involving muscle contractions which result in movement of a limb and lengthening of the muscle unit.
Isotonic—concentric exercise types involving muscle contractions which result in movement of a limb and shortening of the muscle unit.
Isometric exercise types involving muscle contractions which result in no movement of a limb. The muscle unit may shorten marginally as the tendon elongates whilst under a contraction.
Slow resistance slow, isotonic exercises utilising both eccentric and concentric phases. Depending on the load used relative to repetition maximum (maximum load an individual can use for a defined number of repetitions), this can be “heavy” or “moderate” slow resistance.
Results
A total of 2834 studies were initially identified from the searches. An additional four studies were identified from other sources. After removing duplicate results and non-eligible study types, the titles (n = 2102), abstracts (n = 1487) and full texts (n = 77) of the remaining studies were screened for inclusion by two authors independently. A total of 68 studies [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90] were found to be eligible and were included in the systematic review, 31 assessing interventions in Achilles tendinopathy (n = 1792 patients, mean age 46 years), 31 in patellar tendinopathy (n = 1109 patients, mean age 28 years) and 6 in GTPS (n = 907 patients, mean age 55 years). The majority of outcome measures were assessed at short- and mid-term follow-up. Five (5) studies assessed immediate post-intervention outcomes only [31, 44, 63, 64, 82]. Patient and trial characteristics (Additional file 1: Tables S1–S3), risk of bias assessment results (Additional file 1: Tables S4–S6), strength of evidence assessment (Additional file 1: Table S7) and the tabulated results of the compared interventions (Additional file 1: Tables S8–S10) can be found in the supplementary material. The most common type of pain reported was “with single decline squat” in patellar tendinopathy (7 studies), “at rest” in Achilles tendinopathy (11 studies), and “over the last week” (5 studies) for GTPS.
Pairwise Meta-analyses
The pairwise comparisons of interventions are presented below for each tendinopathy type. The forest plots of results based on moderate or high strength of evidence are shown in Fig. 2, whilst those of low or very low strength of evidence can be found in the Additional file 1: Figures S1–S4. For each result, the accompanying parentheses show the MD of the pairwise comparison with its confidence interval and heterogeneity test and the number of pooled studies.
Achilles Tendinopathy
Eccentric Exercise Plus PRP Injection Versus Eccentric Exercise Plus Placebo
Short-term and Mid-term follow-up Based on evidence of low strength, there was no difference in VISA-A between the eccentric exercise plus PRP injection group and the eccentric exercise plus placebo injection groups at short-term follow-up [MD 1.52, 95%CI (− 2.49, 5.53), I2 = 5%, P = 0.46, 3 studies] (Additional file 1: Figure S1a). At mid-term follow-up, there was a difference in VISA-A in favour of the eccentric plus PRP injection group when compared to the eccentric group plus placebo injection based on low strength of evidence, though not likely to be clinically significant [MD 5.05, 95% CI (1.45, 8.64), I2 = 0%, P = 0.006, 3 studies] (Additional file 1: Figure S1b).
Eccentric Exercise Plus Low-Level Laser Therapy Versus Eccentric Exercise Plus Placebo
Short-term follow-up For VISA-A, we found very low strength evidence for a difference in favour of the eccentric exercise plus low-level LT when compared to eccentric exercise plus placebo at short-term follow-up, which is likely not clinically significant [MD 6.29, 95% CI (1.72, 10.85), I2 = 85%, P = 0.007, very low strength, 2 studies] (Additional file 1: Figure S2).
Greater Trochanteric Pain Syndrome
Corticosteroid Injection Versus Exercise Therapy
Short-term and long-term follow-up We found low strength evidence for a difference in pain VAS scores favouring CS injection when compared to exercise therapy (mixed types) in patients with GTPS at short-term follow-up; this is likely to be clinically significant [MD 1.57, 95% CI (1.1. 2.1), I2 = 94%, P = 0.01, 2 studies] (Additional file 1: Figure S3a). There was no difference in reported pain VAS scores between the groups at long-term follow-up based on evidence of low strength [MD − 0.7, 95% CI (− 0.6, 0.45), I2 = 97%, P = 0.78, 2 studies] (Additional file 1: Figure S3b).
Patellar Tendinopathy
Eccentric Exercise Plus ESWT Versus Eccentric Exercise Plus Placebo
Short-term Based on moderate strength evidence, there was no difference in short-term pain VAS or VISA-P between eccentric exercise plus ESWT and eccentric exercise plus placebo ESWT [MD − 0.09, 95% CI (− 1, 0.84), I2 = 0%, P = 0.84, moderate strength, 2 studies; MD − 1.8, 95% CI (− 8, 4.4), I2 = 0%, P = 0.57, 2 studies, respectively] (Fig. 2).
Isotonic Exercise Versus Isometric Exercise
Short-term follow-up We found low strength evidence for no differences in pain VAS immediately post-intervention with isometric versus isotonic exercise in patients with patellar tendinopathy [MD 1.03, 95% CI (− 2.57, 0.5), I2 = 70%, P = 0.19, 3 studies] (Additional file 1: Figure S4).
Network Meta-analysis
The network maps, network forest plots and comparative treatment class effects deriving from all performed network meta-analyses can be found in the Additional file 1: Figures S5–S10. Figure 3 illustrates the median treatment ranks for each tendinopathy for pain VAS and VISA at each follow-up time intervals where data were available. A total of 17 and 19 interventions participated in the network meta-analyses for Achilles and patellar tendinopathy, respectively. Data were insufficient for network meta-analyses in GTPS.
Median ranks of the effectiveness of interventions included in the network meta-analysis for a short-term VISA-A in Achilles tendinopathy. b mid-term VISA-A in Achilles tendinopathy, c for long-term VISA-A in Achilles tendinopathy and d short-term pain VAS in Achilles tendinopathy. PRP platelet-rich plasma, HVI high volume injection, MVCS mucopolysaccharides, type I collagen injection plus vitamin supplementation, ABI autologous blood injection, CS corticosteroid
Achilles Tendinopathy
Short-term follow-up Eccentric exercise plus HVI (with CS) had the highest probability of being the most effective treatment for improving VISA-A and pain VAS scores (probability 56% and 45%, respectively). Passive stretching plus a dietary supplement containing mucopolysaccharides, type I collagen and vitamin C (MCVC) and eccentric exercise plus HVI (without CS) had the highest probability of being the least effective treatment for VISA-A (probability 22% and 46%, respectively) (Fig. 3a, b).
Mid-term follow-up Eccentric exercise plus HVI (with CS) had the highest probability (83%) of being the most effective treatment for improving VISA-A, whilst eccentric exercise plus MCVC dietary supplement had the highest probability (42%) of being the least effective treatment for improving VISA-A (Fig. 3c).
Long-term follow-up Eccentric exercise plus prolotherapy had the highest probability (55%) of being the most effective treatment for improving VISA-A scores, closely followed by eccentric exercise plus HVI (with CS) and heavy slow resistance exercise. Combination treatment with low-level LT plus eccentric exercise had the highest probability of being the least effective treatment (44%) (Fig. 3d).
Patellar Tendinopathy
Short-term follow-up For VISA-P, combination therapy with eccentric exercise plus hyaluronic acid injections had the highest probability (75%) of being the most effective intervention, whilst concentric exercise had the highest probability (42%) of being the least effective intervention (Fig. 4a). For pain VAS, slow resistance exercise of moderate load had the highest probability (22%) of being the most effective treatment, closely followed by eccentric exercise plus topical GTN, whilst concentric exercise had the highest probability (48%) of being the least effective treatment (Fig. 4b).
Median ranks of the effectiveness of interventions included in the network meta-analysis for a short-term VISA-P in patellar tendinopathy and b short-term pain VAS in patellar tendinopathy. PRP platelet-rich plasma, GTN glyceryl trinitrate, rESWT radial extracorporeal shock wave therapy, fESWT focal extracorporeal shock wave therapy
Mid-term and long-term follow-up Data were insufficient for network meta-analyses in patellar tendinopathy at these follow-up time intervals.
Network Meta-analyses Strength of Evidence
For both Achilles and patellar tendinopathy, all results arising from network meta-analyses presented both in text and in figures are considered as of low/very low strength of evidence due to the high overall risk of bias of the included studies and intransitivity.
Discussion
The mainstay of treatment for lower limb tendinopathies is non-operative. Exercise therapy for at least 3 months results in an improvement in function and pain for the majority of patients [3, 6, 91]. Amongst different exercise programmes, eccentric exercise has been thought to be the gold standard management for patellar and Achilles tendinopathy and is usually prescribed as first-line treatment. The effectiveness of eccentric loading for these two tendinopathies is embraced by the research community; the vast majority of non-exercise interventions being assessed were administered alongside eccentric exercise. As a result, conclusions on the effectiveness of these interventions as monotherapies cannot be made. They should always be prescribed as an adjunct to a continuous exercise programme, which should constitute the main focus of the patient’s management. The safety and good tolerability of eccentric exercise make it an even more attractive option. However, it relies heavily on patient compliance, especially where financial constraints at an individual or healthcare system level preclude long-term supervised programmes [81]. For GTPS, eccentric loading on its own is not recommended as the gluteal tendons have short excursion and eccentric exercises alone would be very challenging.
Our study includes all eligible evidence on the comparative effectiveness of exercise interventions for patellar tendinopathy, Achilles tendinopathy and GTPS with or without adjuncts. Our only result which can be used for strong practice recommendations is the lack of benefit on pain or VISA-P of ESWT added to eccentric exercise for patellar tendinopathy (moderate strength of evidence)—therefore, we do not recommend its use. The remaining results were of low or very low strength of evidence. In the following few paragraphs, we discuss promising interventions arising from our findings for each lower limb tendinopathy separately and also summarise the results of other similar systematic reviews and relevant evidence. Based on these, we provide our treatment recommendations for each tendinopathy.
Achilles Tendinopathy
For Achilles tendinopathy, there were no results of comparisons of interventions that were based on moderate or high strength of evidence. Based on evidence of low strength from our pairwise meta-analyses, PRP injections added to eccentric exercise were associated with no short-term benefit and a small, non-clinically significant mid-term benefit in VISA-A. Similarly, low-level laser therapy was associated with a small clinically insignificant short-term benefit in VISA-A when added to eccentric exercise.
From the network meta-analyses, HVI with CS combined with eccentric exercise ranked first in the network meta-analysis for short-term pain VAS and VISA-A, and it also ranked first for mid-term VISA-A improvement. Long-term VISA-A increases were likely to be the highest in patients receiving prolotherapy and eccentric exercise; however, only a single study contributed data for this treatment group. Heavy slow resistance exercise as monotherapy also ranked very highly (second) for mid-term and long-term VISA-A improvements and the single RCT that compared it to eccentric exercise found no differences at 12 weeks or 1 year for pain or VISA-A [28]. In the included RCTs, HVI with CS alongside eccentric exercise consistently demonstrated positive results, whilst those of HVI without CS combined with eccentric exercise were conflicting [30, 81]. It is not impossible that the clinical benefits of HVI with CS may be related to the CS alone, as demonstrated by a recent RCT that found that, added to heavy slow resistance exercise, US-guided CS injection was superior than a placebo injection for VISA-A in the short-, mid- and long-term [92]. HVI was administered under US guidance in all studies, and finally, HVI with or without CS was not associated with any complications; however, the very small (theoretical) risk of tendon rupture associated with locally-administered CS should always be taken into account and communicated to patients [81]. Finally, the limited published evidence on the effectiveness of isometric loading for immediate post-intervention pain relief in Achilles tendinopathy does not support its use [82, 93].
In their similar systematic review and network meta-analysis on Achilles tendinopathy, van der Vlist et al. [94] found no convincing evidence for the superiority of any interventions over others based on the lack of adequate strength of evidence, which is in agreement with our results. As a conclusion, they recommend a calf-muscle exercise programme to be prescribed as first-line treatment as it is easy, widely available, safe and cheap [94]. Another network meta-analysis by Rhim et al. [95] concluded that the addition of HVI with CS and ESWT to eccentric exercise could potentially improve long-term outcomes which is partly in agreement with our recommendations.
We recommend the use of a progressive eccentric or heavy slow resistance exercise for 12 weeks as first-line treatment for Achilles tendinopathy. For resistant cases (no or minimal improvement at the end of the programme), a US-guided HVI with CS could be added to the continuing exercise programme if available.
Patellar Tendinopathy
Pairwise meta-analyses found no interventions that were superior to others for patellar tendinopathy. For short-term pain relief, promising interventions from the network meta-analyses included slow resistance exercise of moderate load and eccentric exercise combined with topical GTN. For short-term VISA-P improvements, eccentric exercise combined with hyaluronic acid injections ranked the highest. Only single studies contributed data to the networks for each of these promising interventions; therefore, clinical practice recommendations for their use can only be weak at this point. No complications were reported with the use of hyaluronic acid injections in the included studies, whilst topical GTN may cause headaches [47, 96]. In the relevant included RCT, two hyaluronic acid injections were administered under US guidance 1 week apart, whilst three injections 1 week apart have also been used successfully for Achilles and patellar tendinopathy elsewhere [47, 97, 98]. Topical GTN was used as one patch (5 mg) daily for 12 weeks in the included RCT [71]. Heavy slow resistance exercise was as effective as eccentric exercise up to 6 months of follow-up in one of the included RCTs and in another RCT slow resistance exercise of moderate load (55% of one repetition maximum) was no less effective than that of heavy load (90% of one repetition maximum) [24, 50]. There was adequate evidence (moderate strength) for strong clinical practice recommendations to suggest that ESWT does not appear to add any benefits when used with eccentric exercise for either pain or VISA-P in patellar tendinopathy.
Isometric exercise has been suggested for immediate pain reduction in patellar tendinopathy as isotonic loading can be painful [63]. Three RCTs compared a bout of isometric versus isotonic muscle contractions for immediate post-intervention pain relief in young athletes and reported conflicting results [44, 63, 64]. Our pairwise meta-analysis found a difference of 1 VAS point favouring isometric loading, which however did not reach clinical or statistical significance (low strength of evidence). Mid- and long-term follow-up results for the effectiveness of isometric exercises on patellar tendinopathy do not exist from RCTs.
In a recent systematic review and network meta-analysis, Chen et al. [99] found no benefits of adding ESWT to eccentric exercise in their pairwise meta-analyses, which is in agreement with our findings. Their network meta-analysis found that dry needling and PRP injections were the highest-ranked interventions. However, their network methodology may have been flawed as some of the studies were not in the network loop (i.e. not all studies had common comparators). Finally, in a previous systematic review of RCTs, topical GTN was superior to placebo in the management of tendinopathy (all types combined); however, its side effects, especially headaches, should be considered and explained to patients [96].
We recommend the use of a progressive eccentric exercise or slow resistance exercise (of “moderate” or “heavy” load) for 12 weeks as first-line treatment for patellar tendinopathy. For resistant cases (no or minimal improvement by the end of the programme), a course of topical GTN or a course of 2–3 US-guided hyaluronic acid injections could be added to the exercise regime depending on availability, and the patient’s and physician’s preference. Where isotonic exercise is not tolerated due to pain, isometric exercise may be used in the initial stages of the treatment regime. ESWT is not recommended for patellar tendinopathy.
Greater Trochanteric Pain Syndrome
Gluteal tendinopathy, which has been renamed as the more generic term “GTPS”, has not received as much attention in the literature as the other two lower limb tendinopathies. This is evident from our inability to perform a network meta-analysis due to insufficient data. Our pairwise meta-analysis results (based on very low strength of evidence) suggest that, for short-term pain relief, a CS injection may be superior to exercise therapy (mixed types). These benefits were likely to be clinically significant. The short-term benefits of CS did not last in the mid-term or long-term. In a RCT that included a CS injection, a ESWT and a home exercise treatment group found that the CS injection group had lower pain scores 1 month after treatment but the reverse was true 15 months after treatment [68]. Also, a significantly larger proportion of those treated with ESWT or exercise had their symptoms resolved or improved at 15 months compared to those treated with a CS injection. The most commonly reported complication with ESWT was skin irritation. In another RCT, combined with a home exercise programme, ESWT was significantly more effective than sham ESWT at 2 months for pain relief. Additionally, ESWT combined with exercise had significantly greater pain-relieving benefits at long-term follow-up compared with CS injection combined with exercise in another RCT [42]. No tendon ruptures or other major complications associated with ESWT or CS injections were observed in any of these studies. All included RCTs administered 3 weekly sessions of ESWT. Finally, isometric loading was found to be as effective as isotonic loading for pain and functional outcomes up to 12 weeks in a RCT [34].
In their systematic review, Barratt et al. [100] also reported the superiority of CS injections compared to exercise for short-term pain relief. Additionally, CS injections yielded more favourable short-term outcomes for pain compared to ESWT and usual care. Their general conclusion on the management of GTPS was that definitive recommendations could not be made due to the lack of high-quality evidence. In a recent network meta-analysis, Gazendam et al. [101] found no interventions superior to no treatment at 6 and 12 months. However, PRP injections and ESWT may provide short-term pain relief, and structured exercise could produce short-term improvement in functional outcomes [101]. Wang et al. [102] found that the existing evidence on the effectiveness of CS injections in GTPS is equivocal. A RCT that was not included in our systematic review as the participants did not perform exercise as part of their treatments found no benefits of CS injections compared to placebo injections [103]. Despite the possible short-term pain-relieving benefits of CS injections in GTPS, we do not recommend its use due to the lack of longer-term benefits and their possible side effects. Finally, education of patients with GTPS may be at least as important as the exercise programme itself and stretching of the gluteal tendons should be avoided as compressive loading may be exacerbating tendinopathic pain [34, 104].
We recommend the use of a progressive isotonic exercise programme (utilising both eccentric and concentric phases) for 12 weeks as first-line treatment for GTPS. Where isotonic loading is not tolerated due to pain, an isometric programme could be prescribed instead, at least in the initial stages of treatment. In patients with restricted hip movements due to concomitant hip osteoarthritis, an isometric programme could be used for the whole duration of the treatment regime. Combined isotonic/isometric programmes could also be used as first-line treatment. For resistant cases (no or minimal improvement by the end of the programme), a ESWT programme (3 weekly sessions) could be added to exercise if available.
Table 1 summarises our treatment recommendations for each one of the three tendinopathies. For all three tendinopathies, patient education should constitute an integral part of management, and it should include explanation of principles of management and gradual load increases, advice on avoiding positions and activities that exacerbate symptoms and as much supervision as possible to maximise compliance and ensure the exercises are performed correctly.
Study Limitations
The most significant limitations of our study arise from the nature and inadequacy of the included evidence rather than methodological flaws at a meta-analysis level. We included all eligible RCTs and performed thorough assessment of the risk of bias and strength of evidence with pooling of results at pre-specified follow-up time points. However, the majority of results were based on low or very low strength of evidence. Placebo interventions were considered the same as no treatment when in reality, placebo could have its independent effects on tendinopathy. Many interventions were not connected to the network and therefore, could not be included in the network meta-analyses. For GTPS, the exercise treatment group was mixed including more than one type of loading. Finally, patient characteristics were not considered as subgroups would further decrease the strength of evidence; in reality, patients of different age groups, activity levels and duration of symptoms may respond differently to interventions.
Conclusion
There appears to be no convincing evidence currently to suggest that any adjuncts administered alone or alongside exercise are superior to exercise monotherapy for the treatment of Achilles tendinopathy, patellar tendinopathy or GTPS. Therefore, we recommend that exercise monotherapy continues to be used as a first-line treatment for these tendinopathies, and it will be effective for most patients. For resistant cases, we provide recommendations for addition of adjuncts/alternative loading programmes based on our results and the rest of the published evidence; the evidence for these is of low or very low strength; therefore, these recommendations may change in the future if evidence of higher strength suggests the superiority of other treatments, further research should focus on promising interventions with inadequate strength of evidence and especially on the comparison of different loading types used as monotherapies to inform clinical practice with more certainty.
Availability of Data and Materials
Data are available from DC upon request.
References
Cook JL, Purdam CR. Is tendon pathology a continuum? A pathology model to explain the clinical presentation of load-induced tendinopathy. Br J Sports Med. 2009;43(6):409–16.
Millar NL, Silbernagel KG, Thorborg K, et al. Tendinopathy. Nat Rev Dis Primers. 2021;7:1.
Alfredson H, Cook J. A treatment algorithm for managing Achilles tendinopathy: new treatment options. Br J Sports Med. 2007;41(4):211–6.
Aström M. Partial rupture in chronic achilles tendinopathy. A retrospective analysis of 342 cases. Acta Orthop Scand. 1998;69(4):404–7.
Edgar N, Clifford C, O’Neill S, Pedret C, Kirwan P, Millar NL. Biopsychosocial approach to tendinopathy. BMJ Open Sport Exerc Med. 2022;8(3): e001326.
Abate M, Silbernagel KG, Siljeholm C, Di Iorio A, De Amicis D, Salini V, et al. Pathogenesis of tendinopathies: inflammation or degeneration? Arthritis Res Ther. 2009;11(3):235.
Merry K, Napier C, Waugh CM, Scott A. Foundational principles and adaptation of the healthy and pathological achilles tendon in response to resistance exercise: a narrative review and clinical implications. J Clin Med. 2022;11(16):4722.
Magnusson SP, Narici MV, Maganaris CN, Kjaer M. Human tendon behaviour and adaptation, in vivo. J Physiol. 2008;586(1):71–81.
Bohm S, Mersmann F, Arampatzis A. Human tendon adaptation in response to mechanical loading: a systematic review and meta-analysis of exercise intervention studies on healthy adults. Sports Med Open. 2015;1:1–18.
Sleeswijk Visser TSO, van der Vlist AC, van Oosterom RF, van Veldhoven P, Verhaar JAN, de Vos RJ. Impact of chronic Achilles tendinopathy on health-related quality of life, work performance, healthcare utilisation and costs. BMJ Open Sport Exerc Med. 2021;7(1): e001023.
Hopkins C, Fu SC, Chua E, Hu X, Rolf C, Mattila VM, Qin L, Yung PS, Chan KM. Critical review on the socio-economic impact of tendinopathy. Asia Pac J Sports Med Arthrosc Rehabil Technol. 2016;22(4):9–20. https://doi.org/10.1016/j.asmart.2016.01.002. (PMID: 29264258; PMCID: PMC5730665).
Paavola M, Kannus P, Paakkala T, Pasanen M, Järvinen M. Long-term prognosis of patients with achilles tendinopathy. An observational 8-year follow-up study. Am J Sports Med. 2000;28(5):634–42.
Challoumas D, Pedret C, Biddle M, Ng NYB, Kirwan P, Cooper B, Nicholas P, Wilson S, Clifford C, Millar NL. Management of patellar tendinopathy: a systematic review and network meta-analysis of randomised studies. BMJ Open Sport Exerc Med. 2021;7(4): e001110.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst Rev. 2021;10(1):89.
Ardern CL, Büttner F, Andrade R, Weir A, Ashe MC, Holden S. Implementing the 27 PRISMA 2020 Statement items for systematic reviews in the sport and exercise medicine, musculoskeletal rehabilitation and sports science fields: the PERSiST (implementing Prisma in Exercise, Rehabilitation, Sport medicine and SporTs science) guidance. Br J Sports Med. 2021;56:175–95.
Robinson JM, Cook JL, Purdam C, Visentini PJ, Ross J, Maffulli N, et al. The VISA-A questionnaire: a valid and reliable index of the clinical severity of Achilles tendinopathy. Br J Sports Med. 2001;35(5):335–41.
Fearon AM, Ganderton C, Scarvell JM, Smith PN, Neeman T, Nash C, et al. Development and validation of a VISA tendinopathy questionnaire for greater trochanteric pain syndrome, the VISA-G. Man Ther. 2015;20(6):805–13.
Visentini PJ, Khan KM, Cook JL, Kiss ZS, Harcourt PR, Wark JD. The VISA score: an index of severity of symptoms in patients with jumper’s knee (patellar tendinosis). J Sci Med Sport. 1998;1(1):22–8.
Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343: d5928.
Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924–6.
White IR. Network meta-analysis. Stata J. 2015;15(4):951–85.
Ma J, Liu W, Hunter A, Zhang W. Performing meta-analysis with incomplete statistical information in clinical trials. BMC Med Res Methodol. 2008;8(1):56.
Abat F, Sánchez-Sánchez JL, Martín-Nogueras AM, Calvo-Arenillas JI, Yajeya J, Méndez-Sánchez R, et al. Randomized controlled trial comparing the effectiveness of the ultrasound-guided galvanic electrolysis technique (USGET) versus conventional electro-physiotherapeutic treatment on patellar tendinopathy. J Exp Orthop. 2016;3(1):34.
Agergaard AS, Svensson RB, Malmgaard-Clausen NM, Couppé C, Hjortshoej MH, Doessing S, Kjaer M, Magnusson SP. Clinical outcomes, structure, and function improve with both heavy and moderate loads in the treatment of patellar tendinopathy: a randomized clinical trial. Am J Sports Med. 2021;49(4):982–93.
Bahr R, Fossan B, Løken S, Engebretsen L. Surgical treatment compared with eccentric training for patellar tendinopathy (Jumper’s Knee). A randomized, controlled trial. J Bone Joint Surg Am. 2006;88(8):1689–98.
Balius R, Álvarez G, Baró F, Jiménez F, Pedret C, Costa E, et al. A 3-arm randomized trial for achilles tendinopathy: eccentric training, eccentric training plus a dietary supplement containing mucopolysaccharides, or passive stretching plus a dietary supplement containing mucopolysaccharides. Curr Ther Res. 2016;78:1–7.
Bell KJ, Fulcher ML, Rowlands DS, Kerse N. Impact of autologous blood injections in treatment of mid-portion Achilles tendinopathy: double blind randomised controlled trial. BMJ. 2013;346:f2310.
Beyer R, Kongsgaard M, Hougs Kjær B, Øhlenschlæger T, Kjær M, Magnusson SP. Heavy slow resistance versus eccentric training as treatment for Achilles tendinopathy: a randomized controlled trial. Am J Sports Med. 2015;43(7):1704–11.
Boesen AP, Hansen R, Boesen MI, Malliaras P, Langberg H. Effect of high-volume injection, platelet-rich plasma, and sham treatment in chronic midportion achilles tendinopathy: a randomized double-blinded prospective study. Am J Sports Med. 2017;45(9):2034–43.
Boesen AP, Langberg H, Hansen R, Malliaras P, Boesen MI. High volume injection with and without corticosteroid in chronic midportion achilles tendinopathy. Scand J Med Sci Sports. 2019;29(8):1223–31.
Bradford B, Rio E, Murphy M, Wells J, Khondoker M, Clarke C, Chan Y, Chester R. Immediate effects of two isometric calf muscle exercises on mid-portion achilles tendon pain. Int J Sports Med. 2021;42(12):1122–7.
Breda SJ, Oei EHG, Zwerver J, Visser E, Waarsing E, Krestin GP, et al. Effectiveness of progressive tendon-loading exercise therapy in patients with patellar tendinopathy: a randomised clinical trial. Br J Sports Med. 2021;55(9):501–9.
Cannell LJ, Taunton JE, Clement DB, Smith C, Khan KM. A randomised clinical trial of the efficacy of drop squats or leg extension/leg curl exercises to treat clinically diagnosed jumper’s knee in athletes: pilot study. Br J Sports Med. 2001;35(1):60–4.
Clifford C, Paul L, Syme G, Millar NL. Isometric versus isotonic exercise for greater trochanteric pain syndrome: a randomised controlled pilot study. BMJ Open Sport Exerc Med. 2019;5(1):558.
de Jonge S, de Vos RJ, Weir A, van Schie HTM, Bierma-Zeinstra SMA, Verhaar JAN, et al. One-year follow-up of platelet-rich plasma treatment in chronic Achilles tendinopathy: a double-blind randomized placebo-controlled trial. Am J Sports Med. 2011;39(8):1623–30.
de Jonge S, de Vos RJ, Van Schie HTM, Verhaar J, Weir A, Tol JL. One-year follow-up of a randomised controlled trial on added splinting to eccentric exercises in chronic midportion Achilles tendinopathy. Br J Sports Med. 2010;44(9):673–7.
de Vos RJ, Weir A, Visser RJA, de Winter T, Tol JL. The additional value of a night splint to eccentric exercises in chronic midportion Achilles tendinopathy: a randomised controlled trial. Br J Sports Med. 2007;41(7): e5.
de Vries A, Zwerver J, Diercks R, Tak I, van Berkel S, van Cingel R, et al. Effect of patellar strap and sports tape on pain in patellar tendinopathy: a randomized controlled trial. Scand J Med Sci Sports. 2016;26(10):1217–24.
Dragoo JL, Wasterlain AS, Braun HJ, Nead KT. Platelet-rich plasma as a treatment for patellar tendinopathy: a double-blind, randomized controlled trial. Am J Sports Med. 2014;42(3):610–8.
Frohm A, Saartok T, Halvorsen K, Renström P. Eccentric treatment for patellar tendinopathy: a prospective randomised short-term pilot study of two rehabilitation protocols. Br J Sports Med. 2007;41(7):e7.
Ganderton C, Semciw A, Cook J, Moreira E, Pizzari T. Gluteal loading versus sham exercises to improve pain and dysfunction in postmenopausal women with greater trochanteric pain syndrome: a randomized controlled trial. J Women’s Health. 2018;27(6):815–29.
Heaver C, Pinches M, Kuiper JH, et al. Greater trochanteric pain syndrome: focused shockwave therapy versus an ultrasound guided injection: a randomised control trial. HIP Int. 2021;33:490–9.
Herrington L, McCulloch R. The role of eccentric training in the management of Achilles tendinopathy: a pilot study. Phys Ther Sport. 2007;8(4):191–6.
Holden S, Lyng K, Graven-Nielsen T, Riel H, Olesen JL, Larsen LH, et al. Isometric exercise and pain in patellar tendinopathy: a randomized crossover trial. J Sci Med Sport. 2020;23(3):208–14.
Horstmann T, Jud HM, Fröhlich V, Mündermann A, Grau S. Whole-body vibration versus eccentric training or a wait-and-see approach for chronic Achilles tendinopathy: a randomized clinical trial. J Orthop Sport Phys Ther. 2013;43(11):794–803.
Jonsson P, Alfredson H. Superior results with eccentric compared to concentric quadriceps training in patients with jumper’s knee: a prospective randomised study. Br J Sports Med. 2005;39(11):847–50.
Kaux J-F, Drion P, Croisier J-L, Crielaard J-M. Tendinopathies and platelet-rich plasma (PRP): from pre-clinical experiments to therapeutic use. J Stem Cells Regen Med. 2015;11(1):7–17.
Knobloch K, Kraemer R, Jagodzinski M, Zeichen J, Meller R, Vogt PM. Eccentric training decreases paratendon capillary blood flow and preserves paratendon oxygen saturation in chronic achilles tendinopathy. J Orthop Sports Phys Ther. 2007;37(5):269–76.
Knobloch K, Schreibmueller L, Longo UG, Vogt PM. Eccentric exercises for the management of tendinopathy of the main body of the Achilles tendon with or without the AirHeelTM Brace. A randomized controlled trial. A: effects on pain and microcirculation. Disabil Rehabil. 2008;30(20–22):1685–91.
Kongsgaard M, Kovanen V, Aagaard P, Doessing S, Hansen P, Laursen AH, et al. Corticosteroid injections, eccentric decline squat training and heavy slow resistance training in patellar tendinopathy. Scand J Med Sci Sports. 2009;19(6):790–802.
Krogh TP, Ellingsen T, Christensen R, Jensen P, Fredberg U. Ultrasound-guided injection therapy of Achilles tendinopathy with platelet-rich plasma or saline: a randomized, blinded, placebo-controlled trial. Am J Sports Med. 2016;44(8):1990–7.
Lee MC, Lee SK, Jung SY, Moon HH. New insight of high-intensity interval training on physiological adaptation with brain functions. J Exerc Nutr Biochem. 2018;22(3):1–5.
López-Royo MP, Ríos-Díaz J, Galán-Díaz RM, Herrero P, Gómez-Trullén EM. A comparative study of treatment interventions for patellar tendinopathy: a randomized controlled trial. Arch Phys Med Rehabil. 2021;102(5):967–75.
Mafi N, Lorentzon R, Alfredson H. Superior short-term results with eccentric calf muscle training compared to concentric training in a randomized prospective multicenter study on patients with chronic Achilles tendinosis. Knee Surg Sport Traumatol Arthrosc. 2001;9(1):42–7.
Mellor R, Grimaldi A, Wajswelner H, Hodges P, Abbott JH, Bennell K, et al. Exercise and load modification versus corticosteroid injection versus ‘wait and see’ for persistent gluteus medius/minimus tendinopathy (the LEAP trial): a protocol for a randomised clinical trial. BMC Musculoskelet Disord. 2016;17(1):196.
Munteanu SE, Scott LA, Bonanno DR, Landorf KB, Pizzari T, Cook JL, et al. Effectiveness of customised foot orthoses for Achilles tendinopathy: a randomised controlled trial. Br J Sports Med. 2015;49(15):989–94.
Petersen W, Welp R, Rosenbaum D. Chronic Achilles tendinopathy: a prospective randomized study comparing the therapeutic effect of eccentric training, the AirHeel brace, and a combination of both. Am J Sports Med. 2007;35(10):1659–67.
Pearson J, Rowlands D, Highet R. Autologous Blood injection to treat Achilles tendinopathy? A randomized controlled trial. J Sport Rehabil. 2012;21(3):218–24.
Rabusin CL, Menz HB, McClelland JA, Evans AM, Malliaras P, Docking SI, Landorf KB, Gerrard JM, Munteanu SE. Efficacy of heel lifts versus calf muscle eccentric exercise for mid-portion Achilles tendinopathy (HEALTHY): a randomised trial. Br J Sports Med. 2021;55(9):486–92.
Ramon S, Russo S, Santoboni F, Lucenteforte G, Di Luise C, de Unzurrunzaga R, Vetrano M, Albano M, Baldini R, Cugat R, Stella G, Balato G, Seijas R, Nusca SM, Servodidio V, Vulpiani MC. Focused shockwave treatment for greater trochanteric pain syndrome: a multicenter, randomized, controlled clinical trial. J Bone Joint Surg Am. 2020;102(15):1305–11.
Resteghini P, Khanbhai TA, Mughal S, Sivardeen Z. Double-blind randomized controlled trial: injection of autologous blood in the treatment of chronic patella tendinopathy-a pilot study. Clin J Sport Med Off J Can Acad Sport Med. 2016;26(1):17–23.
Rigby JH, Mortensen BB, Draper DO. Wireless versus wired iontophoresis for treating patellar tendinopathy: a randomized clinical trial. J Athl Train. 2015;50(11):1165–73.
Rio E, van Ark M, Docking S, Moseley GL, Kidgell D, Gaida JE, et al. Isometric contractions are more analgesic than isotonic contractions for patellar tendon pain: an in-season randomized clinical trial. Clin J Sport Med Off J Can Acad Sport Med. 2017;27(3):253–9.
Rio E, Kidgell D, Purdam C, Gaida J, Moseley GL, Pearce AJ, et al. Isometric exercise induces analgesia and reduces inhibition in patellar tendinopathy. Br J Sports Med. 2015;49(19):1277–83.
Rodas G, Soler-Rich R, Rius-Tarruella J, Alomar X, Balius R, Orozco L, Masci L, Maffulli N. Effect of autologous expanded bone marrow mesenchymal stem cells or leukocyte-poor platelet-rich plasma in chronic patellar tendinopathy (with gap >3 mm): preliminary outcomes after 6 months of a double-blind, randomized, prospective study. Am J Sports Med. 2021;49(6):1492–504.
Rompe JD, Nafe B, Furia JP, Maffulli N. Eccentric loading, shock-wave treatment, or a wait- and-see policy for tendinopathy of the main body of tendo Achillis: a randomized controlled trial. Am J Sports Med. 2007;35(3):374–83.
Rompe JD, Furia J, Maffulli N. Eccentric loading versus eccentric loading plus shock-wave treatment for midportion Achilles tendinopathy: a randomized controlled trial. Am J Sports Med. 2008;37(3):463–70.
Rompe JD, Segal NA, Cacchio A, Furia JP, Morral A, Maffulli N. Home training, local corticosteroid injection, or radial shock wave therapy for greater trochanter pain syndrome. Am J Sports Med. 2009;37(10):1981–90.
Roos EM, Engström M, Lagerquist A, Söderberg B. Clinical improvement after 6 weeks of eccentric exercise in patients with mid-portion Achilles tendinopathy—a randomized trial with 1-year follow-up. Scand J Med Sci Sports. 2004;14(5):286–95.
Scott A, LaPrade RF, Harmon KG, Filardo G, Kon E, Della Villa S, et al. Platelet-rich plasma for patellar tendinopathy: a randomized controlled trial of leukocyte-rich PRP or leukocyte-poor PRP versus saline. Am J Sports Med. 2019;47(7):1654–61.
Silbernagel KG, Thomeé R, Eriksson BI, Karlsson J. Continued sports activity, using a pain-monitoring model, during rehabilitation in patients with Achilles tendinopathy: a randomized controlled study. Am J Sports Med. 2007;35(6):897–906.
Silbernagel K, Thomeé R, Thomeé P, Karlsson J. Eccentric overload training for patients with chronic Achilles tendon pain—a randomised controlled study with reliability testing of the evaluation methods. Scand J Med Sci Sports. 2001;11(4):197–206.
Stasinopoulos D, Stasinopoulos I. Comparison of effects of exercise programme, pulsed ultrasound and transverse friction in the treatment of chronic patellar tendinopathy. Clin Rehabil. 2004;18(4):347–52.
Steunebrink M, Zwerver J, Brandsema R, Groenenboom P, van den Akker-Scheek I, Weir A. Topical glyceryl trinitrate treatment of chronic patellar tendinopathy: a randomised, double-blind, placebo-controlled clinical trial. Br J Sports Med. 2013;47(1):34–9.
Stevens M, Tan C-W. Effectiveness of the Alfredson protocol compared with a lower repetition-volume protocol for midportion Achilles tendinopathy: a randomized controlled trial. J Orthop Sport Phys Ther. 2013;44(2):59–67.
Thijs KM, Zwerver J, Backx FJG, Steeneken V, Rayer S, Groenenboom P, et al. Effectiveness of shockwave treatment combined with eccentric training for patellar tendinopathy: a double-blinded randomized study. Clin J Sport Med Off J Can Acad Sport Med. 2017;27(2):89–96.
Tumilty S, Mani R, Baxter GD. Photobiomodulation and eccentric exercise for Achilles tendinopathy: a randomized controlled trial. Lasers Med Sci. 2016;31(1):127–35.
Tumilty S, McDonough S, Hurley DA, Baxter GD. Clinical effectiveness of low-level laser therapy as an adjunct to eccentric exercise for the treatment of Achilles’ tendinopathy: a randomized controlled trial. Arch Phys Med Rehabil. 2012;93(5):733–9.
van Ark M, Cook JL, Docking SI, Zwerver J, Gaida JE, van den Akker-Scheek I, et al. Do isometric and isotonic exercise programs reduce pain in athletes with patellar tendinopathy in-season? A randomised clinical trial. J Sci Med Sport. 2016;19(9):702–6.
van der Worp H, Zwerver J, Hamstra M, van den Akker-Scheek I, Diercks RL. No difference in effectiveness between focused and radial shockwave therapy for treating patellar tendinopathy: a randomized controlled trial. Knee Surg Sports Traumatol Arthrosc. 2014;22(9):2026–32.
van der Vlist AC, van Oosterom RF, van Veldhoven PLJ, Bierma-Zeinstra SMA, Waarsing JH, Verhaar JAN, et al. Effectiveness of a high volume injection as treatment for chronic Achilles tendinopathy: randomised controlled trial. BMJ. 2020;9(370): m3027.
van der Vlist AC, van Veldhoven PLJ, van Oosterom RF, Verhaar JAN, de Vos RJ. Isometric exercises do not provide immediate pain relief in Achilles tendinopathy: a quasi-randomized clinical trial. Scand J Med Sci Sports. 2020;30(9):1712–21.
Vetrano M, Castorina A, Vulpiani MC, Baldini R, Pavan A, Ferretti A. Platelet-rich plasma versus focused shock waves in the treatment of jumper’s knee in athletes. Am J Sports Med. 2013;41(4):795–803.
Visnes H, Hoksrud A, Cook J, Bahr R. No effect of eccentric training on jumper’s knee in volleyball players during the competitive season: a randomized clinical trial. Clin J Sport Med Off J Can Acad Sport Med. 2005;15(4):227–34.
Wang C-J, Ko J-Y, Chan Y-S, Weng L-H, Hsu S-L. Extracorporeal shockwave for chronic patellar tendinopathy. Am J Sports Med. 2007;35(6):972–8.
Warden SJ, Metcalf BR, Kiss ZS, Cook JL, Purdam CR, Bennell KL, et al. Low-intensity pulsed ultrasound for chronic patellar tendinopathy: a randomized, double-blind, placebo-controlled trial. Rheumatology (Oxford). 2008;47(4):467–71.
Yelland MJ, Sweeting KR, Lyftogt JA, Ng SK, Scuffham PA, Evans KA. Prolotherapy injections and eccentric loading exercises for painful Achilles tendinosis: a randomised trial. Br J Sports Med. 2011;45(5):421–8.
Young MA, Cook JL, Purdam CR, Kiss ZS, Alfredson H. Eccentric decline squat protocol offers superior results at 12 months compared with traditional eccentric protocol for patellar tendinopathy in volleyball players. Br J Sports Med. 2005;39(2):102–5.
Zhang B, Zhong L, Xu S, Jiang H, Shen J. Acupuncture for chronic Achilles tendinopathy: a randomized controlled study. Chin J Integr Med. 2013;19(12):900–4.
Zwerver J, Hartgens F, Verhagen E, van der Worp H, van den Akker-Scheek I, Diercks RL. No effect of extracorporeal shockwave therapy on patellar tendinopathy in jumping athletes during the competitive season: a randomized clinical trial. Am J Sports Med. 2011;39(6):1191–9.
Kannus P. Tendon pathology: basic science and clinical applications. Sport Exerc Inj. 1997;1(3):62–75.
Johannsen F, Olesen JL, Øhlenschläger TF, Lundgaard-Nielsen M, Cullum CK, Jakobsen AS, Rathleff MS, Magnusson PS, Kjær M. Effect of ultrasonography-guided corticosteroid injection vs placebo added to exercise therapy for Achilles tendinopathy: a randomized clinical trial. JAMA Netw Open. 2022;5(7): e2219661.
O’Neill S, Radia J, Bird K, Rathleff MS, Bandholm T, Jorgensen M, Thorborg K. Acute sensory and motor response to 45-s heavy isometric holds for the plantar flexors in patients with Achilles tendinopathy. Knee Surg Sports Traumatol Arthrosc. 2019;27(9):2765–73.
van der Vlist AC, Winters M, Weir A, Ardern CL, Welton NJ, Caldwell DM, Verhaar JAN, de Vos RJ. Which treatment is most effective for patients with Achilles tendinopathy? A living systematic review with network meta-analysis of 29 randomised controlled trials. Br J Sports Med. 2021;55(5):249–56.
Rhim HC, Kim MS, Choi S, Tenforde AS. Comparative efficacy and tolerability of nonsurgical therapies for the treatment of midportion Achilles tendinopathy: a systematic review with network meta-analysis. Orthop J Sports Med. 2020;8(7):2325967120930567.
Challoumas D, Kirwan PD, Borysov D, Clifford C, McLean M, Millar NL. Topical glyceryl trinitrate for the treatment of tendinopathies: a systematic review. Br J Sports Med. 2019;53(4):251–62.
Fogli M, Giordan N, Mazzoni G. Efficacy and safety of hyaluronic acid (500–730 kDa) Ultrasound-guided injections on painful tendinopathies: a prospective, open label, clinical study. Muscles Ligaments Tendons J. 2017;7(2):388–95.
Frizziero A, Vittadini F, Oliva F, et al. Thu0492 efficacy of us-guided hyaluronic acid injections in achilles and patellar mid-portion tendinopathies: a prospective multicentric clinical trial. Ann Rheum Dis. 2019;78:536–7.
Chen PC, Wu KT, Chou WY, Huang YC, Wang LY, Yang TH, Siu KK, Tu YK. Comparative effectiveness of different nonsurgical treatments for patellar tendinopathy: a systematic review and network meta-analysis. Arthroscopy. 2019;35(11):3117-3131.e2.
Barratt PA, Brookes N, Newson A. Conservative treatments for greater trochanteric pain syndrome: a systematic review. Br J Sports Med. 2017;51(2):97–104.
Gazendam A, Ekhtiari S, Axelrod D, Gouveia K, Gyemi L, Ayeni O, Bhandari M. Comparative efficacy of nonoperative treatments for greater trochanteric pain syndrome: a systematic review and network meta-analysis of randomized controlled trials. Clin J Sport Med. 2021;32(4):427–32.
Wang Y, Wang K, Qin Y, Wang S, Tan B, Jia L, Jia G, Niu L. The effect of corticosteroid injection in the treatment of greater trochanter pain syndrome: a systematic review and meta-analysis of randomized controlled trials. J Orthop Surg Res. 2022;17(1):283.
Nissen MJ, Brulhart L, Faundez A, Finckh A, Courvoisier DS, Genevay S. Glucocorticoid injections for greater trochanteric pain syndrome: a randomised double-blind placebo-controlled (GLUTEAL) trial. Clin Rheumatol. 2019;38(3):647–55.
Cook JL, Purdam C. Is compressive load a factor in the development of tendinopathy? Br J Sports Med. 2012;46(3):163–8.
Acknowledgements
None.
Funding
This work was funded by Medical Research Council Grant MR/R020515/1 (NLM).
Author information
Authors and Affiliations
Contributions
DC, NLM, and GC conceived and designed the study. DC and GC performed the analysis. CP, SO and NLM provided expert advice. All authors analysed the data. GC, DC, NLM, SO and CP wrote the paper. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
There are no competing interests for any author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1
. Supplementary meta analysis figures, network maps and tables.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Challoumas, D., Crosbie, G., O’Neill, S. et al. Effectiveness of Exercise Treatments with or without Adjuncts for Common Lower Limb Tendinopathies: A Living Systematic Review and Network Meta-analysis. Sports Med - Open 9, 71 (2023). https://doi.org/10.1186/s40798-023-00616-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40798-023-00616-1