Abstract
Background
Upper gastrointestinal bleeding (UGIB) is a routine medical emergency. The most common non-variceal cause is peptic ulcer disease, while a rare presentation is peptic ulcer-induced splenic artery pseudoaneurysm (SAP). Primary endoscopic treatment is generally attempted for UGIB; however, it sometimes fails when arterial etiology is present. In such cases, either transcatheter arterial embolization (TAE) or surgery is necessary, but the choice of treatment is controversial. We present a case that illustrates the utility of both approaches in a gastric ulcer-induced SAP.
Case presentation
A 33-year-old male presented with hemorrhagic shock secondary to UGIB. The source of bleeding was identified as an SAP that was caused by a gastric ulcer. TAE enabled temporary bleeding control despite the patient’s poor overall condition and limited blood transfusion capability. However, rebleeding occurred soon after stabilization. Ultimately, we performed proximal gastrectomy and splenic artery ligation, and the patient survived.
Conclusions
SAP is an uncommon occurrence, and angiographic information is important for correctly identifying the source of bleeding. The treatment for SAP bleeding is basically the same as for endoscopically unmanageable non-variceal UGIB, since TAE and surgery each have a different utility, depending on the situation. If surgery is performed, especially SA ligation and gastrectomy, it is important to consider the circulation of the spleen and residual stomach. Using TAE and laparotomy, we managed to save the life of the patient with massive hemorrhage under limited circumstances.
Similar content being viewed by others
Background
Upper gastrointestinal bleeding (UGIB) is a common medical emergency with a reported mortality of 2–10% [1]. Within UGIB, variceal and non-variceal etiologies are distinguished, as the management and patient outcomes differ. The most common cause of acute non-variceal UGIB is peptic ulcer disease (PUD) [2], with approximately 50% of all cases attributed to PUD [3]. One rare complication of PUD that can lead to massive UGIB is peptic ulcer-induced splenic artery pseudoaneurysm (SAP). So far, only a few cases have been reported.
Transcatheter arterial embolization (TAE) or surgical intervention are necessary for massive UGIBs that cannot be endoscopically controlled. The decision between surgery and TAE remains controversial, as these patients are often at high risk and are poor surgical candidates. TAE can save high-risk patients [4]; however, compared with surgery, it results in a higher rebleeding rate for uncontrolled peptic ulcer bleeding [5]. Nevertheless, the efficacy and safety of TAE in UGIB is lower than that in lower gastrointestinal bleeding or biliary hemorrhage, because the complex collateral circulation in the upper gastrointestinal tract can inhibit efficient embolization [6]. Here, we report the case of a patient with massive UGIB who required both interventions.
Case presentation
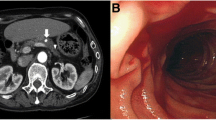
An alcoholic 33-year-old male with a 2-week history of hematemesis presented to the hospital with pallor and impaired consciousness. The patient was hemodynamically unstable on arrival. On examination, his body temperature (BT) was 35.9 °C, heart rate was 120 beats per minute in sinus tachycardia, blood pressure was 84/34 mmHg, respiratory rate was 26 breaths per minute, and oxygen saturation was 100% on 10 L of oxygen. Laboratory results showed severe anemia (hemoglobin 3.7 g/dL) and poor pre-existing nutritional status (cholinesterase 81 U/L, albumin 1.9 g/dL). Dynamic computed tomography (CT) revealed a massive, enhanced fluid collection in the stomach (Fig. 1), while extravasation or free air were not observed. The patient underwent urgent upper gastrointestinal endoscopy. Massive fresh blood pooling was observed in the stomach and a gastric ulcer was located in the upper posterior wall of the gastric body (Fig. 2). No active bleeding was noted, and endoscopy was completed without clipping or cauterization.
Upon admission to the intensive care unit, his general condition improved with conservative treatment, including blood transfusion. On the second day after admission (on a holiday night), he experienced massive rebleeding, hematemesis, and sudden onset of hemorrhagic shock. Blood tests showed fatal anemia (hemoglobin 0.7 g/dL). The patient was managed urgently with massive blood transfusions and catecholamine administration. Cardiopulmonary resuscitation was required for several minutes. Rapid hemostatic intervention was necessary; however, the patient was in poor general condition, because of acidosis (pH 7.09) and hypothermia (BT 33.4 °C). Even worse, the preparation with transfusion was inadequate to conduct a laparotomy procedure. Therefore, emergency angiography and TAE were performed.
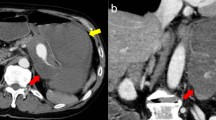
Celiac angiography revealed extravasation from a splenic artery pseudoaneurysm (SAP) (Fig. 3A). First, we aimed to isolate the SAP, but the catheter did not reach distal to the SAP from the celiac artery; there were vascular migrations from chronic pancreatitis and very limited utility of TAE. Reluctantly, we embolized the proximal splenic artery (SA) with a coil. The extravasation persisted through the gastroduodenal artery (GDA) and right gastroepiploic artery (RGEA) (Fig. 3B). The distal SA was then embolized with coils through the GDA and RGEA. Subsequently, the bleeding lessened (Fig. 3C), but it persisted through the intrapancreatic arcade (Fig. 3D). Coiling of the arcade was also difficult because of the vascular migrations. Finally, the arcade was embolized with a gelatin sponge and the extravasation dissipated (Fig. 3E).
A Celiac angiography shows the splenic artery pseudoaneurysm (SAP) and extravasation from it (triangle). B Despite the proximal splenic artery (SA) being embolized with coils (arrow), extravasation persisted through the gastroduodenal artery and right gastroepiploic artery. C, D After the distal SA was embolized with coils (arrow in C), extravasation from the SAP lessened but continued through the intrapancreatic arcade (arrow in D). E The arcade was embolized with a gelatin sponge, and then the extravasation dissipated
Although TAE accomplished a temporary cessation of bleeding, rebleeding occurred the following morning. Initial CT imaging was negative, but a second dynamic CT revealed extravasation from the SAP into the stomach (Fig. 4). The patient responded to fluids and blood transfusions, and vital signs were relatively stable (Fig. 5A). Acidosis and hypothermia were compensating, with pH 7.52 and BT 36.3℃ (Fig. 5B). During the daytime on a weekday, there were sufficient transfusions, clinical staff, and materials were ready for another surgical intervention. We decided to perform emergency laparotomy.
Intraoperatively, the stomach was found to be filled with blood clots. A bleeding ulcer on the upper posterior wall of the stomach body was perforated, with a hole measuring 5.0 cm in diameter (Fig. 6). The SAP was identified as the source of bleeding. First, we ligated the SA proximal to the gastric ulcer perforation where the SAP was evident. At that point, hemostasis was obtained, and vital signs stabilized. Since the perforation was large, and simple suture or omentopexy was difficult, we decided to perform gastrectomy. Distal gastrectomy (DG) would have left the possibility of residual gastric necrosis from SA ligation. Therefore, we chose proximal gastrectomy (PG). The mobilization of gastric antrum was also not easy due to chronic pancreatitis. Therefore, we chose the double-tract method. The spleen and pancreas were preserved, because no visible color change was observed. The operation time was 196 min, and the intraoperative bleeding volume was 3000 mL.
The patient’s postoperative course was generally good. On postoperative day 7, contrast-enhanced CT showed partial splenic infarction; however, there was no intra-abdominal abscess or other abnormal findings. The patient was treated conservatively and discharged from the hospital on day 15 after surgery. The total transfusion during the hospitalization comprised 82 units of concentrated red blood cells, 50 units of fresh frozen plasma, and 40 units of platelets.
Discussion
PUD is the main cause of non-variceal UGIB [3], with duodenal ulcers more likely to experience bleeding than the gastric ulcers [7]. However, SAP is a rare occurrence. In a large series from the Mayo Clinic, only 10 SAPs were identified in patients over 18 years of age. The SAP is most often caused by pancreatitis (52%) or trauma (29%) and rarely by PUD (2%) [8].
To the best of our knowledge, seven cases of PUD-induced SAP have been reported in the past 14 years. Including our case, Table 1 summarizes the characteristics of patients, treatment, and outcomes [9,10,11,12,13,14,15] (Table 1). The most common symptoms were abdominal pain and hematemesis (5/8 cases, 62.5% each), followed by melena (4/8 cases, 50%) and neurological symptoms (3/8 cases, 37.5%). As major bleeding occurred, the patients were highly anemic and vital signs were often unstable. On initial endoscopy, the PUD was often located on the posterior wall of the stomach (6/8 cases, 75%), followed by the duodenum (1/8 case, 12.5%). When PUD is present in the posterior wall of the stomach or duodenum, UGIB by arterial perforation, including SAP bleeding, should be considered in the differential diagnosis. An earlier review mentioned that SAP was most commonly diagnosed by angiography (52%) followed by CT (36%) [8]. In two cases, including ours, a repeat CT scan correctly identified the source of bleeding [13]. Unfortunately, one case was misdiagnosed as a cystic tumor on the initial non-contrast CT and biopsied [14]. The lesson from the cases above is that angiographic information is essential in UGIB.
The treatment of SAP bleeding is fundamentally the same as for endoscopically unmanageable non-variceal UGIB; to date, various methods have been reported. Of the eight published cases mentioned above, primary treatment often included TAE (5/8 cases, 62.5%), with rebleeding occurring in approximately half of the TAE group. According to one previous report, the rebleeding rate was higher in the TAE group than in the surgery group (HR 2.48, 95% CI 1.33–4.72) [5]. On the other hand, considering all secondary hemostatic interventions, both groups achieved approximately 87% durable hemostasis. Rebleeding within 3 days (OR 3.7, p = 0.042), and corticosteroid use (OR 5.2, p = 0.017) had significant negative impacts on survival [16].
Laparotomy was performed in most cases (7/8 cases, 87.5%). In two cases, surgery was performed at the start; the patients survived without rebleeding. In three post-TAE rebleeding cases, including ours, surgery was chosen as the secondary intervention and achieved good results. In one case without rebleeding, early laparotomy was performed for curative purposes. The resuscitative endovascular balloon occlusion of the aorta (REBOA) technique has been reported to be useful for decreasing SAP bleeding volume [15]. Also, resuscitative thoracotomy with aortic cross-clamp (RTACC) is indicated for imminent cardiac arrest. In every case, the procedure was designed to provide rapid control of blood loss, leading to reliable hemostasis.
As regards complications, in one case, splenic infarction occurred after coil embolization of the splenic artery [14]. In other cases, splenic blood flow was maintained despite splenic artery ligation or embolization; collateral blood circulation from the inferior phrenic artery or omentum is thought to be involved. In our case, collateral circulation was expected from the intraoperative findings, so splenectomy was avoided at that time. As a result, the only complication was partial splenic infarction, which was followed conservatively. If a splenic abscess forms postoperatively, splenectomy should be considered.
There is another risk, that of residual gastric necrosis, when DG and splenic artery ligation are performed simultaneously, which Nakata et al. mitigated by using open abdominal postoperative management [15]. In our case, PG and the SA ligation were performed, and residual stomach circulation was preserved through the RGEA and right gastric artery. To avoid residual gastric necrosis, PG or total gastrectomy was found to be safer than DG, because splenic artery ligation or embolization is always necessary in SAP bleeding.
Finally, multidisciplinary and rapid treatment is required to save lives in case of SAP bleeding. Although in limited circumstances, we managed to temporarily control the critical bleeding with TAE. When rebleeding occurred, we considered that blood flow from the intrapancreatic arcade, which had been embolized with a gelatin sponge, had resumed. Therefore, we concluded that no further IVR would be effective; we decided to perform surgery. Retrospectively considering our case, the best way to respond to shock requiring cardiopulmonary resuscitation is REBOA or RTACC; however, each facility’s background, medical resources, timeframe, and other conditions are different. Establishing an emergency medical care system that makes these interventions possible is a future challenge for regional hospitals.
Conclusions
This is a case report of SAP bleeding caused by a gastric ulcer. Using TAE and laparotomy, we managed to save the life of a patient with massive hemorrhage under limited treatment circumstances. When endoscopic treatment fails or is difficult, TAE or surgery are necessary. TAE can save poor surgical candidates like our patient; however, the rebleeding rate is higher than with surgery. If surgery is performed for SAP, especially SA ligation and gastrectomy, it is important to consider the vascularization of the spleen and residual stomach. In fatal circumstances, REBOA or RTACC should be considered for reducing blood loss. The establishment of an emergency medical care system is a future issue.
Availability of data and materials
Data sharing is not applicable to this article, as no datasets were generated or analyzed during the current study.
Abbreviations
- BT:
-
Body temperature
- CT:
-
Computed tomography
- DG:
-
Distal gastrectomy
- GDA:
-
Gastroduodenal artery
- PG:
-
Proximal gastrectomy
- PUD:
-
Peptic ulcer disease
- REBOA:
-
Resuscitative endovascular balloon occlusion of the aorta
- RGEA:
-
Right gastroepiploic artery
- RTACC:
-
Resuscitative thoracotomy with aortic cross-clamp
- SA:
-
Splenic artery
- SAP:
-
Splenic artery pseudoaneurysm
- TAE:
-
Transcatheter arterial embolization
- UGIB:
-
Upper gastrointestinal bleeding
References
Stanley AJ, Laine L. Management of acute upper gastrointestinal bleeding. BMJ. 2019;364: l536. https://doi.org/10.1136/bmj.l536.
Loffroy R, Favelier S, Pottecher P, Estivalet L, Genson PY, Gehin S, et al. Transcatheter arterial embolization for acute nonvariceal upper gastrointestinal bleeding: Indications, techniques and outcomes. Diagn Interv Imaging. 2015;96:731–44. https://doi.org/10.1016/j.diii.2015.05.002.
Laine L, Peterson WL. Bleeding peptic ulcer. N Engl J Med. 1994;331:717–27. https://doi.org/10.1056/NEJM199409153311107.
Ljungdahl M, Eriksson LG, Nyman R, Gustavsson S. Arterial embolisation in management of massive bleeding from gastric and duodenal ulcers. Eur J Surg. 2002;168:384–90. https://doi.org/10.1080/110241502320789050.
Sverdén E, Mattsson F, Lindström D, Sondén A, Lu Y, Lagergren J. Transcatheter arterial embolization compared with surgery for uncontrolled peptic ulcer bleeding: a population-based Cohort Study. Ann Surg. 2019;269:304–9. https://doi.org/10.1097/SLA.0000000000002565.
Defreyne L, Vanlangenhove P, De Vos M, Pattyn P, Van Maele G, Decruyenaere J, et al. Embolization as a first approach with endoscopically unmanageable acute nonvariceal gastrointestinal hemorrhage. Radiology. 2001;218:739–48. https://doi.org/10.1148/radiology.218.3.r01mr05739.
van Leerdam ME. Epidemiology of acute upper gastrointestinal bleeding. Best Pract Res Clin Gastroenterol. 2008;22:209–24. https://doi.org/10.1016/j.bpg.2007.10.011.
Tessier DJ, Stone WM, Fowl RJ, Abbas MA, Andrews JC, Bower TC, et al. Clinical features and management of splenic artery pseudoaneurysm: case series and cumulative review of literature. J Vasc Surg. 2003;38:969–74. https://doi.org/10.1016/s0741-5214(03)00710-9.
Pasumarthy L, Kumar RR, Srour J, Ahlbrandt D. Penetration of gastric ulcer into the splenic artery: a rare complication. Gastroenterology Res. 2009;2:350–2. https://doi.org/10.4021/gr2009.10.1321.
Syed SM, Moradian S, Ahmed M, Ahmed U, Shaheen S, Stalin V. A benign gastric ulcer eroding into a splenic artery pseudoaneurysm presenting as a massive upper gastrointestinal bleed. J Surg Case Rep. 2014. https://doi.org/10.1093/jscr/rju102.
Varshney P, Songra B, Mathur S, Gothwal S, Malik P, Rathi M, et al. Splenic artery pseudoaneurysm presenting as massive hematemesis: a diagnostic dilemma. Case Rep Surg. 2014;2014: 501937. https://doi.org/10.1155/2014/501937.
Sawicki M, Marlicz W, Czapla N, Łokaj M, Skoczylas MM, Donotek M, et al. Massive upper gastrointestinal bleeding from a splenic artery pseudoaneurysm caused by a penetrating gastric ulcer: Case report and review of literature. Pol J Radiol. 2015;80:384–7. https://doi.org/10.12659/PJR.894465.
Cho SB, Park SE, Lee CM, Park JH, Baek HJ, Ryu KH, et al. Splenic artery pseudoaneurysm with splenic infarction induced by a benign gastric ulcer: a case report. Medicine (Baltimore). 2018;97: e11589. https://doi.org/10.1097/MD.0000000000011589.
Menaria P, Muddana V. Unusual case of life-threatening gastrointestinal bleed from a splenic artery pseudoaneurysm: case report and review of literature. Case Rep Gastrointest Med. 2019;2019:8528906. https://doi.org/10.1155/2019/8528906.
Nakata T, Okishio Y, Ueda K, Nasu T, Kawashima S, Kunitatsu K, et al. Life-threatening gastrointestinal bleeding from splenic artery pseudoaneurysm due to gastric ulcer penetration treated by surgical hemostasis with resuscitative endovascular balloon occlusion of the aorta: a case report. Clin Case Rep. 2022;10: e05561. https://doi.org/10.1002/ccr3.5561.
Defreyne L, Schrijver ID, Decruyenaere J, Maele GV, Ceelen W, Looze DD, et al. Therapeutic decision-making in endoscopically unmanageable nonvariceal upper gastrointestinal hemorrhage. Cardiovasc Int Radiol. 2008;31:897–905. https://doi.org/10.1007/s00270-008-9320-x.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Funding
No funding body was involved in the design of the study; collection, analysis, and interpretation of data; or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
HS and NF conceived of the idea, developed the theory, and performed the operations. KT, TA, KN, TN, and MN encouraged investigation of specific aspects and supervised the findings of this study. All authors discussed the results and contributed to the final manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The ethics committee of our institution approved all procedures used in this study.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shidahara, H., Fujikuni, N., Tanabe, K. et al. Massive bleeding from gastric ulcer-induced splenic artery pseudoaneurysm successfully treated with transcatheter arterial embolization and surgery: a case report. surg case rep 8, 196 (2022). https://doi.org/10.1186/s40792-022-01552-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40792-022-01552-0