Abstract
Chitobiose (COS2) efficiently lowers lipids in vivo and facilitates butyric acid enrichment during human fecal fermentation. However, whether COS2 can interact with butyric acid to generate a hypolipidemic effect remains unclear. This study examined the hypolipidemic mechanism of COS2 involving butyric acid, which could alleviate non-alcoholic fatty liver disease (NAFLD). The results revealed that COS2 administration modulated the β-oxidation pathway in the liver and restructured the short chain fatty acids in the fecal of ob/ob−/− mice. Moreover, the hypolipidemic effect of COS2 and its specific accumulated metabolite butyric acid was verified in sodium oleate-induced HepG2 cells. Butyric acid was more effective to reverse lipid accumulation and up-regulate β-oxidation pathway at lower concentrations. Furthermore, structural analysis suggested that butyric acid formed hydrogen bonds with key residues in hydrophilic ligand binding domains (LBDs) of PPARα and activated the transcriptional activity of the receptor. Therefore, the potential mechanism behind the lipid-lowering effect of COS2 in vivo involved restoring hepatic lipid disorders via butyric acid accumulation and liver–gut axis signaling.
Graphical Abstract

Similar content being viewed by others
Introduction
Non-alcoholic fatty liver disease (NAFLD) is defined as lipid accumulation (more than 5%) in hepatocytes without alcohol consumption, viral infection, or other known pathogenic factors. NAFLD includes simple lipid steatosis, non-alcoholic steatohepatitis (NASH), and advanced fibrosis (Vernon et al. 2011) and is the leading cause of liver disease with a prevalence of 25.24% worldwide (Younossi et al. 2016) and 29.2% in China (Zhou et al. 2019). However, the pharmaceutical therapies available for NAFLD clinical treatment are limited due to complex and diverse etiologies and extended disease development. Therefore, treating and preventing NAFLD and chronic liver disease depend on the availability of safe, effective, and diverse therapeutic agents, the development of which is crucial.
Chitosan oligosaccharides (COS) are chitosan degradation products that can regulate body weight and lipid metabolism by modifying the dysfunctional gut microenvironment (He et al. 2020; Wang et al. 2020). Previous studies have shown that COS reduces the intracellular triglyceride (TG) levels in oleic acid-induced HepG2 cells (Cao et al. 2016), displays anti-obesity activity, and improves serum and liver lipid profile abnormalities in high-fat diet-induced C57BL/6N mice (Choi et al. 2012; Li et al. 2022a, b), highlighting the potential of COS in preventing NAFLD. Studies have increasingly focused on the molecular COS mechanism involved in the hypolipidemic effect, including lipid uptake regulation, de novo synthesis, and free fatty acid (FFAs) β-oxidation (Liu et al. 2021; Tao et al. 2019; Zheng et al. 2018). The dimer COS is one of the most promising COS monomers (DP = 2 ~ 6) for alleviating lipid accumulation by decreasing the hepatic lipid uptake and de novo synthesis in oleic acid-induced HepG2 cells (Li et al. 2018). Shen et al. demonstrated that COS2 and COS3 reversed dyslipidemia by reducing the hepatic FFAs’ uptake and de novo synthesis of TG by downregulating the mRNA and protein levels of CD36 and DGAT2 in ob/ob−/− mice (Shen et al. 2021). The regulation of FFAs β-oxidation is also essential for limiting lipid accumulation. The main β-oxidation pathways are as follows: firstly, FFAs were activated and transferred to fatty acyl-CoA by ACSL1 in the outer mitochondrial membrane. Then, carnitine palmitoyl transferase 1A (CPT1A) and carnitine palmitoyl transferase 2 (CPT2), stimulated by PPARα, promote fatty acyl-CoA to cross the mitochondrial membrane and oxidized in the mitochondrial matrix finally under the effects of oxidation enzymes including PPARα (Huh et al. 2020). However, the potential intervention mechanism of COS2 during β-oxidation remains unclear.
Focusing on lipid metabolism regulation is insufficient to elucidate the mechanism behind NAFLD alleviation. Studies have suggested that the "multiple parallel hit" is more suitable for NAFLD pathogenesis (Buzzetti et al. 2016; Tilg et al. 2021). The cause of NAFLD includes hepatic lipid metabolism disruption and is strongly associated with the alteration of gut flora and their metabolic products (Li et al. 2022a, b). Therefore, the interaction and communication of the liver–gut axis cannot be disregarded during NAFLD intervention. COS reportedly prevents metabolic syndrome by promoting the growth of beneficial intestinal bacteria and decreasing the abundance of inflammogenic taxa in HFD mice (Qian 2019). Additionally, gut metabolite alteration is crucial in the NAFLD intervention mechanism. Research has shown that oral supplementation with butyric acid protects mice and rats from hepatic inflammation and lipid steatosis (Hattori et al. 2022; Jin et al. 2015; Sun et al. 2018; Zhao et al. 2021). Therefore, this study hypothesizes that the effects of COS2 in NAFLD were under the promotion of butyric acid in vivo. Butyric acid then circulates into the liver via the enterohepatic axis, playing a role in restoring lipid metabolism.
The objective of this study is to elucidate whether COS2 is involved in the liver–gut axis to alleviate NAFLD and the role the primary secondary metabolite, butyric acid, plays in COS2 enrolled intervention. NAFLD mice and cell models were established to evaluate the anti-hyperlipidemic effect of COS2 and butyric acid. Additionally, the gene and protein levels of the targets related to the β-oxidation pathway were explored to reveal the intervention mechanism. Results will indicate whether the anti-NAFLD effect of COS2 is positively related to butyric acid, which mainly contributes to β-oxidation activation.
Methods
Materials
Briefly, highly efficient and specific chitosanase was developed and used to obtain a series of COS with different degrees of polymerization (Luo et al. 2020). These products were then separated and desalted via ultrafiltration and nanofiltration to produce COS2 (purity > 95%, degree of deacetylation > 95%). The corresponding liquid phase diagram is shown in Additional file 1: Figures S1.
The sodium butyrate (NaB) was purchased from Sigma-Aldrich Chemical (St. Louis, MO, USA), while the tyrisin was obtained from Haoyang Biological Manufacture Co., Ltd. (Tianjin, China). The TG, total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C) assay kits were provided by the Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The total RNA isolation kit and HiScript® III RT SuperMix for the quantitative polymerase chain reaction (qPCR) (+ gDNA wiper) were purchased from Vazyme Biotech Co., Ltd (Nanjing, China). Mei5 Biotechnology, Co., Ltd (Beijing, China) provided the 2X M5 HiPer SYBR Premix EsTaq (with Tli RNaseH), while the RIPA lysis buffer was acquired from BioSharp (Anhui, China). The BCA Protein Assay Kit was purchased from CoWin Biosciences (CWBIO). The CPT1A, CPT2, and GAPDH polyclonal antibodies were acquired from the Proteintech Group, Inc (USA), while the secondary goat-anti-rabbit IgG-HRP antibodies were provided by Abmart, Inc (Shanghai, China). All reagents were analytically pure and obtained from commercial suppliers.
Animal models
Specific-pathogen-free (SPF) C57BL/6-leptin knock-off male mice (4 weeks old), in an initial weight range of 21 ~ 25 g, were purchased from Jiangsu Jicui Yaokang Biotechnology Co., Ltd. (Animal Certificate No. 201908218). The animal treatments involved in this study and their experiments were conducted in accordance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines and the National Research Council's Guide for the Care and Use of Laboratory Animals. These experiments had been reviewed by the Experimental Animal Ethics Committee of Jiangnan University (JN. No20190930b0560215). All animals were housed in standard laboratory conditions (22 °C \(\pm\) 3 °C, 12 h light–dark cycle) with free access to clean drinking water while receiving a standard diet (XieTongShengWu Ltd, Nanjing, Jiangsu, China, no. 101009) for ten weeks. COS2 were dissolved with 0.9% NaCl, while all the solutes were dissolved to the same volume. After an adaptation phase of one week, the animals were randomly divided into three groups (n = 8): The model group was gavaged with 0.9% NaCl as a solvent control. The other two groups received different COS2 dosages of 500 mg kg−1 day−1 (COS2-H) and 250 mg kg−1 day−1 (COS2-L) via intragastric administration. During the intervention, the body weights were measured. At the end of the experiment, all the animals were killed after an 18-h fasting period. Blood samples were collected and centrifuged at 106 g/min for 15 min at 4 °C to separate the serum, after which the liver tissue was collected and weighed. The biological samples were stored at −80 °C for further examination.
Analysis of fecal SCFAs
To determine the contents of SCFAs in mouse fecal samples, 50 mg of fecal sample was weighed accurately and mixed vigorously with 400 μL of sterile water. Then, 50 μL of 50% sulfuric acid was added for sufficient acidification, followed by the addition of 200 μL of ethyl acetate. The mixture was vortexed vigorously and allowed to stand for 2 min. Afterward, it was centrifuged at 8000 g and low temperature for 15 min, and the upper organic phase was collected for SCFAs quantitative analysis. SCFAs were determined by gas chromatography using an Agilent 7890 A gas chromatograph equipped with a flame ionization detector (FID) (California, United States) and an HP-5MS polar column (0.25 μm × 0.25 mm × 30 m). The temperature program was set as follows (Ji et al. 2022): the initial column temperature was 140 °C for 10 min, then ramped at a rate of 5 °C/min to 165 °C and held for 2 min, and finally ramped at a rate of 25 °C/min to 270 °C and held for 2 min. The detector temperature was set at 280 °C, and the injection port temperature was set at 250 °C. The qualitative and quantitative analysis of SCFAs, including acetic acid, propionic acid, and butyric acid, were performed using the standard curve method.
Cell culture
HepG2 cells were cultured in Minimum Essential Medium (Gibco, Grand Island, NY) with 10% fetal bovine serum (Gibco, Grand Island, NY), 1% penicillin–streptomycin solution (Haoyang Biological Manufacture Co., Ltd., Tianjin, China), non-essential amino acids, and sodium pyruvate solution (Sigma-Aldrich Chemical, St. Louis, MO). The cells were incubated at 37 °C in a 5% CO2 atmosphere.
Sodium oleate was used to induce NAFLD cell model with the concentration of 0.01 mM. The application of COS2 (0.02 mΜ, 0.2 mΜ, 0.5 mM, 1 mM) or NaB (0.01 mM, 0.1 mM, 0.3 mM, 0.5 mM) was undertaken into NAFLD cell model. After 24 h of incubation, cells were harvested and samples were prepared.
Biochemical indicators and histological analysis
Enzyme-linked immunosorbent assay (ELISA) kits were used to determine the lipid indicators of serum and liver of mice. The blood samples were centrifuged at 106 g for 15 min and the supernatant was collected and stored at −80 °C. The liver tissues were accurately weighed, mixed with a ninefold weight of ethanol, and homogenized at 4 °C for 120 s (60 Hz) to obtain the 10% homogenate. A commercial kit was used to analyze the TG, TC, LDL-C, and HDL-C levels in the serum and liver tissue samples according to the standard protocols. A histological section of the liver and HepG2 cells were immersed and dyed with Oil Red O solution to detect the lipid accumulation. Photos were taken at 400 × magnification using a Nikon Eclipse TI fluorescent microscope (Nikon, Tokyo, Japan).
Real-time PCR
The total RNA was extracted from the liver and HepG2 cells using a total RNA isolation kit according to the instructions of the manufacturer. The total RNA was reversely transcribed into cDNA using HiScript® III RT SuperMix. The qPCR primers were synthesized by Sangon Biotech (Shanghai, China), and the sequences are listed in Additional file 1: Table S1. The qPCR mixture was prepared using 2 × M5 HiPer SYBR Premix EsTaq (with Tli RNaseH), while the reaction was performed via CFX96 Touch Real-Time PCR (Bio-Rad, CA, USA). All procedures were conducted according to the instructions of the manufacturers. The Cycle threshold (Ct) values of each target gene were obtained and calculated after normalizing the housekeeping gene at 2−ΔΔct.
Western blot
The liver tissues and cells were lysed and homogenized at 4 °C using RIPA lysis buffer, phosphatase inhibitors, and protease inhibitors. The homogenate was centrifuged at 15,000 g for 15 min at 4 °C. The protein concentration was determined using a BCA protein assay kit. The protein samples were separated via 10% SDS-PAGE and then transferred to a polyvinylidene difluoride (PVDF) membrane (Merck Millipore). The membrane was blotted in Tris-buffered saline with tween 20 (TBST) containing 5% skim milk for 1 h at room temperature. Then, the polyclonal antibodies were diluted to 1:1000 with primary antibody dilution buffer, and the membranes were incubated overnight at 4 °C, followed by incubation with horseradish peroxidase-conjugated secondary antibodies (1:3000) for 1 h. The target bands were scanned using a Tannon automatic chemiluminescence image analysis system and examined via Image J software.
Molecular docking simulation
The PPARα protein (PDB code: 2ZNN) was downloaded on Protein Data Bank (RCSB PDB: Homepage). The original ligand was removed by PyMOL (Version 2.1.1_0) to prepare PPARα crystal structure. The molecule structure of COS2 and butyric acid were mapped using ChemBio3D Ultra 14.0. The ligand docking center and the size of the grid box were determined on the native ligand and obtained by AutoDockTools 1.5.6. The docking studies were performed by AutoDock Vina 1.1.2 (Trott et al. 2010). The results were analyzed by PyMOL.
Statistical analysis
The experimental data were generated with GraphPad Prism 8.0 (GraphPad Software, San Diego, USA) and expressed as mean ± SD. The Oil Red O staining images were analyzed using Image J statistical software via one-way ANOVA with multiple comparisons to show the differences between groups. P < 0.05 were considered significant.
Results
COS 2 decreased lipid deposition in the serum and liver of ob/ob −/− mice
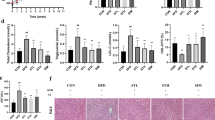
Ten weeks of COS2 intervention distinctly reduced the body weight gain of the ob/ob−/− mice (Fig. 1A, p < 0.05) and decreased the liver index compared with the model group (p < 0.05) (Fig. 1B). The TG, TC, and LDL-C levels in the serum were improved after COS2 intervention (Fig. 1C, D, E), while the HDL-c levels increased (Fig. 1F, p < 0.05). Moreover, the lipid droplets in the liver were smaller in the COS2 treatment groups than in the model group (Fig. 2A). The lipid profiles in the liver, including TG, TC, and LDL-C, decreased while the HDL-C level increased after COS2 administration (Fig. 2B, C, D, E, p < 0.05). These results indicated that COS2 ameliorated lipid deposition in the serum and liver of ob/ob−/− mice.
COS 2 regulated lipid metabolism to promote the lipid β-oxidation levels of ob/ob −/− mice
Farnesoid X receptor (FXR) is crucial for lipid homogenesis and protects the liver from lipid accumulation and hepatic steatosis (Schmitt et al. 2015). This study showed a distinct increase in the hepatic FXR gene expression level in the ob/ob−/− mice at high COS2 gavage concentrations (Fig. 3A, p < 0.05), accounting for the inhibition of lipid uptake and synthesis shown by the previous results (Shen et al. 2021), and contributing to FFA β-oxidation upregulation (Xi et al. 2020). Although the β-oxidation pathways helped eliminate excessive FFAs, the role of COS2 in FFAs β-oxidation regulation remains unclear. Acyl-CoA synthetase long-chain family member 1 (ACSL1) is an enzyme located in the outer mitochondrial membranes, responsible for transferring fatty acids to acyl-CoA (Coleman et al. 2000; Huh et al. 2020). High-dose COS2 treatment significantly increased the ACSL1 mRNA levels in the livers of the ob/ob−/− mice (Fig. 3B, p < 0.05). PPARα, induced by FXR, is a transcription factor that regulates FFAs β-oxidation (Fuchs et al. 2016; Sinal et al. 2001), the gene expression of which was increased by COS2 treatment (Fig. 3C, p < 0.05). As the downstream target of PPARα, CPT1A and CPT2 represent essential regulators for FFA translocation into the mitochondria, denoting the rate-limiting steps of mitochondrial β-oxidation (Xi et al. 2020). COS2 significantly increased the mRNA levels and CPT1A and CPT2 protein content in a dose-dependent manner (Fig. 3D, E, G, p < 0.05). Furthermore, ACOX1 represents the first enzyme in the FFA β-oxidation pathway. High-dose COS2 treatment upregulated the ACOX1 gene level, facilitating FFA oxidation in the liver (Fig. 3F). Therefore, COS2 accelerated FFA conversion and translocation in the liver and increased the mitochondrial β-oxidation rate.
COS 2 regulated the synthesis of SCFAs, leading to a significant increase in the production of butyrate in ob/ob −/− mice
Butyric acid is an SCFAs fermented from dietary fibers by gut microbiota in the colon. Differential analysis was performed on the content of SCFAs in the feces of mice from different treatment groups (Fig. 4). Acetic acid and propionic acid serve as substrates for hepatic gluconeogenesis, and enter the hepatic portal system through intestinal absorption, promoting hepatic gluconeogenesis metabolism and providing energy for liver metabolism. Compared with the model group (Fig. 4A), the acetic acid content in the COS2 high and low dose groups increased significantly (p < 0.05). The propionic acid content in the COS2 high and low dose groups increased significantly (Fig. 4B, p < 0.05). Butyrate is the energy source for intestinal epithelial cells and can have a preventive and therapeutic effect on NAFLD and T2DM. Compared with the model group, the butyric acid content in the COS2 high-dose group increased significantly (Fig. 4C), which is consistent with the results of previous reports on the changes in gut microbiota induced by COS2 (Ji et al. 2021, 2022). Analysis of the total SCFAs content in the intestine of COS-intervened model mice revealed a significant increase in the COS2 treated group.
Recent studies have found that butyric acid can exhibit functions and impact other tissues and organs beyond the gut via the enterohepatic circulation (van der Hee et al. 2021). Our previous research showed that COS2 enhanced butyric acid accumulation by promoting the abundance of Clostridium_sensu_stricto_1, Clostridium_sensu_stricto_13, and Fusobacterium (Ji et al. 2021, 2022). Moreover, butyric acid decreased lipogenesis to alleviate PPARγ in the liver and reversed lipid accumulation via the liver–gut axis (den Besten et al. 2015). Therefore, it can be considered a potential hypolipidemic biomarker during COS2 metabolism in the colon. The role of COS2 during FFAs β-oxidation was correlated with butyric acid to determine its potential lipid-lowering mechanism in conjunction with COS2.
The hypolipidemic effect of COS 2 and the typical metabolite modulated by COS 2 in sodium oleate-induced HepG2 cells
HepG2 cells were incubated with 0.01 mM sodium oleate to obtain the NAFLD cell model. Lipid accumulation was evident in the model group, suggesting the successful establishment of the NAFLD model (Figs. 5, 6, p < 0.05). To examine the lipid-lowering effect of COS2 and butyric acid, the NAFLD cell model was subjected to different COS2 and NaB doses and incubated for 24 h. And the cell availability was detected (the data are shown in Additional file 1: Fig. S2). The Oil Red O staining results indicated that COS2 and NaB intervention significantly alleviated lipid accumulation (Figs. 5D and 6D), while only high COS2 doses (Fig. 5A, B, C, 0.2 mM, 0.5 mM, and 1 mM) reversed the TG, TC, and LDL-C levels in the NAFLD cell model. However, a less distinct lipid-lowering effect was evident in the model group at the lowest COS2 concentration (Fig. 5A, B, C, 0.02 mM). However, after NaB treatment, the TG, TC, and LDL-C levels decreased significantly, induced by sodium oleate (Fig. 6, p < 0.05).
COS 2 and NaB restored lipolysis in sodium oleate-induced HepG2 cells
The mRNA levels of the lipid β-oxidation pathway involving PPARα and its downstream targets, CPT1A and ACOX1, were inhibited in the NAFLD model compared to the control group (Fig. 7A, B, C, p < 0.05). All NaB doses and high COS2 doses elevated the mRNA levels of PPARα, indicating potential lipolysis stimulation, while low COS2 doses showed less effects to activate PPARα gene levels compared to the model group (Fig. 7A, p < 0.05). Therefore, low COS2 doses were less successful than NaB in reversing the PPARα protein levels (Fig. 7D). Similarly, all NaB doses and high COS2 doses were more effective in reversing CPT1A gene and protein levels, while low COS2 concentrations were less successful in increasing CPT1A (Fig. 7B, E). Furthermore, all COS2 and NaB doses restored CPT2 protein expression (Fig. 7F). Moreover, ACOX1, a rate-limiting enzyme involved in FFAs β-oxidation, had been more facilitated by NaB than COS2 in mRNA levels (Fig. 7C). Consequently, NaB was superior to COS2 at low concentrations in promoting oxidative lipid metabolism. Therefore, the butyric acid in the liver–gut axis may exert a more positive effect, warranting further attention.
Structures of COS 2 and butyric acid bound to PPARα
PPARα involves in fatty acid metabolism and can be activated by ligands binding to its LBDs (Oyama et al. 2009). The LBDs of PPARα including hydrophilic, hydrophobic and amphiphilic pockets forming Y-shape domains (Han et al. 2020). The optimal docking modes of COS2 suggested that the ligand mainly contact with H3, H2’ helix and β3 strand at the hydrophobic pocket of PPARα, which is the entrance of the LBDs (Fig. 8A). While due to the size and hydrophily of butyric acid, the hydrophilic pocket between Helix 3 and Helix 12 including the activation factor-2 (AF-2) domain was occupied and hydrogen bonds were formed with amino acid residues Tyr464 on Helix 12, Try314 on Helix 5, Ser280 on Helix 3 and His440 on helix 11 (Fig. 8B, C). These interactions stabilized AF2 helix to recruit co-activators of receptor and facilitated the transcriptional activity of PPARα (Capelli et al. 2016; Han et al. 2020; Xu et al. 2001). Thus, these results well explained that butyric acid could be more effective than COS2 at low concentrations in promoting PPARα expression and stimulating oxidative lipid metabolism.
Discussion
This study proposed that the lipid-lowering mechanism of COS2 was related to a specific metabolite, butyric acid. COS2 effectively alleviated lipid dysfunction by regulating the mitochondrial β-oxidation pathways in ob/ob−/− mice. Additionally, COS2 exhibited a prebiotic effect by facilitating the accumulation of butyric acid, a specific metabolite with the potential to relieve hepatic lipid abnormality more effectively. Therefore, this study was extended to examine the lipid-lowering mechanism of COS2.
NAFLD diagnosis typically occurs when lipid accumulation accounts for 5% of the weight of the liver. Lipid metabolism dysbiosis represents the main pathogenic factor of NAFLD and can manifest in various ways, such as lipid uptake, de novo synthesis, and oxidative metabolism. It is been proved that COS2 can be absorbed and circulated through blood and exhibited its effects in liver (Chen et al. 2022). Our previous studies showed that COS2 downregulated hepatic lipogenesis-related targets and reduced lipid uptake (Shen et al. 2021). In this study, we found that COS2 enhanced the FFA oxidation process in the liver to reverse NAFLD lipid accumulation in the ob/ob−/− mice. Mitochondrial FFA β-oxidation represents the main lipid oxidation pathway. The upstream regulator of lipid metabolism is FXR, a ligand-activated receptor belonging to the nuclear receptor superfamily and essential for regulating FFA β-oxidation by controlling PPARα (Proctor et al. 2006; Kast et al. 2001; Li et al. 2021; Xi et al. 2020). COS2 improved FXR gene expression, activating liver lipolysis (Fig. 3A). PPARα, the downstream target of FXR, represents the key enzyme that controls the FFA β-oxidative system. As shown in Fig. 3C, high COS2 doses significantly upregulated the PPARα mRNA levels. Moreover, the mitochondrial entry process represented the FFA β-oxidation rate-limiting step and mainly occurred via ACSL1, CPT1A, and CPT2 catalysis. The FFAs were activated to form acyl-CoA via ACSL1 (Huh et al. 2020), which was transferred successfully into the mitochondria with the help of CPT1A and CPT2, finally initiating the β-oxidation process (Xi et al. 2020). COS2 promoted the expression of gene and protein levels of ACSL1, CPT1A, and CPT2 to facilitate the rate-limited process of lipolysis, demonstrating the internal mechanism of COS2 in improving hepatic lipid metabolism of NAFLD in ob/ob−/− mice.
In addition, COS2 has been found to significantly increase the production of butyric acid by enriching the microbial communities in the intestinal microenvironment. In an in vitro anaerobic fermentation study using fecal samples from healthy individuals, COS2 was shown to promote the growth of functional microbial communities, such as Clostridium_butyricum, Clostridium, and Parabacteroides, which are known to produce substantial amounts of butyric acid (Ji et al. 2022). Similarly, in the fermentation of fecal samples obtained from patients with NAFLD, COS2 enriched the core functional microbial communities responsible for butyric acid production, including Clostridium sensu stricto 13, Parabacteroides, Romboutsia, Holdemanella, Bacteroides, Bacterium NLAE zl_G201, Erysipelatoclostridium, and Lactococcus (Ji et al. 2021). Moreover, COS2 and COS3 were found to promote the growth of intestinal probiotics such as Bifidobacterium and Lactobacillus in ob/ob model mice and significantly increase the abundance of functional microbial communities, such as Akkermansia, Clostridiales, Faecalibaculum, Roseburia, Ruminiclostridium, and Alistipes, which are also known to produce butyric acid. Previous study reported that butyrate had been reported to induce the reduction of lipid accumulation (Zong et al. 2023). For the intervention mechanism investigation, NaB-induced PPARα activation stimulates fatty acid β oxidation, thus contributing to amelioration of high-fat diet-induced NAFLD in adult rats (Sun et al. 2018). Thus, enterohepatic circulation is possibly responsible for the COS2 anti-NAFLD effect.
Due to the complexity of NAFLD pathogenesis, the following hypotheses could explain the anti-NAFLD impact of COS2: 1. COS2 alleviated hepatic lipid accumulation via direct liver lipid homeostasis. 2. COS2 increased the butyrate levels, producing a distinct hepatic anti-hyperlipidemic effect via enterohepatic circulation. To confirm this, HepG2 cell experiments were established using sodium oleate to induce lipid accumulation and create a NAFLD cell model. The metabolic kinetics study results of COS2 revealed a physiological concentration of 0.02 mM in the serum of the rats after intragastric administration of 500 mg kg−1. The physiological butyric acid concentration was around 3 μM in human serum (Behary et al. 2021) and between 26 μM to 48 μM in the portal serum of mice (Jakobsdottir et al. 2013). Therefore, 0.02 mM COS2 and 0.01 mM NaB were selected as the low dose interventions in the NAFLD cell model, showing that both treatments displayed anti-hyperlipidemia activity. However, at the physiological concentration, NaB was more successful in restoring the lipid levels, as well as the gene and protein expression of PPARα, CPT1A, ACOX1, and CPT2 than COS2. Moreover, in molecular docking analysis, the results showed that COS2 interacted with PPARα in hydrophobic pocket and stuck in the entrance of LBDs of PPARα protein. While butyric acid could crush into the hydrophilic pocket and form hydrogen bonds with AF2 domain, which was important for the receptor to activate the transcriptional activity (Xu et al. 2001). Therefore, butyric acid was superior in stimulating FFA β-oxidation and preventing lipid accumulation. The intestinal microenvironment and enterohepatic circulation status are crucial for NAFLD remission. COS2 accelerated the FFA β-oxidation pathway by promoting butyric acid production in the intestinal tract, reaching the liver via enterohepatic circulation, and binding to the FFA β-oxidation targets PPARα to improve lipid metabolism abnormalities.
Conclusion
COS2 was found to induce the FFA β-oxidation pathway, which mitigated NAFLD by regulating gut microenvironment, particularly butyric acid metabolism, and its interaction with hepatic lipid metabolism. The results provide new insights into the mechanism of COS2 in lipid-lowering effects.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- COS2 :
-
Chitobiose
- NAFLD:
-
Non-alcoholic fatty liver disease
- LBDs:
-
Ligand binding domains
- NASH:
-
Non-alcoholic steatohepatitis
- COS:
-
Chitosan oligosaccharides
- TG:
-
Triglyceride
- FFAs:
-
Free fatty acid
- CPT1A:
-
Carnitine palmitoyl transferase 1A
- CPT2:
-
Carnitine palmitoyl transferase 2
- SCFAs:
-
Short-chain fatty acids; NaB: Sodium butyrate
- TC:
-
Total cholesterol
- LDL-c:
-
Low-density lipoprotein cholesterol
- HDL-c:
-
High-density lipoprotein cholesterol
- SPF:
-
Specific-pathogen-free
- PVDF:
-
Polyvinylidene difluoride
- TBST:
-
Tris-buffered saline with tween 20
- FXR:
-
Farnesoid X receptor
- ACSL1:
-
Acyl-CoA synthetase long-chain family member 1
References
Behary J, Amorim N, Jiang XT, Raposo A, Gong L, McGovern E, Ibrahim R, Chu F, Stephens C, Jebeili H, Fragomeli V, Koay YC, Jackson M, O’Sullivan J, Weltman M, McCaughan G, El-Omar E, Zekry A (2021) Gut microbiota impact on the peripheral immune response in non-alcoholic fatty liver disease related hepatocellular carcinoma. Nat Commun 12:187–199
Buzzetti E, Pinzani M, Tsochatzis EA (2016) The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 65:1038–1048
Cao PQ, Huang GD, Yang QY, Guo J, Su ZQ (2016) The effect of chitooligosaccharides on oleic acid-induced lipid accumulation in HepG(2) cells. Saudi Pharm J 24:292–298
Capelli D, Cerchia C, Montanari R, Loiodice F, Tortorella P, Laghezza A, Cervoni L, Lavecchia PG, A, (2016) Structural basis for PPAR partial or full activation revealed by a novel ligand binding mode. Sci Rep 6:34792–34804
Chen J, Ji C, Li and Zhao, (2022) Pharmacokinetics, bioavailability and tissue distribution of chitobiose and chitotriose in rats. Biores Bioprocess 9:1–15
Choi EH, Yang HP, Chun HS (2012) Chitooligosaccharide ameliorates diet-induced obesity in mice and affects adipose gene expression involved in adipogenesis and inflammation. Nutr Res 32:218–228
Coleman RA, Lewin TM, Muoio DM (2000) Physiological and nutritional regulation of enzymes of triacylglycerol synthesis. Annu Rev Nutr 20:77–103
Den Besten G, Bleeker A, Gerding A, van Eunen K, Havinga R, van Dijk TH, Oosterveer MH, Jonker JW, Groen AK, Reijngoud DJ, Bakker BM (2015) Short-Chain Fatty Acids Protect Against High-Fat Diet-Induced Obesity via a PPARγ-Dependent Switch From Lipogenesis to Fat Oxidation. Diabetes 64(7):2398–2408
Fuchs CD, Traussnigg SA, Trauner M (2016) Nuclear receptor modulation for the treatment of nonalcoholic fatty liver disease. Semin Liver Dis 36(1):69–86
Gijs B, Aycha B, Albert G, Karen E, Rick H, Theo H, Maaike HO, Johan WJ, Albert KG, Dirk-Jan R, Barbara MB (2015) Short-chain fatty acids protect against high-fat diet-induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation. Diabetes 64:2398–2408
Han Y, Liu J, Ahn S, An S, Ko H, Shin JC, Jin SH, Ki MW, Lee SH, Lee KH, Shin SS, Choi WJ, Noh M (2020) Diallyl biphenyl-type neolignans have a pharmacophore of PPARα/γ dual modulators. Biomol Ther (seoul) 28:397–404
Hattori Y, Tsutsui S, Yamada C (2022) Dietary supplementation with sodium butyrate reduces high-sucrose diet-induced hepatic accumulation of triacylglycerols and expression of fatty acid synthesis enzymes in rats. J Oleo Sci 71:1189–1193
He N, Wang S, Lv Z, Zhao W, Li S (2020) Low molecular weight chitosan oligosaccharides (LMW-COSs) prevent obesity-related metabolic abnormalities in association with the modification of gut microbiota in high-fat diet (HFD)-fed mice. Food Funct 11:9947–9959
Huh JY, Reilly SM, Abu-Odeh M, Murphy AN, Mahata SK, Zhang JY, Cho Y, Seo JB, Hung CW, Green CR, Metallo CM, Saltiel AR (2020) Tank-binding kinase 1 regulates the localization of acyl-CoA synthetase ACSL1 to control hepatic fatty acid oxidation. Cell Metab 32:1012–1027
Jakobsdottir G, Xu J, Molin G, Ahrne S, Nyman M (2013) High-fat diet reduces the formation of butyrate, but increases succinate, inflammation, liver fat and cholesterol in rats, while dietary fibre counteracts these effects. PLoS ONE 8:80476
Ji XG, Chen M, Zhao MY, Song YD, Lin Y, Yin ZLM (2021) Effects of chitooligosaccharides on the rebalance of gut microorganisms and their metabolites in patients with nonalcoholic fatty liver disease. J Funct Foods 77:104333
Ji XG, Zhu LL, Chang KL, Zhang R, Chen YJ, Yin H, Jin JY, Zhao LM (2022) Chitooligosaccahrides: digestion characterization and effect of the degree of polymerization on gut microorganisms to manage the metabolome functional diversity in vitro. Carbohyd Polym 275:118716
Jin CJ, Sellmann C, Engstler AJ, Ziegenhardt D, Bergheim I (2015) Supplementation of sodium butyrate protects mice from the development of non-alcoholic steatohepatitis (NASH). Brit J Nutr 114:1745–1755
Kast HR, Nguyen CM, Sinal CJ, Jones SA, Laffitte BA, Reue K, Gonzalez FJ, Willson TM, Edwards PA (2001) Farnesoid X-activated receptor induces apolipoprotein C-II transcription: a molecular mechanism linking plasma triglyceride levels to bile acids. J Mol Endocrinol 15:1720–1728
Li XD, Zhao MY, Fan LQ, Cao XN, Chen LH, Chen JH, Lo YM, Zhao LM (2018) Chitobiose alleviates oleic acid-induced lipid accumulation by decreasing fatty acid uptake and triglyceride synthesis in HepG2 cells. J Funct Foods 46:202–211
Li YQ, Tian YY, Cai WZ, Wang QH, Chang YG, Sun YH, Dong P, Wang JF (2021) Novel i-carrageenan tetrasaccharide alleviates liver lipid accumulation via the bile acid-FXR-SHP/PXR pathway to regulate cholesterol conversion and fatty acid metabolism in insulin-resistant Mice. J Agric Food Chem 69:9813–9821
Li T, Gong H, Zhan BY, Mao XY (2022a) Chitosan oligosaccharide attenuates hepatic steatosis in HepG2 cells via the activation of AMP-activated protein kinase. J Food Biochem 46:14045
Li XP, Yao Y, Wang Y (2022b) Effect of hesperidin supplementation on liver metabolomics and gut microbiota in a high-fat diet-induced NAFLD mice model. J Agric Food Chem 70:11224–11235
Liu SH, Chen RY, Chiang MT (2021) Effects and mechanisms of chitosan and chitosan oligosaccharide on hepatic lipogenesis and lipid peroxidation, adipose lipolysis, and intestinal lipid absorption in rats with high-fat diet-induced obesity. Int J Mol Sci 22:1139–1154
Luo S, Qin Z, Chen Q, Fan L, Jiang L, Zhao L (2020) High level production of a Bacillus amyloliquefaciens chitosanase in Pichia pastoris. Int J Biol Macromol 149:1034–1041
Oyama T, Toyota K, Waku T, Hirakawa Y, Nagasawa N, Kasuga JI, Hashimoto Y, Miyachi H, Morikawa K (2009) Adaptability and selectivity of human peroxisome proliferator-activated receptor (PPAR) pan agonists revealed from crystal structure. Acta Crystallogr D Biol Crystallogr 65:786–795
Proctor G, Jiang T, Iwahashi M, Wang Z, Li J, Levi M (2006) Regulation of renal fatty acid and cholesterol metabolism, inflammation, and fibrosis in akita and OVE26 mice with type 1 diabetes. Diabetes 55:2502–2509
Qian M, Lyu Q, Liu Y, Hu H, Wang S, Pan C, Duan X, Gao Y, Qi L, Liu W, Wang L (2019) Chitosan oligosaccharide ameliorates nonalcoholic fatty liver disease (NAFLD) in diet-induced obese mice. Mar Drugs 17:391–405
Schmitt J, Kong B, Stieger B, Tschopp O, Schultze SM, Rau M, Weber A, Müllhaupt B, Guo GL, Geier A (2015) Protective effects of farnesoid X receptor (FXR) on hepatic lipid accumulation are mediated by hepatic FXR and independent of intestinal FGF15 signal. Liver Int 35:1133–1144
Shen X, Liang XY, Ji XG, You JS, Zhuang XY, Song YD, Yin H, Zhao MY, Zhao LM (2021) CD36 and DGAT2 facilitate the lipid-lowering effect of chitooligosaccharides via fatty acid intake and triglyceride synthesis signaling. Food Funct 12:8681–8693
Sinal CJ, Yoon M, Gonzalez FJ (2001) Antagonism of the actions of peroxisome proliferator-activated receptor-α by bile acids. JBC 276:47154–47162
Sun B, Jia YM, Hong J, Sun QW, Gao SX, Hu Y, Zhao NN, Zhao RQ (2018) Sodium butyrate ameliorates high-fat-diet-induced non-alcoholic fatty liver disease through peroxisome proliferator-activated receptor alpha-mediated activation of beta oxidation and suppression of inflammation. J Agric Food Chem 66:7633–7642
Tao WJ, Sun WJ, Liu LJ, Wang G, Xiao ZP, Pei X, Wang M (2019) Chitosan oligosaccharide attenuates nonalcoholic fatty liver disease induced by high fat diet through reducing lipid accumulation, inflammation and oxidative stress in C57BL/6 mice. Mar Drugs 17:645–660
Tilg H, Adolph TE, Moschen AR (2021) Multiple parallel hits hypothesis in NAFLD - revisited after a decade. Hepatology 73:833–842
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31(2):455–461
van der Hee B, Wells JM (2021) Microbial regulation of host physiology by short-chain fatty acids. Trends Microbiol 29:700–712
Vernon G, Baranova A, Younossi ZM (2011) Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharm Ther 34:274–285
Wang Q, Jiang Y, Luo X, Wang C, Wang N, He H, Zhang T, Chen L (2020) Chitooligosaccharides modulate glucose-lipid metabolism by suppressing SMYD3 pathways and regulating gut microflora. Mar Drugs 18:69–84
Xi YF, Li HS (2020) Role of farnesoid X receptor in hepatic steatosis in nonalcoholic fatty liver disease. Biomed Pharmacother 121:109609
Xu HE, Lambert MH, Montana VG, Plunket KD, Moore LB, Collins JL, Oplinger JA, Kliewer SA, Gampe RT, McKee DD, Moore JT, Willson TM (2001) Structural determinants of ligand binding selectivity between the peroxisome proliferator-activated receptors. PNAS 98:13919–13924
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M (2016) Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64:73–84
Zhao ZH, Wang ZX, Zhou D, Han YM, Ma FG, Hu ZM, Xin FZ, Liu XL, Ren TY, Zhang FF, Xue YQ, Cui AY, Liu ZS, Bai JY, Liu YX, Cai GX, Su WT, Dai XZ, Shen F, Pan Q, Li Y, Fan JG (2021) Sodium butyrate supplementation inhibits hepatic steatosis by stimulating liver kinase B1 and insulin-induced gene. CMGH 12:857–871
Zheng JP, Cheng G, Li QY, Jiao SM, Feng C, Zhao XM, Yin H, Du YG, Liu HT (2018) Chitin oligosaccharide modulates gut microbiota and attenuates high-fat-diet-induced metabolic syndrome in mice. Mar Drugs 16:66–83
Zhou F, Zhou JH, Wang WX, Zhang XJ, Ji YX, Zhang P, She ZG, Zhu LH, Cai JJ, Li HL (2019) Unexpected rapid increase in the burden of NAFLD in China from 2008 to 2018: a systematic review and meta-analysis. Hepatology 70:1119–1133
Zong QF, Qu H, Zhao YH, Liu HY, Wu SL, Wang S, Bao WB, Cai DM (2023) Sodium butyrate alleviates deoxynivalenol-induced hepatic cholesterol metabolic dysfunction via RORγ-mediated histone acetylation modification in weaning piglets. J Anim Sci Biotechno 14:792–805
Acknowledgements
Not applicable.
Funding
This study was supported by the Shanghai Natural Science Foundation General program (23ZR1417300), Shanghai Post-doctoral Excellence Program (2022153), the National Key R&D Program of China (Grant Number 2019YFD090180302), and the 111 Project (B18022).
Author information
Authors and Affiliations
Contributions
ZXY and ZMY contributed equally to this work. ZXY and ZMY conceived and wrote the manuscript. ZXY, JXG and YSH performed the experiments. ZMY revised the manuscript, Funding acquisition, Project administration. YH: validation. ZLM: funding acquisition, project administration, and supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The Animal Care and Use Committee of Laboratory Animals provided research ethics approval [JN. No. 20190930b0560215(245)] License number: SYXK (Su) 2016-0045.
Consent for publication
The authors approved consent for publishing the manuscript.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1.
The HPLC-ELSD chromatogram of COS2. Figure. S2. The cell viability of COS2 and NaB treatment in SO-induced NAFLD cell model. Table S1. Primers of the target sequence.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhuang, X., Zhao, M., Ji, X. et al. Chitobiose exhibited a lipid-lowering effect in ob/ob−/− mice via butyric acid enrolled liver–gut crosstalk. Bioresour. Bioprocess. 10, 79 (2023). https://doi.org/10.1186/s40643-023-00696-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40643-023-00696-7