Abstract
The leaves of Stauntonia hexaphylla (SHL) are a very popular herbal medicine in Korea because it can be used to treat rheumatic osteoporosis and other diseases. However, owing to the inconsistency in harvesting time and growth years, their quality is uneven, which indirectly negatively affects the safety of this medication. Therefore, the difference of constituents in SHL harvested at different seasons and years were analyzed by high-performance liquid chromatography coupled to photodiode-array and electrospray ionization mass spectrometry detectors (HPLC–PDA–ESI/MS). A total of 15 components were tentatively characterized in samples of SHL, including 5 compounds reported for the first time in this plant. Moreover, the relative content of these constitutions was simultaneously determined by HPLC coupled with evaporative light scattering detection (ELSD). Hierarchical clustering analysis (HCA) and principal component analysis (PCA) revealed that the quality has a certain extent difference in different harvest times, the best harvest time was 3 years old growing in autumn. The same harvest time was also suggested based on the anti-inflammatory evaluation.
Similar content being viewed by others
Introduction
Stauntonia hexaphylla (Thunb.) Decne. (Lardizabalaceae) is a native shrub that grows on the southern coast of South Korea [1, 2]. Stauntonia hexaphylla leaves (SHL) have been used as medicine and functional food to treat diabetes, rheumatic arthralgia, and menstrual pain [3]. At the same time, according to modern pharmacological research, SHL also has many biological activities such as antioxidant, anti-inflammatory, and anti-osteoporosis properties [2,3,4]. Owing to its biological activities, many pharmaceutical companies in Korea have developed drugs and functional foods to treat prostate diseases, osteoporosis, and rheumatic diseases using SHL. Also, it is very common for people to use it as a health care product to prevent these diseases.
According to our previous research and reported literature, SHL contains many saponins, which are considered to be bioactive compounds with analgesic and anti-inflammatory activities [2, 3]. Besides, flavonoids and chlorogenic acid derivatives are essential for the treatment of rheumatic disease and osteoporosis [3,4,5]. However, there are various types of SHL on the market, and differences in their growth year and harvesting season make it difficult to develop clinically rational drugs. At the same time, there are several researches on the anti-inflammatory activity of SHL without a specific focus on the harvest season and growth year of the samples [2]. It is known that there are large differences in the chemical composition of medicinal materials depending on the growth year and harvesting season, and these differences may lead to different or even opposite efficacy. For example, some studies have determined that the content and type of ginsenosides from different growth years of ginseng were considerably different; thus, ginseng samples acquired from non-optimal growth years did not have an expected pharmacological effect such as anti-proliferative activity [6, 7].
Therefore, the purpose of this study is to identify the chemical composition of SHL from different growth years and harvesting seasons by high-performance liquid chromatography coupled to photodiode-array and electrospray ionization mass spectrometry detectors (HPLC–PDA–ESI–MS/MS) and establish an easy HPLC method coupled with chemometrics methods to discriminate SHL from different harvest seasons and growth years. In addition, we evaluated the anti-inflammatory activity of SHL from different growth years and harvesting seasons, which will provide an opportunity for the development, utilization, and quality control of SHL.
Materials and methods
Chemicals and materials
The extract samples of SHL were provided by Chong Kun Dang Healthcare (Seoul, Korea) (Table 1). The samples of SHL were harvested in the area of Goheung-gun, Jeollanam-do, Republic of Korea, at the geographical coordinates of 34° 38′ 22.8″ N 126° 56′ 34.1″ E, and authenticated by professor Jong Seong Kang and Young Ho Kim. An authentic sample (CNU 17002) of the collections was deposited at the Herbarium of Chungnam National University. The samples of SHL were dried at 60 °C for 12 h. After drying, the samples were crushed and extracted with 70% ethanol at 60 °C for 12 h and followed by concentrated and dried to obtain extract powder. The reference compounds of ciwujianoside A1, anhuienside C, ciwujianoside A2, mutongsaponin E, hedercoside D, hexaphyloside A, and ciwujianoside C3 (Fig. 1) were donated by the Laboratory of Natural Products, College of Pharmacy, Chungnam National University. Dexamethasone (DEX), neochlorogenic acid, chlorogenic acid, and cryptochlorogenic acid were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). HPLC-grade acetonitrile and methanol were purchased from Burdick & Jackson (Muskegon, MI, USA); a buffer containing formic acid, acetic acid, and trifluoroacetic acid (MS grade) was purchased from Sigma-Aldrich (St. Louis, Mo, USA). Distilled water was purified using a Milli-Q system (Sinhan, Seoul, Korea). Escherichia coli lipopolysaccharide (LPS) and 3-(4,5-dimethylthiazoL-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) were purchased from Sigma-Aldrich Chemie (St. Louis, MO, USA).
Sample and standard preparation
To completely dissolve the extract of SHL, 2.25 g of samples was mixed with 50 mL of 50% methanol in a conical flask and sonicated at 50 °C for 60 min by an SD-D400H Mujigae ultrasonic machine (Seoul, Korea). All reference standards were prepared at the concentrations of 100 μg/ml in a methanol solution. Then, the extract and standard solution were store in a refrigerator at 4 °C before use. In addition, reference standards and sample solutions were filtered through a 0.22 μm syringe filter before being injected into the HPLC system.
HPLC analysis conditions
To analysis the different types of components in SHL, the samples were analyzed with an HPLC–UV system (Shimzadu LC-20A series system) coupled with SEDEX 55 ELSD. A Hector C18 column (250 × 4.6 mm, 5 μm, RStech, Daejeon, Korea) was employed as the stationary phase. The optimized flow rate of a carrier N2 gas and drift tube temperature were 1.5 L/min and 65 °C, respectively. Solvent A (water, 0.1% formic acid, v/v) and solvent B (acetonitrile, 0.1% formic acid, v/v) were employed for gradient elution according to the following program: 12–28% B (0–30 min) and 28–40% (30–70 min). The column temperature and flow rate were set to 30 °C and 1.0 ml/min, respectively.
The HPLC–PDA–ESI–MS/MS analysis was carried out using a Shimadzu LCMS-8040 system (Kyoto, Japan) connected to a photodiode array (PDA) detector. Electrospray ionization (ESI) mass spectrometry was performed at the interface voltage of − 3.5 kV for the negative mode and at 4.5 kV for the positive mode. The parameters of optimized ionization conditions are as follows: drying gas, 15 L/min; desolvation line temperature, 250 °C; heat block temperature, 400 °C; nebulizing gas, 3 L/min. The scan range of MS1 was set up at m/z 100–1500. The wavelength range of PDA was set up at 190–400 nm. The MS/MS analysis was automatically performed by a product ion survey scan at positive and negative modes. The elution program was the same as the one used previously.
Cell culture
Macrophage cell lines RAW 264.7 were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA), and they were cultured in Dulbecco’s modified Eagle’s medium (Sigma-Aldrich, MO, USA) supplemented with 10% fetal bovine serum (GIBCO, NY, USA) and 1% penicillin–streptomycin in an incubator with 5% CO2 at 37 °C.
Determination of NO production and cell viability
The method for determining the amount of NO produced by macrophages has been described in previous studies [8, 9]. Briefly, RAW 264.7 cells (1 × 105 cells/ml) were cultured in a 96-well plate for 2 h. Then, SHL extracts (200 μg/ml) from different harvest time and DEX (10 μM) were added to the cell and stimulated with LPS (1 μg/ml) at 37 °C for 18 h. Finally, the mixtures of the Griess reagent and cell culture supernatants were measured at 550 nm using a Tecan infinite® F200 microplate reader (Tecan Group, Ltd., Männedorf, Switzerland).
The cytotoxicity of SHL extracts was evaluated by the MTT assay, and cells were cultured in a 96-well plate at 1 × 105 cells/mL. Then, different concentrations of SHL extracts (50, 100 and 200 μg/mL) and DEX (10 μM) were added to the cells and treated with LPS at 37 °C for 18 h. After 18 h of incubation, 20 μL of an MTT solution (5 mg/mL) was added to the supernatant, followed by 4 h of incubation. Then, 200 μL of dimethyl sulfoxide was supplemented; then, the supernatant was aspirated and measured at 570 nm using a microplate reader.
Data analysis
Hierarchical clustering analysis (HCA) and principal components analysis (PCA) were performed using the SIMCA 14.1 software (Umetric, Umea, Sweden) to discriminate different SHL samples. For the anti-inflammatory experiment, all data were analyzed by GraphPad Prism 8.0.2 (GraphPad Software, Inc., LaJolla, CA, USA). The values of P < 0.05 and P < 0.01 were considered to be statistically significant and very significant, respectively; this analysis was carried out using a one-way analysis of variance (ANOVA). Dunnett’s test was employed to compare the difference between the SHL extract-treated and LPS-stimulated groups.
Results and discussion
Identification and characterization of chemical constitution in SHL by HPLC–PDA-MS/MS
The rationality and scientificity of collection is an important factor to ensure the quality and uniformity of herbal medicine, which is also essential for clinical use and pharmaceutical production. By exploring the differences in chemical components between different growth seasons and years, we can provide scientific basis for the harvesting period of medicinal materials. To identify the bioactive ingredients in SHL, HPLC–PDA–ESI–MS/MS was performed to obtain the mass and UV spectrum. Finally, fifteen components were identified from SHL samples, which included chlorogenic acid and its isomers neochlorogenic acid, cryptochlorogenic acid, YJ-002 (flavonoid), and eleven triterpenoid saponins. The chemical structures of these compounds are shown in Fig. 1; the detailed information on identified compounds is summarized in Table 2 and Additional file 1: Figures S1–S12.
Peaks 1, 2, 3, 6, and 10 were definitively identified to be due to neochlorogenic acid, chlorogenic acid, cryptochlorogenic acid, hederacoside D, and ciwujianoside A1 by comparison with reference standards, respectively. Peak 4 produced [M−H]− and [M+H]+ ions at m/z 547.1 and 549.3, respectively, which indicated that the molecular weight was 548 (Additional file 1: Figure S1). In the MS/MS spectrum, the major fragment at m/z 271.1 ([M−Rha−Ara+H]+) was observed, which indicated the loss of 278 (Ara–Rha) from the parent ion. By searching the known compounds isolated from SHL, peak 4 was tentatively identified as YJ-002 [5].
In our previous study, triterpenoid saponins separated from SHL exhibited three types of aglycones, which included oleanolic acid, hederagenin, and akebonic acid [10]. Peak 5 exhibited pseudomolecular ions [M−H]− and [M+NH4]+ at m/z 1057.5 and 1076.6 in negative and positive modes, respectively (Additional file 1: Figure S2). The fragmentation of parent ions for peak 5 primarily yielded product ions by the loss of sugar chains (Glc–Glc, Rha, Ara). Moreover, in the MS and MS/MS spectra in positive mode, aglycone ions were observed at m/z 439.4 and 457.2, which corresponded to oleanolic acid. According to our previous experimental data and previous literature, peak 5 was tentatively identified as anhuienside C [11, 12]. Peaks 13,14, and 15 are attributed to the same aglycone ions as that of peak 5; they were tentatively assigned as araliasaponin II, hexaphyloside A, and ciwujianoside C3, respectively, by comparison with known compounds isolated from S. hexaphylla and their families [13,14,15,16,17]. The MS spectrum of Peak 7 shows an identical precursor ion at m/z 1222.6 ([M+NH4]+) and 1249.4 [M+HCOO−]− in positive and negative modes, respectively (Additional file 1: Figure S4). The fragmentation of parent ion produced daughter ions at m/z 928.5, 752.5, and 603.2 attributed to the loss of Ara–Glc, Rha, and Glc. The produced identical aglycone ions were observed at m/z 423.4, which corresponded to akebonic acid. By comparing the MS and MS/MS spectra with a reference standard and previous literature, peak 7 was characterized as ciwujianoside A2 [18, 19]. The same aglycone ions were also observed in the MS spectrum of peaks 9 and 11. By analyzing and comparing previous studies and data, the compounds were finally identified as mutongsaponin E and yemuoside YM12 [19,20,21,22]. Similarly, according to fragment ions and our previous experimental data, peaks 8 and 12 were tentatively identified as akebia saponin D and cussonoside A [10, 23,24,25].
Changes in saponins and flavonoid in SHL from different growth years and seasons
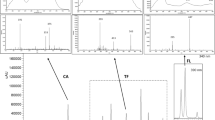
Figure 2 clearly shows that the chemical composition and relative content in SHL from different years and seasons are considerably different. First, peak 4 (YJ-002) is a very representative flavonoid identified from SHL, which can promote bone or cartilage formation by inhibiting osteoclast differentiation and promoting the expression and production of type II collagen and aggrecan [5]. The content of YJ-002 is the highest in one-year-old SHL and then decreased from the third year. Moreover, peak 5 (anhuienside C), peak 7 (ciwujianoside A2), peak 9 (mutongsaponin E), and peak 11 (yemuoside YM12) exhibit the same tendency as YJ-002. Among them, the content of mutongsaponin E decreased the most, and it was difficult to detect yemuoside YM12 in five-year-old SHL.
Representative HPLC–ELSD chromatograms of SHL harvested at different growth years and seasons. a One-year-old SHL harvested in autumn; b three-years-old SHL harvested in autumn; c five-years-old SHL harvested in autumn; d three-years-old SHL harvested in spring. (Identified compounds 1–15 are shown in Table 2.)
However, with an increase in the growth year, the content of some components considerably increased [e.g., peak 6 (hederacoside D), peak 10 (ciwujianoside A1), and peak 15 (ciwujianoside C3)] but there were also some slight differences between them. The content of hederacoside D and ciwujianoside A1 sharply increased from the first to third year and stabilized after the third year. Although the amount of ciwujianoside C3 increased each year, its growth trend was relatively flat compared to hederacoside D and ciwujianoside A1. Second, some specific triterpenoid saponins can be only detected in SHL of three and five-years-old plants such as akebia saponin D, cussonoside A, and araliasaponin II. Of note, these specific triterpenoid saponins exhibit various biological activities. For example, akebia saponin D can suppress both Alzheimer’s disease-related neuro inflammation and memory system dysfunction [26, 27]; cussonoside A exhibits a pronounced sedative effect [25]; araliasaponin II has strong anti-inflammatory activity [17]. This means that if SHL was chosen to treat Alzheimer’s disease, inflammation, insomnia, and other diseases, SHL that was older than three years could be used. Conversely, triterpenoid saponins (e.g., anhuienside C, ciwujianoside A2, mutongsaponin E, and yemuoside YM12) only existed in or their content was relatively high in one-year-old SHL; most of these compounds were isolated from the family of Lardizabalaceae. Previous studies have indicated that these compounds can be used to treat diabetes, prostate diseases, and obesity [28, 29]. This indicates that one-year-old SHL can be used to produce functional foods for weight reduction and diabetes resistance.
To consider the influence of seasonal factors, the chemical constituents of SHL in spring and autumn are quite different (Fig. 2b, d). As can be clearly seen from the chromatograms, only peak 4 (YJ-002), peak 10 (ciwujianoside A1), peak 15 (ciwujianoside C3), and peak 12 (cussonoside A) were detected in SHL harvested in spring, which hinted that SHL was not suitable for harvesting in spring since the content of various components was relatively low or even non-existent. Additionally, these results proved that the accumulation of chemical components is closely related to seasonal factors, which is consistent with previous reports [30, 31]. The sampling site for this study was Goheung, Jeollanam-do province, which is located in the northern region of South Korea. A conceivable explanation may involve the low temperature and rainfall totals that are typical of spring, which may contribute to a mild drought-induced stress in the S. hexaphylla. Furthermore, stress caused by water poverty may decrease the consumed content of NADPH (nicotinamide adenine dinucleotide phosphate) and H+ within the Calvin cycle, reducing plant growth [32]. Excess NADPH and H+ are used in consolidation with carbon fixed during photosynthesis to the formation of secondary metabolites [33].
HCA
HCA is one of the most important systemic statistical methods, which can be used to evaluate the similarities and differences of SHL with different growth years and seasons by their peak characteristics such as relative peak area (RPA) and relative retention time (RRT). In this study, peak 6 was definitively identified to be due to hederacoside D, which is one of the most representative components of SHL with a consistently high content [34, 35]. Therefore, this peak was chosen to calculate the RPA for fifteen common peaks identified from different SHL samples. Thus, the 29 × 15 matrix was obtained, which represented the relative amount of these fifteen components in different SHL extracts. The result of HCA (Fig. 3) clearly revealed that SHL extracts could be classified into two main clusters. Cluster I was formed by S7 to S11, which were three-year-old SHL plants harvested in the spring. Cluster II was further divided into three groups, which were one, three, and five-year-old plants harvested in autumn, respectively. Thus, it is easy to conclude that seasonal factors considerably affect the classification of SHL samples. Additionally, the rescaled distance between group 3 and group 1 is greater than the rescaled distance between group 3 and group 4, which could demonstrate that compared with group 1, the quality of group 3 was more similar to group 4. In other words, the qualities of SHL harvested at 3 years old had a close relationship with those harvested from 5 years old but were quite different from those collected from 1 year old. This result is highly consistent with the visual comparison of their chromatograms (Fig. 2), suggesting their similarities in the content and distribution of chemical constitutions. However, how to more exactly explain the difference between the individual samples in the same cluster was found to be a complication of HCA. From the point of view, the chemical pattern recognition approaches, such as PCA, should be taken into account for further evaluate sample clustering based on their different growing years.
PCA
To identify promising marker components and improve the discrimination capacity to control the quality of SHL, PCA was performed by the RPA of main compounds identified in 29 batches of SHL. The data were computed and input in the same way as for HCA. The first and second principal components were employed to clarify the difference, and they can explain the 93.2% variance in the plot (Fig. 4a). As shown in Fig. 4a, the two-dimensional PCA score plot indicated that 29 batches of SHLs were clearly divided into four domains, which were marked as group 1 (S1–S6), group 2 (S7–S11), group 3 (S12–23), and group 4 (S24–S29). PCA results were highly consistent with HCA. For example, in PCA and HCA plots, S1 to S6 with the same growth year were clustered in the same group. Meanwhile, on the basis of the loading plot of the PCA results (Fig. 4b), peaks 9 and 10 demonstrated better discrimination ability on SHL than other components. At the same time, many in vivo and in vitro studies have proved that these two compounds have anti-osteosarcoma, memory enhancement, antiplatelet aggregation, anti-inflammatory and antinociceptive activities [14, 36,37,38,39]. Therefore, these two bioactive components can be selected as marker compounds to effectively control the quality of SHL with different ages and seasons.
Anti-inflammatory activities
To determine whether different concentrations of SHL extracts affect the viability of RAW 264.7 cells, different growth years and seasons of SHL extracts were added to the cells at the concentration of 50–200 μg/mL. As shown in Fig. 5, there was no significant difference between the treated and control groups (P > 0.05), which revealed that the SHL extract was not cytotoxic to RAW 264.7 cells.
To compare the anti-inflammatory activity of different SHL extracts in vitro, the production of NO was measured by the Griess assay. NO is a typical inflammatory mediator indicator in the formation of inflammation, which is primarily produced by activated macrophages [9]. NO also can regulate the immune function of the body at a normal level, or cause various diseases when overproduced [40, 41]. Figure 6 illustrated that SHL extracts with different growth years and seasons have a considerably different NO content. The SHL harvested at spring did not significantly reduce the NO production in LPS-induced cells relative to the LPS model group (P > 0.05), which demonstrated that the SHL collected from spring was not suitable for treating inflammation-related diseases. Interestingly, the elevated NO content induced by LPS has decreased after treated with different harvested years of SHL at a concentration of 200 μg/mL, and three and five-year-old SHLs exhibited a very significant (P < 0.01) suppressive effect on the content of NO. Besides, there was no significant difference (P > 0.05) between three and 5 years old of SHL and positive control DEX group on the reduction of the NO content, which was suggesting that three-year-old SHLs harvested in autumn could also play the same anti-inflammatory role as the chemical drug of DEX in inhibition of the NO production. Therefore, three-year-old SHLs harvested in autumn was a promising agent that could be used for treating inflammatory diseases.
Effect of the S. hexaphylla leaves extract (200 μg/mL) from different growth years and seasons on the production of nitric oxide. The values of *P < 0.05 and **P < 0.01 were compared with the LPS stimulated group followed by ANOVA. DEX was selected as the positive control group at the concentration of 10 μM
Availability of data and materials
All data generated or analysed during this study are included in this published article and its supplementary information files
Abbreviations
- SHL:
-
Stauntonia hexaphylla leaf
- HPLC–PDA–ESI/MS:
-
High-performance liquid chromatography coupled to photodiode-array and electrospray ionization mass spectrometry detectors
- ELSD:
-
Evaporative light scattering detection
- HCA:
-
Hierarchical clustering analysis
- PCA:
-
Principal component analysis
- LPS:
-
Escherichia coli lipopolysaccharide
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- ESI:
-
Electrospray ionization
- ANOVA:
-
One-way analysis of variance
- NADPH:
-
nicotinamide adenine dinucleotide phosphate
- RPA:
-
Relative peak area
- RRT:
-
Relative retention time
References
Hwang SH, Kwon SH, Kim SB (2017) Lim SS (2017) Inhibitory activities of Stauntonia hexaphylla leaf constituents on rat lens aldose reductase and formation of advanced glycation end products and antioxidant. Biomed Res Int. 2017:4273257
Vinh LB, Jang H-J, Phong NV, Cho K, Park SS, Kang JS (2019) Isolation, structural elucidation, and insights into the anti-inflammatory effects of triterpene saponins from the leaves of Stauntonia hexaphylla. Bioorg Med Chem Lett 29(8):965–969
Choi W-G, Kim J-H, Kim D, Lee Y, Yoo J, Shin D (2018) Simultaneous determination of chlorogenic acid isomers and metabolites in rat plasma using LC–MS/MS and its application to a pharmacokinetic study following oral administration of Stauntonia hexaphylla leaf extract (YRA-1909) to rats. Pharmaceutics. 10(3):143
Cheon Y-H, Baek JM, Park S-H, Ahn S-J, Lee MS, Oh J (2015) Stauntonia hexaphylla (Lardizabalaceae) leaf methanol extract inhibits osteoclastogenesis and bone resorption activity via proteasome-mediated degradation of c-Fos protein and suppression of NFATc1 expression. BMC Complement Altern Med. 15(1):280
Zhang K, Lei J, He Y, Yang X, Zhang Z, Hao D (2018) A flavonoids compound inhibits osteoclast differentiation by attenuating RANKL induced NFATc-1/c-Fos induction. Int Immunopharmacol 61:150–155
Vo HT, Ghimeray AK, Vu NT, Jeong Y-H (2015) Quantitative estimation of ginsenosides in different ages of Panax vietnamensis and their anti-proliferation effects in hela cells. Afr J Tradit Complement Altern Med 12(4):79–83
Cui S, Wang J, Yang L, Wu J, Wang X (2015) Qualitative and quantitative analysis on aroma characteristics of ginseng at different ages using E-nose and GC–MS combined with chemometrics. J Pharmaceut Biomed. 102:64–77
Tian C, Chang Y, Liu X, Zhang Z, Guo Y, Lan Z (2020) Anti-inflammatory activity in vitro, extractive process and HPLC-MS characterization of total saponins extract from Tribulus terrestris L. fruits. Ind Crop Prod. 150:112343
Zhen J, Villani TS, Guo Y, Qi Y, Chin K, Pan M-H (2016) Phytochemistry, antioxidant capacity, total phenolic content and anti-inflammatory activity of Hibiscus sabdariffa leaves. Food Chem 190:673–680
Vinh LB, Jo SJ, Nguyen Viet P, Gao D, Cho KW, Koh E-J (2019) The chemical constituents of ethanolic extract from Stauntonia hexaphylla leaves and their anti-inflammatory effects. Nat Prod Res 15:1–4
Xia Y-G, Gong F-Q, Guo X-D, Song Y, Li C-X, Liang J (2016) Rapid screening and characterization of triterpene saponins in Acanthopanax senticosus leaves via untargeted MSAll and SWATH techniques on a quadrupole time of flight mass spectrometry. J Pharmaceut Biomed. 170:68–82
Li T, Ferns K, Yan Z-Q, Yin S-Y, Kou J-J, Li D (2016) Acanthopanax senticosus: photochemistry and anticancer potential. Am J Chinese Med. 44(08):1543–1558
Vinh LB, Jang H-J, Phong NV, Dan G, Cho KW, Kim YH (2019) Bioactive triterpene glycosides from the fruit of Stauntonia hexaphylla and insights into the molecular mechanism of its inflammatory effects. Bioorg Med Chem Lett 29(16):2085–2089
Yamauchi Y, Ge Y-W, Yoshimatsu K, Komastu K, Kuboyama T, Yang X (2019) Memory enhancement by oral Administration of extract of Eleutherococcus senticosus leaves and active compounds transferred in the brain. Nutrients. 11(5):1142
Jiang W-L, Chen X-G, Zhu H-B, Hou J, Tian J-W (2009) Cornuside attenuates apoptosis and ameliorates mitochondrial energy metabolism in rat cortical neurons. Pharmacology 84(3):162–170
Miyase T, Shiokawa K-I, Zhang DM, Ueno A (1996) Araliasaponins I-XI, triterpene saponins from the roots of Aralia decaisneana. Phytochemistry 41(5):1411–1418
Seo YS, Lee SJ, Li Z, Kang OH, Kong R, Kim SA (2017) Araliasaponin II isolated from leaves of Acanthopanax henryi (Oliv.) Harms inhibits inflammation by modulating the expression of inflammatory markers in murine macrophages. Mol Med Rep 16(1):857–864
Shao C-J, Kasai R, Xu J-D, Tanaka O (1988) Saponins from leaves of Acanthopanax senticosus Harms., Ciwujia: structures of Ciwujianosides B, C1, C2, C3, C4, D1, D2 and E. Chem Pharm Bull 36(2):601–608
Gao H, Wang Z (2006) Triterpenoid saponins and phenylethanoid glycosides from stem of Akebia trifoliata var. australis. Phytochemistry 67(24):2697–2705
Ling Y, Zhang Q, Zhu D-d, Chen F, Kong X-h, Liao L (2015) Identification and characterization of the major chemical constituents in Fructus Akebiae by High-Performance liquid chromatography coupled with electrospray ionization-quadrupole-time-of-flight mass spectrometry. J Chromatogr Sci 54(2):148–157
Wang H-B, Yu D-Q, Liang X-T, Watanabe N, Tamai M, Omura S (1990) Structures of two nortriterpenoid saponins from Stauntonia chinensis. J Nat Prod 53(2):313–318
Sun S, Yan Y-Z, Xie P-S, Lam W (2013) Analysis of bioactive fraction of Caulis Stauntoniae by HPLC fingerpringing. J Liq Chromatogr R T. 36(13):1822–1834
Dubois M, Ilyas M, Wagner H (1986) Cussonosides A and B, two triterpene-saponins from Cussonia barteri. Planta Med 52(02):80–83
Zhou YQ, Yang ZL, Xu L, Li P, Hu YZ (2009) Akebia saponin D, a saponin component from Dipsacus asper Wall, protects PC 12 cells against amyloid-β induced cytotoxicity. Cell Biol Int 33(10):1102–1110
Wu Y, Ji D, Liu Y, Zhang C, Yang Z (2012) Industrial-scale preparation of akebia saponin D by a two-step macroporous resin column separation. Molecules 17(7):7798–7809
Yu X, Wang L-n, Ma L, You R, Cui R, Ji D (2012) Akebia saponin D attenuates ibotenic acid-induced cognitive deficits and pro-apoptotic response in rats: involvement of MAPK signal pathway. Pharmacol Biochem Be. 101(3):479–486
Yu X, Wang L-N, Du Q-M, Ma L, Chen L, You R (2012) Akebia saponin D attenuates amyloid β-induced cognitive deficits and inflammatory response in rats: involvement of Akt/NF-κB pathway. Behav Brain Res 235(2):200–209
Li F, Li W, Fu H, Zhang Q, Koike K (2007) Pancreatic lipase-inhibiting triterpenoid saponins from fruits of Acanthopanax senticosus. Chem Pharm Bull 55(7):1087–1089
Hu X, Wang S, Xu J, Wang D-B, Chen Y, Yang G-Z (2014) Triterpenoid saponins from Stauntonia chinensis ameliorate insulin resistance via the AMP-activated protein kinase and IR/IRS-1/PI3K/Akt pathways in insulin-resistant HepG2 cells. Int J Mol Sci 15(6):10446–10458
Zhang T, Han M, Yang L, Han Z, Cheng L, Sun Z (2019) The effects of environmental factors on ginsenoside biosynthetic enzyme gene expression and saponin abundance. Molecules 24(1):14
Ncube B, Finnie J, Van Staden J (2011) Seasonal variation in antimicrobial and phytochemical properties of frequently used medicinal bulbous plants from South Africa. S Afr J Bot 77(2):387–396
Selmar D, Kleinwächter M (2013) Influencing the product quality by deliberately applying drought stress during the cultivation of medicinal plants. Ind Crop Prod. 42:558–566
Hauf W, Schlebusch M, Huege J, Kopka J, Hagemann M, Forchhammer K (2013) Metabolic changes in synechocystis PCC6803 upon nitrogen-starvation: excess NADPH sustains polyhydroxybutyrate accumulation. Metabolites. 3(1):101–118
Hong G-L, Park S-R, Jung D-Y, Karunasagara S, Lee KP, Koh E-J (2020) The therapeutic effects of Stauntonia hexaphylla in benign prostate hyperplasia are mediated by the regulation of androgen receptors and 5α-reductase type 2. J Ethnopharmacol 250:112446
Jung JY, Karunasagara SSL, Hong GL, Koh EJ, Cho KW, Park SS (2020) Assortment of Stauntonia hexaphylla and Cornus officinalis protect against testosterone-induced benign prostatic hyperplasia through anti-inflammatory and anti-proliferative activity. BioRxiv 15(8):e0236879
Yang C, An Q, Xiong Z, Song Y, Yu K, Li F (2009) Triterpenes from Acanthopanax sessiliflorus fruits and their antiplatelet aggregation activities. Planta Med 75(06):656–659
Xu Q-L, Wang J, Dong L-M, Zhang Q, Luo B, Jia Y-X (2016) Two new pentacyclic triterpene saponins from the leaves of Akebia trifoliata. Molecules 21(7):962
Jung H-J, Park H-J, Kim R-G, Shin K-M, Ha J, Choi J-W (2003) In vivo anti-inflammatory and antinociceptive effects of liriodendrin isolated from the stem bark of Acanthopanax senticosus. Planta Med 69(07):610–616
Hwang JT, Park K-S, Ryuk JA, Kim HJ, Ko BS (2019) Development of an oriental medicine discrimination method through analysis of steroidal saponins in Dioscorea nipponica Makino and their anti-osteosarcoma effects. Molecules 24(22):4022
Limmongkon A, Nopprang P, Chaikeandee P, Somboon T, Wongshaya P, Pilaisangsuree V (2018) LC-MS/MS profiles and interrelationships between the anti-inflammatory activity, total phenolic content and antioxidant potential of Kalasin 2 cultivar peanut sprout crude extract. Food Chem 239:569–578
Jeong H-Y, Choi Y-S, Lee J-K, Lee B-J, Kim W-K, Kang H (2017) Anti-inflammatory activity of citric acid-treated wheat germ extract in lipopolysaccharide-stimulated macrophages. Nutrients. 9(7):730
Acknowledgements
The authors are grateful to Professor Inkyu Hwang and Ye Liu from Laboratory for Immunology and Immunotherapeutics (College of Pharmacy, Chungnam National University) for providing us anti-inflammatory experiments date of different samples of Stauntonia hexaphylla leaves. The authors thank Institute of Drug Research and Development in College of Pharmacy, Chungnam National University.
Funding
The research was supported by the research fund of Chungnam National University.
Author information
Authors and Affiliations
Contributions
DG and JSK conceived and design the experiments. LBV and YHK edited the draft manuscript and provided us the standard compounds. KWC provided us the different samples and analyzed the experimental data. DG and CWC performed the HPLC analysis and wrote the manuscript. All authors helped prepare the manuscript. All authors read and approved the final manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Additional file 1.
The MS1 and MS2 spectra of identified compounds 4 to 15.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, D., Vinh, L.B., Cho, C.W. et al. Discrimination and quality evaluation of fifteen components in Stauntonia hexaphylla leaves from different harvest time by HPLC–PDA–ESI–MS/MS and ELSD coupled with multivariate statistical analysis and anti-inflammatory activity evaluation. Appl Biol Chem 63, 60 (2020). https://doi.org/10.1186/s13765-020-00540-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13765-020-00540-y