Abstract
Background
Recent studies have shown that mesenchymal stem cell (MSC) therapy has potential therapeutic effects for patients with end-stage liver diseases. However, a consensus on the efficacy and safety of MSCs has not been reached.
Methods
A systemic literature review was conducted by searching the Cochrane Library and PubMed databases for articles that evaluated the impact of MSC therapy on the outcomes among patients with end-stage liver disease. Various parameters, including pre- and post-treatment model of end-stage liver disease (MELD) score, serum albumin (ALB), total bilirubin (TB), coagulation function, aminotransferase, and survival rate, were evaluated.
Results
This meta-analysis included a final total of 13 studies and 854 patients. The results indicated improved liver parameters following MSC therapy at different time points, including in terms of MELD score, TB level, and ALB level, compared with conventional treatment. Furthermore, the MSC treatment increased the overall survival rate among patients with liver cirrhosis and acute-on-chronic liver failure (ACLF). The changes in transaminase level and coagulation function differed between the different therapies at various post-treatment time points, indicating that MSC therapy provided no significant benefits in this regard. The further subgroup analysis stratified by liver background revealed that patients with ACLF benefit more from MSC therapy at most time points with improved liver function, including in terms of MELD score, TB level, and ALB level. In addition, no serious side effects or adverse events were reported following MSC therapy.
Conclusions
The meta-analysis results suggest that MSC therapy is safe and results in improved liver function and survival rates among patients with end-stage liver disease. The subgroup analysis stratified by liver background indicated that patients with ACLF benefit more from MSC therapy than patients with liver cirrhosis at most time points.
Similar content being viewed by others
Introduction
Mesenchymal stem cells (MSCs), as one of the multipotent cells, have the potential to self-renew and differentiate into multiple types of cells, such as epithelial cells or hepatocytes [1, 2]. Studies in animal models have shown that MSC therapy can improve liver function, ameliorate liver fibrosis and reverse acute hepatic failure [3,4,5]. Therefore, MSCs are believed to repair damaged hepatocytes and livers, providing therapeutic approaches for end-stage liver disease.
In the clinical setting, autologous and allogeneic MSC infusion is most often instituted in the treatment of liver cirrhosis and liver failure [6]. Compared with autologous MSC treatment, allogenic MSCs overcome the problems of long preparation and delays [7]. In addition, improved cell differentiation, proliferation, and cytokine secretion are provided by allogeneic MSCs derived from healthy donors [8]. In recent decades, studies have shown that MSC treatment can significantly improve liver function and ameliorate liver fibrosis in patients with decompensated liver cirrhosis [9]. Survival rates for patients with acute-on-chronic liver failure (ACLF) are also improved without increased side effects in the long-term [10].
Although a number of studies have been performed to evaluate the benefits of MSC treatment in end-stage liver disease, its clinical efficacy and safety remain unclear. Few meta-analyses of MSC therapy assessed treatment based on controlled trials or consistent evaluation variables. Additionally, detailed analyses of different end-stage liver diseases, including decompensated liver cirrhosis and acute-on-chronic liver failure, were not performed. Therefore, we conducted a meta-analysis of available comparative research to assess the clinical value and safety of MSCs in decompensated liver cirrhosis and ACLF.
Materials and methods
Literature search
Two independent investigators searched the PubMed and Cochrane Library databases (April 2022) to retrieve relevant studies. Comparative trials evaluating the therapeutic value and safety of MSCs versus a control in the treatment of decompensated liver cirrhosis and ACLF were included. No restrictions were set for language, publication date, or publication status. The search strategy was based on the following keywords: “mesenchymal stem cells” and “liver cirrhosis” or “decompensated liver cirrhosis” or “liver failure” or “acute-on-chronic liver failure” and “clinical study” or “clinical trial” or “randomized controlled trial” or “randomized clinical trial”.
Inclusion and exclusion criteria
The main inclusion criteria were comparative studies evaluating outcomes between MSC therapy versus a control in the treatment of decompensated liver cirrhosis and ACLF. The exclusion criteria were as follows: (1) non-comparative studies, case reports, letters, reviews, editorials; (2) studies that lacked clinical data or outcomes; (3) if multiple studies were reported by the same institution, only the highest quality study was included.
Data extraction
Two independent investigators reviewed texts, figures, and tables to extract information from the included studies. The following data were collected: (1) first author name, year of publication, country and study type; (2) study sample size; (3) cell type, cell dosage of MSCs, and time of treatment; (4) study outcomes including albumin (ALB), total bilirubin (TB), model for end-stage liver disease (MELD) score, coagulation function, liver transaminase level, adverse events, and survival rates.
Quality assessment
The Newcastle–Ottawa scale (NOS) was used to assess the quality of included studies [11].
Statistical analysis
The dichotomous variables were evaluated using odds ratios (ORs) with a 95% confidence interval (CI). In the survival analysis, the OR indicated the relative likelihood of death in each group. Continuous variables were assessed by weighted mean differences (WMD). Higgins I2 statistic was used to assess statistical heterogeneity among studies. When I2 < 50%, a fixed-effects model was used for calculations. On the other hand, when heterogeneity was greater than 50%, a random-effects model was recommended. Funnel plots, Egger’s and Begg’s tests were used to identify publication bias. This meta-analysis was performed using Review Manager version 5.3 (Revman, The Cochrane Collaboration, Oxford, UK). P < 0.05 was considered statistically significant.
Results
Study selection and eligibility
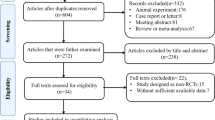
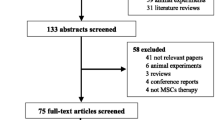
The search strategy initially generated 47 studies, as shown in the flowchart (Fig. 1). Twenty-nine of these studies were excluded because of lack of relevance, duplication, or review article type. Another five studies did not meet the inclusion criteria due to insufficient data or improper study type. Finally, a total of 13 studies were enrolled in our study [12,13,14,15,16,17,18,19,20,21,22,23,24].
A total of 854 patients from five countries were included in the meta-analysis. The patient demographics and baseline characteristics of the included studies are given in Table 1. The studies included patients with end-stage liver diseases including decompensated liver cirrhosis (n = 7) and ACLF (n = 6), of which eight were conducted in China, two in Egypt, one in Korea, one in Brazil, and one in Iran. Four hundred and three patients received MSC therapy, and 451 patients in the control group underwent supportive therapy. MSCs were divided into two types: originating from the umbilical cord (UC-MSCs, n = 5) and from the bone marrow (BM-MSCs, n = 8).
Quality assessment
Newcastle–Ottawa scale (NOS) was used to assess the quality of included studies. Four studies had NOS scores of nine, indicating high quality. The other nine studies were considered to be of moderate quality.
Meta-analysis results
Survival rate
Survival rates of patients at 4 weeks, 8 weeks, 12 weeks, and 24 weeks were analyzed (Fig. 2). Patients receiving MSC therapy had a higher chance of survival at 8 (OR 2.47, 95% CI 1.38–4.43, P = 0.002) and 12 weeks (OR 2.21, 95% CI 1.31–3.74, P = 0.003) compared with controls. While MSC therapy did not show a significant survival benefit at 4 weeks (OR 4.12, 95% CI 0.52–32.53 P = 0.18) or 24 weeks (OR 1.71, 95% CI 0.92–3.19, P = 0.09), it tended to increase survival rate at these points. Subgroup analysis of survival at 8 weeks and 12 weeks by different liver disease backgrounds was performed. All included patients were diagnosed as ACLF before treatment; thus, MSC therapy was associated with increased survival rate at 8 weeks and 12 weeks in the ACLF group.
Meta-analysis of the comparison between mesenchymal stem cell (MSC) therapy and conventional treatment in terms of survival rate. B and C The patients receiving MSC therapy had a higher chance of survival at 8 and 12 weeks compared with the controls. A and D The MSC therapy did not have a significant survival benefit at 4 or 24 weeks
MELD score
The MELD score was calculated according to a formula using three laboratory test results for bilirubin, prothrombin time, and creatinine. Nine studies included an analysis of the MELD score to enable a rapid evaluation of the urgent need of a liver transplantation among the candidates (Fig. 3). Baseline MELD score was not different between the MSC and control groups (MD -0.04, 95% CI -0.63–0.54, p = 0.88). The MELD score decreased significantly at 4 weeks (MD -2.35, 95% CI -3.41- -1.29, P < 0.0001), 12 weeks (MD -3.41, 95% CI -5.41- -1.40, P = 0.0009) and 24 weeks (MD -2.55, 95%CI -3.32- -1.77, P < 0.0001) through MSC therapy. No significant difference was found between the two therapies at 48 weeks. Further subgroup analysis showed that patients with ACLF had a significantly decreased MELD score by MSC therapy at 4 weeks, 12 weeks (MD -4.09, 95% CI -6.26- -1.92, P = 0.0002), and 24 weeks (MD -4.12, 95% CI -6.21- -2.02, P = 0.0001). For patients with cirrhosis, a decreased MELD score at 24 weeks was observed after MSC therapy (MD -2.30, 95% CI -3.13- -1.47, P < 0.00001) (Fig. 4).
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of model of end-stage liver disease (MELD) score. A There was no difference in baseline MELD score between the MSC group and the control group. C, D and E The MELD score decreased significantly at 4, 12, and 24 weeks following MSC therapy. B and F No significant difference was found between the two therapy types at 2 and 48 weeks
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of MELD score, as stratified by different liver diseases. A and B The patients with acute-on-chronic liver failure (ACLF) had a significantly decreased MELD score following MSC therapy at 12 and 24 weeks. B A decreased MELD score at 24 weeks was observed among the patients with cirrhosis following MSC therapy
ALB level
All thirteen studies were enrolled in the analysis of ALB level (Fig. 5). Little difference was seen between the MSC and control groups at baseline (MD 0.71, 95% CI 0.14–1.28, P = 0.02). Compared with controls, ALB was significantly elevated in those who received MSC therapy at 4 weeks (MD 2.08, 95% CI 1.53–2.63, P < 0.00001), 12 weeks (MD 2.05, 95% CI 0.43–3.66, P = 0.01), and 24 weeks (MD 4.03, 95% CI 3.26–4.81, P < 0.00001). We did further subgroup analysis to explore whether liver disease background influenced ALB level after treatment, which showed that MSC therapy was related to an increased level of ALB at 4 weeks (MD 1.88, 95% CI 1.31–2.46, P < 0.00001) and 24 weeks (MD 4.55, 95% CI 3.20–5.91, P < 0.0001) in both the ACLF and cirrhosis subgroups (Fig. 6).
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of albumin (ALB) level. A, B and F There was no significant difference between the MSC group and the control group at baseline and at 2 and 48 weeks. C, D and E Compared with the controls, the ALB level was significantly elevated at 4, 12 and 24 weeks among the patients who received MSC therapy
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of ALB level, as stratified by different liver diseases. A and C The MSC therapy was related to an increased level of ALB at 4 and 24 weeks in both the ACLF subgroup and the cirrhosis subgroup. B and D No benefits of MSC therapy were observed at all other time points
TB level
We included eleven studies in the analysis of TB level (Fig. 7). TB level before treatment was not significantly different between the two groups (MD -1.61, 95% CI -11.14–7.92, P = 0.74). TB level decreased significantly at 2 weeks (MD -16.93, 95%CI -29.64- -4.21, p = 0.009) and at 12 weeks (MD -10.79, 95%CI -21.34- -0.25, p = 0.04) after MSC therapy. No significant changes were found after two treatments at 4 weeks and at 24 weeks. Further subgroup analysis showed that MSC therapy led to a reduction in TB level at 12 weeks in patients with ACLF (MD -15.13, 95% CI -26.94- -3.33, P = 0.01), and at 4 weeks in patients with cirrhosis (MD -24.10, 95% CI -43.11- -5.09, P = 0.01) (Fig. 8).
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of total bilirubin (TB) level. A The TB level before treatment was not significantly different between the two groups. B and D The TB level decreased significantly at 2 and 12 weeks following MSC therapy. C and E No significant changes were observed following the two treatments at 4 and 24 weeks
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of total bilirubin (TB) level, as stratified by different liver diseases. A The MSC therapy led to a reduction in TB level at 4 weeks among the patients with cirrhosis and B at 12 weeks among the patients with ACLF. C No benefit of MSC therapy in terms of TB level was observed at 24 weeks
Coagulation function
Four studies and six studies reported changes in prothrombin time (PT) and international normalized ratio (INR) at different time points, respectively (Figs. 9 and 10). Baseline PT level was not different between the two groups (MD -2.17, 95% CI -2.17–0.64, P = 0.29). Compared with control groups, MSC treatment decreased participants’ PT level significantly at 4 weeks (MD -2.69, 95% CI -4.19–1.19, P = 0.0004). However, PT level increased at 12 weeks after MSCs treatment (MD 6.40, 95% CI 3.21–9.58, P < 0.0001) and did not differ between the groups at 24 weeks (MD 2.51, 95% CI -14.50–19.52, P = 0.77). For INR level, no significant changes were found before and after both treatments at other time points. Due to limited included studies, subgroup analysis was not performed.
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of prothrombin activity (PT). A There was no difference in baseline PT level between the two groups. B Compared with the control groups, the MSC treatment significantly decreased the participants’ PT level at 4 weeks. C The PT level increased at 12 weeks following MSC treatment and D did not differ between the groups at 24 weeks
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of international normalized ratio (INR). No significant changes were observed before and after both treatments at all time points. A There was no difference in baseline INR level between the two groups. B, C, D and E The INR level did not differ between the groups at 2, 4, 12 and 24 weeks
Transaminase level (ALT and AST)
An analysis of ALT and AST levels was reported by eight and five studies, respectively (Figs. 11 and 12). There was no significant difference in baseline transaminase level between groups. Through MSC treatment, the ALT level at 2 weeks (MD − 12.53, 95% CI − 20.56 to − 4.5, P = 0.002) decreased significantly compared to control group. However, ALT level after MSC treatment at 4, 12, and 24 weeks did not show significant changes. As for AST level, MSC therapy significantly decreased the AST level at 4 weeks (MD − 10.77, 95% CI − 20.50 to − 1.04, P = 0.03) and 12 weeks (MD -25.48, 95%CI -48.92- -2.04, p = 0.03).
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of alanine aminotransferase (ALT) level. A There was no significant difference in baseline ALT level between the two groups. B Following MSC treatment, the ALT level at 2 weeks decreased significantly compared with the control group (B). C, D and E Following MSC treatment, there were no significant changes in ALT level at 4, 12 and 24 weeks
Meta-analysis of the comparison between MSC therapy and conventional treatment in terms of aspartate aminotransferase (AST) level. A There was no significant difference in baseline AST level between the two groups. B and C Following MSC therapy, the AST level was significantly decreased at 4 and 12 weeks
Adverse events and complications
No significant adverse events or complications related to MSC therapy were reported by seven studies. Six studies reported major complications, including encephalopathy, gastrointestinal hemorrhage, rash, and infection. Analysis showed that encephalopathy was significantly reduced after MSC therapy (OR 0.41, 95% CI 0.18–0.91 P = 0.03), while the clinical symptoms of gastrointestinal hemorrhage, rash, and infection did not differ between MSC therapy and control groups. There was no statistically significant difference in long-term risk of development of hepatocellular carcinoma between the two groups (OR 0.58, 95% CI 0.29–1.15 P = 0.12) (Table 2).
Discussion
In recent years, studies have demonstrated that MSC therapy is a safe and effective treatment for chronic liver diseases [25, 26]. This systematic review and meta-analysis aimed to evaluate the safety and efficacy of MSC treatment for decompensated liver cirrhosis and ACLF. Our results showed that MSC therapy might improve liver function, assessed by MELD score, TB, and ALB levels. Additionally, MSC treatment appeared to improve survival in patients. No significant difference in transaminase levels or coagulation function was observed between MSC and conventional treatment.
In the present study, the first controversial issue is the effect of MSC treatment on TB and transaminase [27]. Though MSC treatment could improve liver function compared with the baseline, pooled results suggested that MSC treatment could not markedly improve TB and transaminase at all time points. In 2021, Schacher et al. [18] pointed out that end-stage liver disease that results in persistent liver injury may be the reason for the discrepancy. The different liver diseases and the limited sample sizes may explain why statistically significant differences in ALT levels were not detected between the groups, which was consistent with studies conducted in 2017 and 2021 [15, 16, 18].
Another important point relates to the impact of MSC treatment on the survival of patients. For patients with ACLF, it is important to determine whether patients can survive for the first 3 months, since the mortality of whom can be as high as 65%. In our analysis, all included patients were diagnosed as ACLF before treatment and they had a higher chance of survival at 4, 8 and 12 weeks after treatment with MSC. Besides, there was also a trend toward higher survival rates at 24 weeks treated with MSC. The results showed that MSC treatment could help ACLF patients survive for the first 3 months and even longer. It is noteworthy that long-term survival (beyond 48 weeks) was infrequently reported by the included studies. Recently in 2021, Shi et al. [20] reported a survival benefit in decompensated liver cirrhosis patients with MSC treatment over a 75-month follow-up, indicating that MSC therapy could improve the long-term outcomes in liver cirrhosis. Future long-term studies are required to confirm the survival benefits of MSC treatment in end-stage liver disease.
To further explore whether liver disease background influences the efficacy of MSC therapy, subgroup analysis stratified by decompensated liver cirrhosis and ACLF was performed. Owing to the small number of eligible studies, subgroup analysis was limited to TB, ALB, MELD score, and survival rate. It turns out that patients with ACLF might benefit more from MSC therapy at most time points. The results were in line with several studies and highlighted the regenerative role of MSCs in ACLF in 2021[14, 19, 28]. Previously, liver transplantation was regarded as the only therapeutic alternative in end-stage liver disease, especially ACLF [29]. The present study showed that MSC therapy may provide another potential choice in the treatment of ACLF.
Apart from the efficacy of MSC therapy in end-stage liver disease, different routes of MSC transfusion, which can either be through the peripheral vein or through the hepatic artery, are another concern in clinical practice. Theoretically, hepatic arterial injection is more effective than the peripheral vein route due to less loss of MSCs and the higher homing ability. However, the disadvantages of the invasive procedure and the higher risk of bleeding through hepatic arterial injection have been reported in many studies. Meanwhile, peripheral intravenous infusion is considered an ideal route as it is convenient to perform and the MSCs migrate well into the liver parenchyma and differentiate into hepatocytes in vivo. In our analysis, no significant difference was observed between the two routes (Additional file 1: Table S1–S3). More clinical studies are required to determine both the effectiveness and the convenience of the two different transfusion routes.
In addition, we also compared different cell types of MSCs on efficacy for end-stage liver disease. The results showed that BM-MSCs and UC-MSCs had little difference on improvement of liver function. However, previous studies suggested that UC-MSCs had better efficacy, since UC-MSCs showed low alloreactivity and young cellular age. Therefore, comparison of therapeutic effects between BM-MSCs and UC-MSCs calls for more clinical trials. It is noted that MSCs were originally named to represent a class of cells from human bone marrow and periosteum that could maintain their in vitro capacity to be induced to hepatocytes and tissues. Several reports in the early 2000s have described MSC-into-Hep maturation, which impelled clinical studies to confirm the beneficial effects of MSCs. However, recently, Dr. Caplan recommended to change the name of MSCs to Medicinal Signaling Cells since the assumption that MSCs differentiate into mature and functional hepatocytes has never been totally described or approved [30]. Instead, the paracrine action of the multipotent cells rather than differentiation capacity is believed to lead to regeneration induction. The controversy motivates more experimental and clinical studies to explore the differentiation capacity of these cells in end-stage liver disease.
Our meta-analysis has some limitations. First, only 13 reports were included in our study. Second, subgroup analysis of coagulation function and transaminase levels was missing owing to limited included studies. Also, subgroup analysis stratified by different cell types, times of treatment, and administration routes was not conducted, which may cause selection bias. Moreover, the sample size of most included studies was relatively small and long-term follow-up was lacking. Future multi-center large-scale studies are required to further evaluate the efficacy of MSC treatment. Finally, most of the included studies were performed by countries in Asia. This mainly contributes to high incidence of viral hepatitis and liver cirrhosis in Asian countries. This factor is perhaps another potential source of bias.
Conclusions
Despite the limitations noted above, our results incorporated the data from 854 patients to evaluate the safety and efficacy of MSC therapy in the treatment of end-stage liver disease. The results indicated that MSC therapy improved the liver function at most time points, including in terms of MELD score, TB level, and ALB level, compared with conventional treatment. Furthermore, the MSC treatment increased the overall survival rate among the patients. The further subgroup analysis stratified according to liver background revealed that patients with ACLF benefit more from MSC therapy at most time points, with improved liver function. However, there remain concerns regarding MSC source, administration route, and long-term outcomes. Therefore, future multi-center large-scale studies are required to confirm the efficacy and safety of MSC treatment in decompensated liver cirrhosis and ACLF.
Availability of data and materials
The data are available from the corresponding authors upon reasonable request.
Abbreviations
- MSC:
-
Mesenchymal stem cells
- ACLF:
-
Acute-on-chronic liver failure
- MELD:
-
End-stage liver disease
- ALB:
-
Serum albumin
- TB:
-
Total bilirubin
- OR:
-
Odds ratios
- CI:
-
Confidence interval
- WMD:
-
Weighted mean differences
- PT:
-
Prothrombin time
- INR:
-
International normalized ratio
- ALT:
-
Alanine aminotransferase level
- AST:
-
Aspartate aminotransferase level
References
Fu X, Liu G, Halim A, Ju Y, Luo Q, Song AG. Mesenchymal stem cell migration and tissue repair. Cells. 2019;8(8):784.
Sun H, Shi C, Ye Z, Yao B, Li C, Wang X, et al. The role of mesenchymal stem cells in liver injury. Cell Biol Int. 2022;46(4):501–11.
Jang YO, Cho MY, Yun CO, Baik SK, Park KS, Cha SK, et al. Effect of function-enhanced mesenchymal stem cells infected with decorin-expressing adenovirus on hepatic fibrosis. Stem Cells Transl Med. 2016;5(9):1247–56.
Stock P, Bruckner S, Winkler S, Dollinger MM, Christ B. Human bone marrow mesenchymal stem cell-derived hepatocytes improve the mouse liver after acute acetaminophen intoxication by preventing progress of injury. Int J Mol Sci. 2014;15(4):7004–28.
Zagoura DS, Roubelakis MG, Bitsika V, Trohatou O, Pappa KI, Kapelouzou A, et al. Therapeutic potential of a distinct population of human amniotic fluid mesenchymal stem cells and their secreted molecules in mice with acute hepatic failure. Gut. 2012;61(6):894–906.
Watanabe Y, Tsuchiya A, Terai S. The development of mesenchymal stem cell therapy in the present, and the perspective of cell-free therapy in the future. Clin Mol Hepatol. 2021;27(1):70–80.
Zhang K, Sun H, Cao H, Jia Y, Shu X, Cao H, et al. The impact of recipient age on the effects of umbilical cord mesenchymal stem cells on HBV-related acute-on-chronic liver failure and liver cirrhosis. Stem Cell Res Ther. 2021;12(1):466.
Lim YL, Eom YW, Park SJ, Hong T, Kang SH, Baik SK, et al. Bone marrow-derived mesenchymal stem cells isolated from patients with cirrhosis and healthy volunteers show comparable characteristics. Int J Stem Cells. 2020;13(3):394–403.
Wang L, Li J, Liu H, Li Y, Fu J, Sun Y, et al. Pilot study of umbilical cord-derived mesenchymal stem cell transfusion in patients with primary biliary cirrhosis. J Gastroenterol Hepatol. 2013;28(Suppl 1):85–92.
Jia Y, Shu X, Yang X, Sun H, Cao H, Cao H, et al. Enhanced therapeutic effects of umbilical cord mesenchymal stem cells after prolonged treatment for HBV-related liver failure and liver cirrhosis. Stem Cell Res Ther. 2020;11(1):277.
Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25(9):603–5.
El-Ansary M, Abdel-Aziz I, Mogawer S, Abdel-Hamid S, Hammam O, Teaema S, et al. Phase II trial: undifferentiated versus differentiated autologous mesenchymal stem cells transplantation in Egyptian patients with HCV induced liver cirrhosis. Stem Cell Rev Rep. 2012;8(3):972–81.
Li YH, Xu Y, Wu HM, Yang J, Yang LH, Yue-Meng W. Umbilical cord-derived mesenchymal stem cell transplantation in hepatitis B virus related acute-on-chronic liver failure treated with plasma exchange and entecavir: a 24-month prospective study. Stem Cell Rev Rep. 2016;12(6):645–53.
Peng L, Xie DY, Lin BL, Liu J, Zhu HP, Xie C, et al. Autologous bone marrow mesenchymal stem cell transplantation in liver failure patients caused by hepatitis B: short-term and long-term outcomes. Hepatology. 2011;54(3):820–8.
Lin BL, Chen JF, Qiu WH, Wang KW, Xie DY, Chen XY, et al. Allogeneic bone marrow-derived mesenchymal stromal cells for hepatitis B virus-related acute-on-chronic liver failure: a randomized controlled trial. Hepatology. 2017;66(1):209–19.
Mohamadnejad M, Alimoghaddam K, Bagheri M, Ashrafi M, Abdollahzadeh L, Akhlaghpoor S, et al. Randomized placebo-controlled trial of mesenchymal stem cell transplantation in decompensated cirrhosis. Liver Int. 2013;33(10):1490–6.
Salama H, Zekri AR, Medhat E, Al Alim SA, Ahmed OS, Bahnassy AA, et al. Peripheral vein infusion of autologous mesenchymal stem cells in Egyptian HCV-positive patients with end-stage liver disease. Stem Cell Res Ther. 2014;5(3):70.
Schacher FC, Pezzi Martins, da Silva A, Silla L, Alvares-da-Silva MR. Bone marrow mesenchymal stem cells in acute-on-chronic liver failure grades 2 and 3: a phase I-II randomized clinical trial. Can J Gastroenterol Hepatol. 2021;20:3662776.
Shi M, Zhang Z, Xu R, Lin H, Fu J, Zou Z, et al. Human mesenchymal stem cell transfusion is safe and improves liver function in acute-on-chronic liver failure patients. Stem Cells Transl Med. 2012;1(10):725–31.
Shi M, Li YY, Xu RN, Meng FP, Yu SJ, Fu JL, et al. Mesenchymal stem cell therapy in decompensated liver cirrhosis: a long-term follow-up analysis of the randomized controlled clinical trial. Hepatol Int. 2021;15(6):1431–41.
Suk KT, Yoon JH, Kim MY, Kim CW, Kim JK, Park H, et al. Transplantation with autologous bone marrow-derived mesenchymal stem cells for alcoholic cirrhosis: phase 2 trial. Hepatology. 2016;64(6):2185–97.
Xu L, Gong Y, Wang B, Shi K, Hou Y, Wang L, et al. Randomized trial of autologous bone marrow mesenchymal stem cells transplantation for hepatitis B virus cirrhosis: regulation of Treg/Th17 cells. J Gastroenterol Hepatol. 2014;29(8):1620–8.
Xu WX, He HL, Pan SW, Chen YL, Zhang ML, Zhu S, et al. Combination treatments of plasma exchange and umbilical cord-derived mesenchymal stem cell transplantation for patients with hepatitis B virus-related acute-on-chronic liver failure: a clinical trial in China. Stem Cells Int. 2019;2019:4130757.
Zhang Z, Lin H, Shi M, Xu R, Fu J, Lv J, et al. Human umbilical cord mesenchymal stem cells improve liver function and ascites in decompensated liver cirrhosis patients. J Gastroenterol Hepatol. 2012;27(Suppl 2):112–20.
Forbes SJ, Gupta S, Dhawan A. Cell therapy for liver disease: from liver transplantation to cell factory. J Hepatol. 2015;62(1 Suppl):S157–69.
Zhou GP, Jiang YZ, Sun LY, Zhu ZJ. Therapeutic effect and safety of stem cell therapy for chronic liver disease: a systematic review and meta-analysis of randomized controlled trials. Stem Cell Res Ther. 2020;11(1):419.
Carvalho JR, Verdelho MM. New insights about albumin and liver disease. Ann Hepatol. 2018;17(4):547–60.
Zheng Y, Zhu S, Zheng X, Xu W, Li X, Li J, et al. Serum from acute-on-chronic liver failure patients may affect mesenchymal stem cells transplantation by impairing the immunosuppressive function of cells. J Clin Transl Hepatol. 2021;9(4):503–13.
Belli LS, Duvoux C, Artzner T, Bernal W, Conti S, Cortesi PA, et al. Liver transplantation for patients with acute-on-chronic liver failure (ACLF) in Europe: Results of the ELITA/EF-CLIF collaborative study (ECLIS). J Hepatol. 2021;75(3):610–22.
Caplan AI. Mesenchymal stem cells: time to change the name! Stem Cells Transl Med. 2017;6(6):1445–51.
Acknowledgements
We thank Medjaden Inc. for editing this manuscript.
Funding
The specific research fund of the Innovation Platform for Academicians of Hainan Province (YSPTZX202216). The Fund of National Clinical Center for Infectious Diseases, PLA General Hospital (NCRC-ID202105,413FZT6). The Fund of Science and Technology Project of Wuhan (No: 2020020602012112). The Fund of Haihe Laboratory of Cell Ecosystem Innovation Fund (HH22KYZX0046). The Fund of Tianjin Free Trade Zone Innovation Development Project 211 (ZMCY-03–2021002-01). The funders of the study had no role in the study design, data collection, data analysis, data interpretation, or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
WY and LS designed the methods. HW, WY, and YW drafted the manuscript. HD, TD, and WZ carried out the acquisition, analysis, and interpretation of the data. LC and LZ were responsible for data curation. YZ, LS, and YJ performed the statistical analysis and were responsible for the integrity of the data and accuracy of the data analysis. All authors had full access to all data in the study and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Weiqi Yao, Haibo Dong, Tengyun Dong, Lulu Zhao, and Yu Zhang are employees of Wuhan Optics Valley Vcanbiopharma Co., Ltd. All other authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1
: Table S1 Results of administration route and cell type of MSCs therapy on MELD score. Table S2 Results of administration route and cell type of MSCs therapy on ALB level. Table S3 Results of administration route and cell type of MSCs therapy on TBIL level..
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, H., Yao, W., Wang, Y. et al. Meta-analysis on last ten years of clinical injection of bone marrow-derived and umbilical cord MSC to reverse cirrhosis or rescue patients with acute-on-chronic liver failure. Stem Cell Res Ther 14, 267 (2023). https://doi.org/10.1186/s13287-023-03494-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13287-023-03494-2