Abstract
Introduction
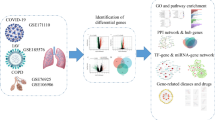
Coronavirus disease 2019 (COVID-19)-associated tracheal stenosis (COATS) may occur as a result of prolonged intubation during COVID-19 infection. We aimed to investigate patterns of gene expression in the tracheal granulation tissue of patients with COATS, leverage gene expression data to identify dysregulated cellular pathways and processes, and discuss potential therapeutic options based on the identified gene expression profiles.
Methods
Adult patients (age ≥ 18 years) presenting to clinics for management of severe, recalcitrant COATS were included in this study. RNA sequencing and differential gene expression analysis was performed with transcriptomic data for normal tracheal tissue being used as a control. The top ten most highly upregulated and downregulated genes were identified. For each of these pathologically dysregulated genes, we identified key cellular pathways and processes they are involved in using Gene Ontology (GO) and KEGG (Kyoto Encyclopedia of Genes and Genomes) applied via Database for Annotation, Visualization, and Integrated Discovery (DAVID).
Results
Two women, aged 36 years and 37 years, were included. The profile of dysregulated genes indicated a cellular response consistent with viral infection (CXCL11, PI15, CCL8, DEFB103A, IFI6, ACOD1, and DEFB4A) and hyperproliferation/hypergranulation (MMP3, CASP14 and HAS1), while downregulated pathways included retinol metabolism (ALDH1A2, RBP1, RBP4, CRABP1 and CRABP2).
Conclusion
Gene expression changes consistent with persistent viral infection and dysregulated retinol metabolism may promote tracheal hypergranulation and hyperproliferation leading to COATS. Given the presence of existing literature highlighting retinoic acid’s ability to favorably regulate these genes, improve cell-cell adhesion, and decrease overall disease severity in COVID-19, future studies must evaluate its utility for adjunctive management of COATS in animal models and clinical settings.
Similar content being viewed by others
Background
Acquired tracheal stenosis may occur in up to 20% of patients after prolonged intubation [1,2,3], inflicting significant limitations on patients’ respiratory function, vocal ability, and overall quality of life [4]. During the coronavirus disease 2019 (COVID-19) pandemic, studies from across the world reported intubation rates ranging from 5 to 88% amongst patients with COVID-19 [5]. Moreover, the median duration of intubation may be as long as 17 days, with more than 18% of patients requiring reintubation within one week of extubation [6, 7]. It is believed that in addition to airway mucosal damage due to intubation, tracheitis due to COVID-19 may also contribute to tracheal stenosis [8]. As a result, the European Laryngological Society urges physicians to maintain a high index of suspicion for tracheal stenosis amongst patients with COVID-19 requiring intubation [9].
The management of tracheal stenosis is mainly interventional, with options including tracheal resection and reconstruction, bronchoscopic dilation, laser therapy, spray cryotherapy, or airway stent placement [10, 11]. Adjunctive medical therapies have been explored with limited success, including local mitomycin C, local or inhaled steroids, oral proton pump inhibitors, trimethoprim-sulfamethoxazole, penicillin, and macrolide antibiotics [12,13,14]. Recently, an increased understanding of molecular and genetic profiles of tracheal granulation tissue is guiding the exploration of novel therapies for tracheal stenosis [15]. However, data is sparse and there is an urgent need to explore genetic mechanisms underlying the development of tracheal stenosis [16], particularly in the context of COVID-19 infection. Thus, we aimed to investigate patterns of gene expression in the tracheal granulation tissue of patients with COVID-19-associated tracheal stenosis (COATS), and leverage gene expression data to identify key dysregulated pathways and processes. We also discuss potential therapeutic options based on the identified gene expression profiles. Although this Research Note presents data for a small sample, we believe the novelty of the results warrant its sharing with the scientific community.
Main text
Methods
This study was conducted between July 2020-July 2021 at Nuvance Health in Connecticut, USA, after receiving ethical approval from the institutional review board (ID: 2019-19).
Patient enrollment and sample collection
We sought to include adult patients (age ≥ 18 years) presenting to clinics for management of severe, recalcitrant COATS, as visualized by upper airway endoscopy. Patients were diagnosed with COATS if they developed tracheal stenosis in the setting of active COVID-19 infection during airway interventions performed for the management of COVID-19. Informed consent was acquired prior to patient enrolment and data collection. Patient data collected included demographics, baseline health status, and clinical history. Samples were collected at the index visit and subsequent visits at time of reintervention (with each reintervention and sample collection being roughly six months apart), making for a total of five tissue samples (two from patient 1 and three from patient 2).
Tracheal tissue biopsies of granulation tissue were collected at the time of endoscopic intervention, which consisted of balloon dilation and spray cryotherapy. If enrolled patients returned to clinics requiring reintervention for re-stenosis, they were reapproached for collection of additional samples. Additional biopsies were collected from consenting patients, with these being considered as unique samples for analysis.
RNA sequencing of samples (transcriptomics)
Tissue samples were sent to Azenta Life Sciences (Burlington, Massachusetts, USA) for processing and RNA sequencing. A next-generation sequencing platform (HiSeq ®; Illumina, Inc., San Diego, California, USA) with Poly(A) selection was used to prepare the cDNA (complementary DNA) libraries.
Normal control
Publicly accessible transcriptomics data for normal tracheal tissue was sourced from the public data repositories of the National Center for Biotechnology Information (NCBI: SRR16760102) and the European Nucleotide Archive (ENA: ERR2022844).
Data analysis
CLC Genomics Workbench by QIAGEN (Venlo, Netherlands) was used for analysis of the sequenced data. Trimmed reads were aligned and annotated with Ensembl 91: Dec 2017 (GRCh38.p10). Differential gene expressions were explored between tissue samples of COATS and normal control data. Gene expression was considered significant if the false discovery rate (FDR) p-value was < 0.05 and the fold change (ratio of value in specimen to value in normal control) was > 1.5. The top 10 most highly upregulated and downregulated genes were identified by calculating a change coefficient that accounted for both FDR p-value and fold change, as follows:
The protein class of the gene products and their relevant functions were retrieved from the Human Protein Atlas [17], an open-source repository containing data on all proteins coded by the human genome. Based on their relevant functions, we determined whether the upregulation or downregulation of each gene was likely part of the body’s protective response to COVID-19-infection (e.g., immune system activation) or contributing towards pathological mechanisms causing tracheal stenosis. In addition to comparing the COATS samples to normal control data, we also compared the COATS samples against each other.
Gene Ontology (GO) and KEGG (Kyoto Encyclopedia of Genes and Genomes) functional enrichment were applied via DAVID (Database for Annotation, Visualization, and Integrated Discovery) for significantly upregulated and downregulated genes separately. Significantly upregulated and downregulated cellular pathways and biological processes (i.e., those with a Benjamini p-value < 0.05), along with their GO identification numbers, were noted.
Results
We included two patients in this study. Patient 1 was a 37-year-old woman with obstructive sleep apnea and hypertension who had developed tracheal stenosis two months after undergoing tracheostomy tube placement during COVID-19 infection. She had undergone three prior spray cryotherapy and balloon dilation procedures for tracheal stenosis prior to presenting to our institution for her index visit with our team. She had been diagnosed with obstructive sleep apnea three years ago and hypertension recently. Patient 2 was a 36-year-old woman with interstitial lung disease and a history of smoking who developed tracheal stenosis a month after prolonged intubation during COVID-19 infection. She was diagnosed with interstitial lung disease a year ago. Patient 2 had not undergone any prior procedures for tracheal stenosis. These details are shown in Supplementary File 1 - Table 1.
The top 10 most highly upregulated and downregulated genes amongst these five tissue samples are shown in Tables 1 and 2, respectively. Significantly upregulated cellular pathways included those related to COVID-19 and other viral infections (influenza A, human papillomavirus, hepatitis C virus, Epstein-Barr virus, and measles virus), cytokine-cytokine receptor interaction, viral protein interaction with cytokine and cytokine receptor, and NOD (nucleotide oligomerization domain)-like receptor signaling pathways (Supplementary File 1 - Table 2). Significantly upregulated biological processes included antiviral defense, innate immunity, inflammatory response, chemotaxis, angiogenesis, and collagen degradation; significantly downregulated biological processes included cell adhesion & ion transport (Supplementary File 1 - Table 3).
Notably, retinol metabolism was also identified as the sole significantly downregulated cellular pathway (Supplementary File 1 - Table 2). Thus, we conducted a deep-dive into the genes directly involved in the metabolism and action of retinoic acid (active form of retinol). Gene expression appeared to indicate a state of compensation for relative retinoic acid deficiency, with CRABP1 (cellular retinoic acid-binding protein 1: functions to inhibit retinoic acid’s activity) being downregulated by a factor of 18.04 and CRABP2 (cellular retinoic acid-binding protein 1: functions to enable retinoic acid’s cellular activity) being upregulated by a factor of 99.96. In addition, RBP1 (retinol-binding protein 1: facilitates enzymatic conversion of retinol to retinoic acid) was upregulated by a factor of 22.02. The local retinoic acid deficiency may arise due to suppressed retinoic acid transport from the liver to the trachea: RBP4 (retinol-binding protein 4: functions to transport retinoic acid from the liver to peripheral tissues) was downregulated by a factor of 26.01. Moreover, enzymatic conversion of retinoid precursors to retinoic acid was also suppressed: RALDH2/ALDH1A2 (retinaldehyde dehydrogenase 2/ aldehyde dehydrogenase 1 family member A2: catalyzes synthesis of retinoic acid from retinaldehyde) was downregulated by a factor of 12.71.
In addition, we also compared the significantly dysregulated genes for Patient 1 vs. Patient 2. Genes that were uniquely upregulated in Patient 2 (vs. Patient 1) were involved in cellular pathways related to carcinogenesis.
The raw transcriptomic data is shown in Supplementary File 2.
Discussion
This is the first study reporting patterns of gene expression in COATS. RNA sequencing analysis indicated an upregulation of genes and pathways involved in a persistent cellular antiviral response, confirming the notion that the pathophysiology of COATS features an infective component. Gene expression indicated cellular processes typical of tracheal stenosis, such as hyperproliferation (due to downregulated CRNN and CAPN14 causing decreased cell-cycle regulation), hypergranulation (due to downregulated SPRR3, RPTN, TGM3, and SLURP1 causing dysregulated keratinization), and extracellular matrix remodeling (upregulated MMPs and HAS1). Several of the upregulated genes indicated a cellular response to viral infection (CXCL11, CCL8, DEFB103A, IFI6, ACOD1, and DEFB4A). CXCL11 and CCL8 are involved in chemotaxis of immune cells, while the defensins (DEFB103A and DEFB4A) are broadly involved in innate immunity at the epithelial surface of the trachea. IFI6 plays an important role in the innate immunity against viruses, while ACOD1 also acts in an anti-viral capacity by suppressing replication of viral genomes. Our results also indicated downregulation of cellular pathways involved in retinol metabolism and suggested a state of relative retinoic acid deficiency. Figure 1 summarizes and synthesizes the proposed interactions between the key cellular processes contributing to COATS.
We performed a literature search to identify potential therapeutic options that could inhibit pathologically upregulated gene products or promote pathologically downregulated gene products (Table 3). Retinoic acid suppresses MMP3 [18] and CASP14 [19] (upregulated in COATS) and promotes SLURP1 [20] and CRNN [21] (downregulated in COATS). MMP3 and CASP14 may also serve as molecular targets for the prevention and management of COATS, as their expression may be suppressed using their respective inhibitors. Thus, retinoic acid, MMP3 inhibitors, and CASP14 inhibitors may hold therapeutic promise and should be prioritized for future investigation.
Retinoic acid is well-known for its role in maintaining epithelial integrity and promoting mucosal immunity [22]. Topical retinoic acid can improve the regeneration of a mucociliary respiratory epithelium after iatrogenic mucosal injury [23]. Retinoic acid has been shown to suppress MMPS [18] and CASP14 [19], while promoting expression of SLURP1 [20] and CRNN [21]. Moreover, retinoic acid has also been shown to promote cell-cell adhesion [24], which was also downregulated in the COATS tissue samples. Lastly, we also observed downregulation of pathways concerned with retinol metabolism. In fact, evidence suggests that retinoic acid depletion may be a common feature of COVID-19 infection [25, 26] and that administration of retinoic acid may reduce overall disease severity by targeting a variety of key molecular players [27,28,29]. Systemic retinoic acid can prevent virus-induced airway hyper-reactivity due to its anti-inflammatory and anti-viral effects [30]. This evidence collectively points towards retinoic acid being a potential adjunct therapy to prevent or treat COATS, and future studies must evaluate its benefits in animal models and clinical settings.
Other potential therapies that may be explored include MMP3 inhibitors and caspase inhibitors. MMP3, like other matrix metalloproteinases, plays a key role in extracellular matrix degradation during tissue remodeling that occurs as part of hyperproliferation and stenosis [31]. In COVID-19 infection, the upregulation of MMP3 has been identified previously as both a biomarker and a potential therapeutic target to prevent systemic complications of the disease [32, 33]. The main role of CASP14 (caspase-14) in the human body is the maintenance of the stratum corneum by promoting epidermal cornification [34]. However, formation of a cornified layer has been shown to occur in tracheal stenosis [35], presumably leading to a severe and recalcitrant manifestation of the disease. Caspases are widely upregulated in severe and chronic forms of COVID-19 infection, and caspase inhibitors have been identified as possible therapeutic options for these conditions [36]. While CASP-14 specific inhibitors have not been explored, pan-caspase inhibitors may be used in COVID-19 [36]. Thus, MMP3 and caspase inhibition also warrant attention with regards to their role in preventing and managing COATS.
In conclusion, we identified three upregulated genes (MMP3, CASP14, and HAS1) and five downregulated genes (SPRR3, CRNN, CAPN14, SLURP1, and RPTN) that likely promote tracheal hypergranulation and hyperproliferation leading to COVID-19-associated tracheal stenosis (COATS). In addition, retinol metabolism pathways were dysregulated, and gene expression indicated a relative local retinoic acid deficiency. Given the presence of existing literature highlighting retinoic acid’s ability to favorably regulate these genes, improve cell-cell adhesion, and decrease overall disease severity in COVID-19, future studies must evaluate its utility for adjunctive management of COATS in animal models and clinical settings.
Limitations
This study has limitations. First this research note presents data from only five samples from two patients. Second, genes upregulated in Patient 2 indicated possible carcinogenesis (may be indicative of early neoplastic processes attributable to the patient’s smoking history), which may confound results. Third, our work only discusses on the biological plausibility of potential therapeutic options, and future work is required to evaluate actual benefits of these drugs. Lastly, we did not evaluate changing trends in the gene expression at different timepoints within the continuum of the disease course.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request. The genomic data for the patients with COATS can be found in Supplementary File 2.
References
Pearson FG, Andrews MJ. Detection and management of tracheal stenosis following cuffed tube tracheostomy. Ann Thorac Surg. 1971;12(4):359–74. Epub 1971/10/01.
Grillo HC, Donahue DM, Mathisen DJ, Wain JC, Wright CD. Postintubation tracheal stenosis. Treatment and results. J Thorac Cardiovasc Surg. 1995;109(3):486–92. discussion 92– 3. Epub 1995/03/01.
Stauffer JL, Olson DE, Petty TL. Complications and consequences of endotracheal intubation and tracheotomy. A prospective study of 150 critically ill adult patients. Am J Med. 1981;70(1):65–76. Epub 1981/01/01.
Bibas BJ, Cardoso PFG, Minamoto H. Pêgo-Fernandes PMJTcr. Quality-of-life evaluation in patients with laryngotracheal diseases. 2020;9(3):2099.
Mohammadi M, Khamseh AKP, Varpaei HAJA, Medicine P. Invasive Airway Intubation in COVID-19 Patients; Statistics, Causes, and Recommendations: A Review Article. 2021;11(3).
Allgood S, Peters J, Benson A, Maragos C, McIltrot K, Slater T et al. Acquired laryngeal and subglottic stenosis following COVID-19—Preparing for the coming deluge. 2021.
Fleuren LM, Dam TA, Tonutti M, de Bruin DP, Lalisang RC, Gommers D et al. Predictors for extubation failure in COVID-19 patients using a machine learning approach. 2021;25(1):1–10.
Tyagi R, Arunachalam M, Mittal S, Hadda V, Mohan A, Guleria R et al. COVID-19-Associated Tracheal Stenosis (COATS): A New Entity? 2022:10.1097.
Piazza C, Filauro M, Dikkers FG, Nouraei SR, Sandu K, Sittel C, et al. Long-term intubation and high rate of tracheostomy in COVID-19 patients might determine an unprecedented increase of airway stenoses: a call to action from the European. Laryngological Soc. 2021;278:1–7.
Herrington HC, Weber SM, Andersen PEJTL. Mod Manage Laryngotracheal Stenosis. 2006;116(9):1553–7.
Bhora FY, Ayub A, Forleiter CM, Huang CY, Alshehri K, Rehmani S, et al. Treatment of Benign Tracheal Stenosis using Endoluminal Spray Cryotherapy. JAMA otolaryngology– head neck Surg. 2016;142(11):1082–7. Epub 2016/08/18.
Hoffman MR, Patro A, Huang LC, Chen SC, Berry LD, Gelbard A et al. Impact of adjuvant medical therapies on surgical outcomes in idiopathic subglottic stenosis. 2021;131(12):E2880–6.
Croft CB, Zub K, Borowiecki B. Therapy of iatrogenic subglottic stenosis: a steroid/antibiotic regimen. Laryngoscope. 1979;89(3):482–9. Epub 1979/03/01.
Zhou L, Li Y, Gan LM, Qin EY, Meng XY, Gan JH, et al. [Effects of different drugs on bronchial stenosis by TGF-β/mTOR signaling pathway in rabbit model]. Zhonghua Yi Xue Za Zhi. 2019;99(24):1898–903. Epub 2019/07/05.
Wei P, Huang Z, Gan L, Li Y, Qin C, Liu GJA. Nintedanib ameliorates tracheal stenosis by activating HDAC2 and suppressing IL-8 and VEGF in rabbit. 2020;12(8):4739.
Farzanegan R, Feizabadi M, Ghorbani F, Movassaghi M, Vaziri E, Zangi M, et al. Overv Tracheal Stenosis Res Trends hot Top. 2017;20(9):–.
Gogna P, Narain TA, O’Sullivan DE, Villeneuve PJ, Demers PA, Hystad P, et al. Estimates of the current and future burden of lung cancer attributable to PM2.5 in Canada. Prev Med. 2019;122:91–9.
Liu Y, Kimura K, Orita T, Teranishi S, Suzuki K, Sonoda KH. All-trans-retinoic acid inhibition of transforming growth factor-β-induced collagen gel contraction mediated by human Tenon fibroblasts: role of matrix metalloproteinases. Br J Ophthalmol. 2015;99(4):561–5. Epub 2015/01/24.
Rendl M, Ban J, Mrass P, Mayer C, Lengauer B, Eckhart L, et al. Caspase-14 expression by epidermal keratinocytes is regulated by retinoids in a differentiation-associated manner. J Invest Dermatol. 2002;119(5):1150–5. Epub 2002/11/26.
Mastrangeli R, Donini S, Kelton CA, He C, Bressan A, Milazzo F, et al. ARS Component B: structural characterization, tissue expression and regulation of the gene and protein (SLURP-1) associated with Mal De Meleda. Eur J Dermatology: EJD. 2003;13(6):560–70. Epub 2004/01/15.
Cheong KA, Kim HJ, Kim JY, Kim CH, Lim WS, Noh M, et al. Retinoic acid and hydroquinone induce inverse expression patterns on cornified envelope-associated proteins: implication in skin irritation. J Dermatol Sci. 2014;76(2):112–9. Epub 2014/09/23.
McCullough FS, Northrop-Clewes CA, Thurnham DI. The effect of vitamin A on epithelial integrity. Proc Nutr Soc. 1999;58(2):289–93. Epub 1999/08/31.
Fang KM, Wang CT, Chen YW, Huang TW. Reduction of adhesions and antrostomy stenosis with topical vitamin A after endoscopic sinus surgery. Am J Rhinol Allergy. 2015;29(6):430–4. Epub 2015/12/08.
Whelan JT, Chen J, Miller J, Morrow RL, Lingo JD, Merrell K, et al. 9-cis-retinoic acid promotes cell adhesion through integrin dependent and independent mechanisms across immune lineages. J Nutr Biochem. 2013;24(5):832–41. Epub 2012/08/29.
Sarohan AR. COVID-19: endogenous retinoic acid theory and retinoic acid depletion syndrome. Med Hypotheses. 2020;144:110250. Epub 2020/12/02.
Sarohan AR, Akelma H, Araç E, Aslan Ö, Cen O. Retinol Depletion in COVID-19. Clin Nutr open Sci. 2022;43:85–94. Epub 2022/06/07.
Stephensen CB, Lietz G. Vitamin A in resistance to and recovery from infection: relevance to SARS-CoV2. Br J Nutr. 2021;126(11):1663–72. Epub 2021/01/21.
Tepasse PR, Vollenberg R, Fobker M, Kabar I, Schmidt H, Meier JA et al. Vitamin A plasma levels in COVID-19 patients: a prospective Multicenter Study and Hypothesis. Nutrients. 2021;13(7). Epub 2021/07/03.
Li R, Wu K, Li Y, Liang X, Tse WKF, Yang L, et al. Revealing the targets and mechanisms of vitamin A in the treatment of COVID-19. Aging. 2020;12(15):15784–96. Epub 2020/08/18.
Moreno-Vinasco L, Verbout NG, Fryer AD, Jacoby DB. Retinoic acid prevents virus-induced airway hyperreactivity and M2 receptor dysfunction via anti-inflammatory and antiviral effects. Am J Physiol Lung Cell Mol Physiol. 2009;297(2):L340–6. Epub 2009/05/26.
Jarmuz T, Roser S, Gal A, Rivera H, Roman J. Transforming growth Factor-β1, myofibroblasts, and tissue remodeling in the pathogenesis of Tracheal Injury: potential role of gastroesophageal reflux. 2004;113(6):488–97.
Almuntashiri S, Zhang D, Somanath PR, Sikora A. MMP3 in severe COVID-19: a biomarker or therapeutic target? Infect Disord Drug Targ. 2023;23(1):e190622206159. Epub 2022/06/22.
Kadry R, Newsome AS, Somanath PR. Pharmacological inhibition of MMP3 as a potential therapeutic option for COVID-19 Associated Acute Respiratory Distress Syndrome. Infect Disord Drug Targ. 2021;21(6):e170721187996. Epub 2020/11/18.
Denecker G, Ovaere P, Vandenabeele P, Declercq W. Caspase-14 reveals its secrets. J Cell Biol. 2008;180(3):451–8. Epub 2008/02/06.
Li XZ, Wang ZC, Qiu Y, Ma SX, Meng LB, Wu WH, et al. Bioinformatics analysis and verification of gene targets for benign tracheal stenosis. Mol Genet Genom Med. 2020;8(6):e1245. Epub 2020/04/21.
Plassmeyer M, Alpan O, Corley MJ, Premeaux TA, Lillard K, Coatney P, et al. Caspases and therapeutic potential of caspase inhibitors in moderate-severe SARS-CoV-2 infection and long COVID. Allergy. 2022;77(1):118–29. Epub 2021/05/17.
Arredondo J, Chernyavsky AI, Webber RJ, Grando SA. Biological effects of SLURP-1 on human keratinocytes. J Invest Dermatol. 2005;125(6):1236–41. Epub 2005/12/16.
Chernyavsky AI, Galitovskiy V, Shchepotin IB, Grando SA. Anti-inflammatory effects of the nicotinergic peptides SLURP-1 and SLURP-2 on human intestinal epithelial cells and immunocytes. Biomed Res Int. 2014;2014:609086. Epub 2014/05/31.
Mitchell JK, Pitcher D, McArdle BM, Alnefelt T, Duffy S, Avery V, et al. Identifying common metalloprotease inhibitors by protein fold types using Fourier transform mass spectrometry. Bioorg Med Chem Lett. 2007;17(23):6521–4. Epub 2007/10/16.
Paolocci N, Tavazzi B, Biondi R, Gluzband YA, Amorini AM, Tocchetti CG, et al. Metalloproteinase inhibitor counters high-energy phosphate depletion and AMP deaminase activity enhancing ventricular diastolic compliance in subacute heart failure. J Pharmacol Exp Ther. 2006;317(2):506–13. Epub 2006/01/27.
Okamoto Y, Satomura K, Nakayama K, Tanaka N, Ohsuzu F, Imaki J, et al. A matrix metalloproteinase inhibitor, ONO-4817, suppresses the development of aortic intimal hyperplasia in experimental hyperlipidemic rabbit. Int Heart J. 2007;48(3):369–78. Epub 2007/06/27.
Sindermann JR, Köbbert C, Voss R, Ebbing J, March KL, Breithardt G, et al. Transgenic model of smooth muscle cell cycle reentry: expression pattern of the collageneous matrix. Cardiovasc Pathology: Official J Soc Cardiovasc Pathol. 2008;17(2):72–80. Epub 2008/03/11.
Johnson LL, Bornemeier DA, Janowicz JA, Chen J, Pavlovsky AG, Ortwine DF. Effect of species differences on stromelysin-1 (MMP-3) inhibitor potency. An explanation of inhibitor selectivity using homology modeling and chimeric proteins. J Biol Chem. 1999;274(35):24881–7. Epub 1999/08/24.
Jacobsen EJ, Mitchell MA, Hendges SK, Belonga KL, Skaletzky LL, Stelzer LS, et al. Synthesis of a series of stromelysin-selective thiadiazole urea matrix metalloproteinase inhibitors. J Med Chem. 1999;42(9):1525–36. Epub 1999/05/07.
Matthews KH, Stevens HN, Auffret AD, Humphrey MJ, Eccleston GM. Formulation, stability and thermal analysis of lyophilised wound healing wafers containing an insoluble MMP-3 inhibitor and a non-ionic surfactant. Int J Pharm. 2008;356(1–2):110–20. Epub 2008/02/19.
Fray MJ, Dickinson RP. Discovery of potent and selective succinyl hydroxamate inhibitors of matrix metalloprotease-3 (stromelysin-1). Bioorg Med Chem Lett. 2001;11(4):571–4. Epub 2001/03/07.
Hibino T, Fujita E, Tsuji Y, Nakanishi J, Iwaki H, Katagiri C, et al. Purification and characterization of active caspase-14 from human epidermis and development of the cleavage site-directed antibody. J Cell Biochem. 2010;109(3):487–97. Epub 2009/12/05.
Kao JJ. The NF-kappaB inhibitor pyrrolidine dithiocarbamate blocks IL-1beta induced hyaluronan synthase 1 (HAS1) mRNA transcription, pointing at NF-kappaB dependence of the gene HAS1. Exp Gerontol. 2006;41(6):641–7. Epub 2006/05/26.
Marzoll A, Wördehoff L, Dai G, Fries S, Grosser T, Schrör K et al. Abstract 1161: cyclooxygenase 2 inhibitors reduce Hyaluronan Synthesis in Murine atherosclerosis. 2007;116(suppl_16):II234–5.
Stuhlmeier KM, Pollaschek C. Glucocorticoids inhibit induced and non-induced mRNA accumulation of genes encoding hyaluronan synthases (HAS): hydrocortisone inhibits HAS1 activation by blocking the p38 mitogen-activated protein kinase signalling pathway. Rheumatology. 2003;43(2):164–9.
Stuhlmeier KM. Effects of leflunomide on hyaluronan synthases (HAS): NF-kappa B-independent suppression of IL-1-induced HAS1 transcription by leflunomide. J Immunol (Baltimore Md: 1950). 2005;174(11):7376–82. Epub 2005/05/21.
Pasonen-Seppänen SM, Maytin EV, Törrönen KJ, Hyttinen JMT, Hascall VC, MacCallum DK, et al. All-trans Retinoic Acid-Induced Hyaluronan Production and Hyperplasia are partly mediated by EGFR Signaling in Epidermal Keratinocytes. J Invest Dermatology. 2008;128(4):797–807.
Davis BP, Stucke EM, Khorki ME, Litosh VA, Rymer JK, Rochman M, et al. Eosinophilic esophagitis-linked calpain 14 is an IL-13-induced protease that mediates esophageal epithelial barrier impairment. JCI Insight. 2016;1(4):e86355. Epub 2016/05/10.
Narumoto O, Horiguchi K, Horiguchi S, Moriwaki Y, Takano-Ohmuro H, Shoji S, et al. Down-regulation of secreted lymphocyte antigen-6/urokinase-type plasminogen activator receptor-related peptide-1 (SLURP-1), an endogenous allosteric alpha7 nicotinic acetylcholine receptor modulator, in murine and human asthmatic conditions. Biochem Biophys Res Commun. 2010;398(4):713–8. Epub 2010/07/14.
Hohl D, de Viragh PA, Amiguet-Barras F, Gibbs S, Backendorf C, Huber M. The small proline-rich proteins constitute a multigene family of differentially regulated cornified cell envelope precursor proteins. J Invest Dermatol. 1995;104(6):902–9. Epub 1995/06/01.
Krieg P, Schuppler M, Koesters R, Mincheva A, Lichter P, Marks F. Repetin (Rptn), a new member of the fused gene subgroup within the S100 gene family encoding a murine epidermal differentiation protein. Genomics. 1997;43(3):339–48. Epub 1997/08/01.
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
JW, KP, ALAS, MJL, SSR, RSL, and FB were involved in obtaining the specimens. JW was involved in lab work and sample processing. RSM performed the data analysis and wrote the preliminary draft of the manuscript. All authors provided their intellectual input on the manuscript and approved the final version for publication.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The institutional review board at Nuvance Health granted ethical approval for this study. All methods were carried out in accordance with relevant guidelines and regulations. Written informed consent was obtained from the study participants.
Consent for publication
Written informed consent for publication was obtained from the study participants.
Competing interests
Faiz Bhora, MD has consulting relationships with the following, that are unrelated to this study: AstraZeneca, Genentech, Biodesix, Johnson & Johnson/Ethicon, Boston Scientific, and Medtronic. The other authors have no relevant financial disclosures or competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Martins, R.S., Weber, J., Poulikidis, K. et al. Gene expression profiles in COVID-19-associated tracheal stenosis indicate persistent anti-viral response and dysregulated retinol metabolism. BMC Res Notes 17, 140 (2024). https://doi.org/10.1186/s13104-024-06775-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13104-024-06775-y