Abstract
Background
Toxoplasma gondii and Neospora caninum are closely related protozoan parasites that are considered important causes of abortion in livestock, causing huge economic losses. Hunan Province ranks 12th in the production of beef and mutton in China. However, limited data are available on the seroprevalence, risk factors and molecular characterization of T. gondii and N. caninum in beef cattle and goats in Hunan province, China.
Methods
Sera of 985 beef cattle and 1147 goats were examined for the presence of specific antibodies against T. gondii using indirect hemagglutination test (IHAT) and anti-N. caninum IgG using competitive-inhibition enzyme-linked immunoassay assay (cELISA). Statistical analysis of possible risk factors was performed using PASW Statistics. Muscle samples of 160 beef cattle and 160 goats were examined for the presence of T. gondii DNA (B1 gene) and N. caninum DNA (Nc-5 gene) by nested PCR. The B1 gene-positive samples were genotyped at 10 genetic markers using the multilocus nested PCR-RFLP (Mn-PCR-RFLP).
Results
Specific IgG against T. gondii were detected in 8.3% (82/985) and 13.3% (153/1147) and against N. caninum in 2.1% (21/985) and 2.0% (23/1147) of the beef cattle and goats, respectively. Based on statistical analysis, the presence of cats, semi-intensive management mode and gender were identified as significant risk factors for T. gondii infection in beef cattle. Age was a significant risk factor for T. gondii infection in goats (P < 0.05), and age > 3 years was a significant risk factor for N. caninum infection in beef cattle (P < 0.05). PCR positivity for T. gondii was observed in three beef samples (1.9%; 3/160) and seven chevon samples (4.4%; 7/160). Genotyping of PCR positive samples identified one to be ToxoDB#10. The N. caninum DNA was observed in one beef sample (0.6%; 1/160) but was negative in all chevon samples.
Conclusions
To our knowledge, this is the first large-scale serological and molecular investigation of T. gondii and N. caninum and assessment of related risk factors in beef cattle and goats in Hunan Province, China. The findings provide baseline data for executing prevention and control of these two important parasites in beef cattle and goats in China.
Graphical Abstract

Similar content being viewed by others
Background
Toxoplasmosis and neosporosis are two parasitic diseases with worldwide distribution, which are caused by Toxoplasma gondii and Neospora caninum, respectively [1]. Felids are the only definitive hosts of T. gondii, and all warm-blooded animals, including humans, can be infected as intermediate hosts [2]. Cattle and goats are two important intermediate hosts of T. gondii and N. caninum. Toxoplasma gondii and N. caninum are prevalent in ruminants worldwide, and they are associated with reproductive problems, particularly in cattle infected with N. caninum and in goats infected with T. gondii, causing significant economic losses [1, 3,4,5]. In China, the pooled seroprevalence of T. gondii in cattle and goats was 10.1% and 9.9%, respectively [6, 7]. The seroprevalence of N. caninum in cattle and goats varied in different regions of China, ranging from 9.4–20.9% to 3.9–7.2%, respectively [8, 9]. Infection in immunocompetent non-pregnant hosts is usually asymptomatic [10]. However, primary infection of humans and animals with T. gondii during pregnancy may result in abortion or congenital anomalies in the fetus [11]. In immunocompromised hosts, it may lead to severe generalized toxoplasmosis, which may even be fatal [12]. On the other hand, dogs are the most common definitive host of N. caninum, another important apicomplexan parasite in animals [3, 13, 14]. This parasite has been recognized as one of the most important causes of abortion in bovines worldwide [15]. In addition to dogs and cattle, natural infections have also been reported in goats, sheep, deer and horses, and these animals may also be potentially affected by N. caninum [16].
Although the average T. gondii prevalence in the Chinese population is relatively low, the total number of infected people is quite large because of the 1.4 billion total population of China [17]. An important route of infection in humans, next to contamination of food and water with oocysts, is consuming raw or undercooked meat (especially pork, chevon and beef) containing tissue cysts [18]. Toxoplasma gondii in cattle and goats is prevalent in some regions of China, which is a potential risk for transmitting the infection to humans. China is the major production and consumption market of beef and chevon worldwide, where production was about 7.18 million tons of beef and 5.25 million tons of mutton in 2022 (http://www.stats.gov.cn/tjsj/zxfb/202302/t20230227_1918980.html). Although N. caninum has not been proven to be a zoonotic pathogen, its impact on possible abortion and milk production in cattle is significant [13].
Hunan Province is located in the central-southern region of China, where its subtropical monsoon climate has endowed it with abundant vegetation. Additionally, the province boasts rich local breeds of cattle and goats, such as the Xiangxi Yellow Cattle, Xiangdong Black Goat and Matou Goat. These unique breeds have provided vast prospects for the livestock industry, further driving the expansion of the consumer market scale. According to statistics, beef and mutton production in Hunan Province has reached 356,500 tons, ranking 12th nationwide (https://m.voc.com.cn/xhn/news/202207/14095448.html). Considering the abundance of local breeds of cattle and goat in Hunan Province, as well as their potential in the breeding industry, investigations and molecular detection of T. gondii and N. caninum can play a crucial role in ensuring the quality and quantity of beef and mutton production in Hunan Province. However, the prevalence of these parasites in beef cattle and goats in Hunan Province has never been thoroughly investigated, with only one previous report [19]. Therefore, the objectives of the present study were to examine the seroprevalence and risk factors and to genetically characterize T. gondii and N. caninum in beef cattle and goats in Hunan Province, China.
Methods
Serum and muscle samples
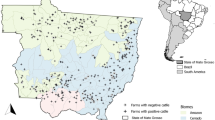
Based on geographical locations, Hunan Province is divided into five regions: central, western, eastern, southern and northern Hunan. In this study, a total of 985 beef cattle blood samples were collected from all regions except eastern Hunan (Fig. 1). These blood samples were collected from intensive farms as well as small holder farms. Meanwhile, 1147 goat blood samples were collected from intensive farms across the five regions of Hunan Province. The beef cattle and goats were randomly selected, with collection taking place between May 2018 and January 2019. Blood was drawn into glass tubes without anticoagulant from each animal on site. Samples were refrigerated and transferred to the laboratory to prepare sera and frozen at −20 °C until analysis. Information about the gender and age of the animals and the presence of free-roaming cats/dogs was recorded by local veterinarians.
Sample collection sites for beef cattle and goat serum samples from different regions of Hunan Province are marked on the map using ArcGIS 10.8 software (ESRI Inc., Redlands, CA, USA). (http://www.esri.com/software/arcgis/arcgis-for-desktop). The red hexagon represents the collection point of cattle serum samples, and the blue triangle represents the collection point of goat serum samples
A total of 320 intercostal muscle samples (160 beef and 160 chevon) were purchased from markets (farmers’ markets and supermarkets) in Hunan Province from September 2019 to November 2020. Each muscle tissue sample was cut into small pieces that weighed < 1 g and placed in a 2 ml centrifuge tube. The samples were then labeled and stored at −20 ℃.
Serological examination
Specific antibodies against T. gondii in animals were detected using a commercial indirect hemagglutination test (IHAT) kit (Lanzhou Veterinary Research Institute, Chinese Academy of Agricultural Science, Lanzhou, China) according to the manufacturer’s instructions [20, 21]. The serum samples were deemed positive when an agglutination reaction was observed in wells diluted at ≥ 1:64. Antibodies (IgG) to N. caninum were examined by competitive-inhibition enzyme-linked immunoassay assay (cELISA) using ID Screen® N. caninum Competition kit (IDVET, France) following the manufacturer’s recommendations [22]. Each ELISA plate included negative (nc) and positive controls (pc) provided by the manufacturer, and the samples' optical density (OD) was measured at 450 nm. The validity of the test was confirmed when both OD of negative control was > 0.700 and OD positive control/OD negative control < 0.3. For each sample, the competition percentage (S/N%) was determined using the formula: S/N % = (ODsample/OD negative control) × 100. Results were interpreted as follows: samples with S/N % ≤ 50% were considered positive, inconclusive if 50% < S/N % ≤ 60% and negative if S/N% > 60%.
DNA extraction, PCR detection and genetic characterization
Total genomic DNA was extracted from 200 mg muscle samples from each sample purchased from markets using a commercia kit (Promega, USA). A semi-nested PCR targeting the B1 gene (131 bp) was used to examine the T. gondii DNA [23]. Although the sensitivity of the 529-bp repeat element is higher than that of the B1 gene, the B1 gene has been commonly used as genetic marker for the molecular detection of T. gondii DNA [24]. Positive (DNA extracted from GT1 strain) and negative (ultrapure water) controls were included in all PCR reactions. The B1 gene-positive samples were genotyped at 10 genetic markers [SAG1, SAG2 (5′ + 3′ SAG2, alter.SAG2), SAG3, BTUB, GRA6, c22–8, c29–2, L358, PK1 and Apico] using the multiplex multilocus nested polymerase chain reaction-restriction fragment length polymorphism technology (Mn-PCR-RFLP), and eight reference T. gondii strains (GT1, PTG, CTG, MAS, TgCgCa1, TgCatBr5, TgCatBr64 and TgRsCr1) were included as controls as described previously [25]. The PCR products were digested with specific restriction endonucleases, followed by electrophoretic separation of the resulting restriction fragments. Subsequently, typing data were analyzed using the ToxoDB database (http://toxodb.org/toxo/) and compared against reference strain profiles.
The Nc-5 gene was used to detect N. caninum infection as described previously [26]. Positive (N. caninum NC-1 strain) and negative (ultrapure water) controls were included in all PCR reactions. The PCR products were validated by electrophoresis using 1% agarose gels containing GoldView™ (Beijing, China). The positive PCR products were sequenced by Tsingke Biotech (Beijing, China). The sequences acquired were subjected to analysis and alignment using MEGA 7 software [27]. The aligned sequences were further assessed and compared through BLAST analysis.
Statistical analysis
We categorized the samples by region, gender and age group (0–1, 1–2, 2–3 and > 3 years) for the analysis of potential infection risks. Additionally, as some beef cattle were semi-intensive, factors like presence of free-roaming cats/dogs and management modes (intensive or semi-intensive) were included for beef cattle samples. Variables associated with T. gondii and N. caninum infections among animals and different variables were analyzed using a chi-square test in PASW Statistics 18 (IBM Corporation, Somers, NY, USA), and 95% confidence intervals (CI) are also given. Probability (P) value < 0.05 was considered statistically significant.
Results
Sera from 82 of 985 beef cattle (8.3%) and 153 of 1147 goats (13.3%) were positive for the presence of T. gondii antibodies. Twenty-one of 985 beef cattle (2.1%) and 23 of 1147 goats (2.0%) were positive for N. caninum antibodies. Co-infection of both T. gondii and N. caninum was detected in five of the 2132 (0.2%) animals, with three of 985 beef cattle (0.3%) and two of 1147 (0.2%) goats being infected (Table 1).
The analysis results of possible risk factors of T. gondii infection in beef cattle and goats in Hunan Province are shown in Table 2 and Table 3. The presence of cats, semi-intensive management mode and gender were considered important risk factors for T. gondii infection in beef cattle (P < 0.05). The overall risk of cows being infected with T. gondii is nearly four times that of bulls (OR = 3.5, P < 0.001). The risk of T. gondii infection in beef cattle raised with cats in the environment is nearly twice that of beef cattle without cats (OR = 1.8, P < 0.05). The risk factors of semi-intensively raised beef cattle being infected with T. gondii were nearly two times those of intensively raised beef cattle (OR = 1.7, P < 0.05). In addition, for beef cattle in different regions of Hunan Province, the risk of beef cattle being infected with T. gondii in southern Hunan is nearly four times that in central Hunan (OR = 3.8, P < 0.05), and the risk of beef cattle being infected with T. gondii in northern Hunan is nearly three times of that in central Hunan (OR = 2.8, P < 0.05). Other factors including age were not statistically significant (P > 0.05).
For goats, we found that age was statistically significant among all possible risk factors for T. gondii infection. The seroprevalences of goats aged 0–1 (OR = 2.09, P < 0.05), 1–2 (OR = 1.88, P < 0.05) and > 3 years old (OR = 2.47, P < 0.05) are nearly twice those of goats aged 2 to 3 years old. In addition, for goats in different regions of Hunan Province, the risk of goats being infected with T. gondii in western Hunan is nearly two times that in central Hunan (OR = 2.34, P < 0.05).
The results of analysis of risk factors for N. caninum seroprevalence in beef cattle and goats in Hunan Province are shown in Additional file 1: Tables S1, S2. It was found that age > 3 years is a statistically significant risk factor for N. caninum seroprevalence in beef cattle (P < 0.05). Additionally, seroprevalence may vary between regions in goats. Goats in central and western Hunan have much higher risks [nearly 10 times (OR = 10.02, P < 0.05) and 12 times (OR = 12.36, P < 0.05), respectively] of being infected with N. caninum compared to goats in eastern Hunan.
PCR positivity for T. gondii was observed in three beef samples (1.9%; 3/160) and seven chevon samples (4.4%; 7/160) targeting the B1 gene. Only one of the three positive DNA samples provided data at all genetic loci and was identified as ToxoDB#10 (Table 4). However, the genotypes of seven chevon samples remained uncertain because of the ambiguity associated with two genetic markers. The N. caninum DNA was amplified in one beef sample (0.6%; 1/160) but was negative in all chevon samples. The Nc-5 sequence obtained in this study (accession no. MW590805) had 98.33% identity to a previously published corresponding N. caninum sequence (accession no. KP715562).
Discussion
This study revealed the widespread presence of T. gondii and N. caninum infections in common ruminants in Hunan Province. The present and previous results showed the presence of T. gondii infection in beef and chevon in China, posing a potential threat to human health [28, 29]. In certain regions of China, some people like to eat raw beef because of the mistaken belief that raw beef is highly nutritious [30]. Therefore, the risk of T. gondii infection in humans is greatly increased by eating raw and undercooked infected meat (beef and chevon).
In this study, serological assays showed that co-infection of beef cattle (0.3%) and goats (0.2%) with both parasites was rare, which is consistent with the results of many previous studies [21, 31, 32]. However, seroprevalence of T. gondii and N. caninum in cattle and goat varied greatly between countries and between different regions within the same country. The seroprevalence rate of T. gondii infection in beef cattle reported in this study (8.3%) is roughly similar to that in central Ethiopia (6.6%) and Indonesia (9%) but lower than that in Algeria (28.7%) and Libya (27.4%) [33,34,35,36]. We also compared the seroprevalence of T. gondii in cattle between Hunan Province and other provinces in China. The prevalence is higher than that previously found in Liaoning (6.0%) and Guangdong (5.7%) in China but is significantly lower than that reported in Guizhou (26.9%), Chongqing (27.2%), Qinghai (35.5%) and Xinjiang (46.4%) [37].
In goats, the seroprevalence of T. gondii infection observed here (13.3%) was similar to that reported in central Ethiopia (11.6%) but lower than in Algeria (33.6%), Pakistan (30%) and Egypt (38.2%) [33, 34, 38, 39]. The seroprevalence of T. gondii infection in goats in Hunan Province was higher than that found in Inner Mongolia (7.9%), Jiangxi (10.3%) and Heilongjiang (3.8%) but was significantly lower than that in Chongqing (42.5%), Yunnan (34.9%) and Shanxi (29.8%) [40]. These variations in seroprevalence may be related to the diagnostic techniques, climate, management mode of the farm and geographical conditions [41].
Our study showed that the N. caninum seroprevalence in beef cattle (2.1%) was similar to that in Malaysia (2.7%) but significantly lower than that in Argentina and Serbia (7.2%) [42,43,44,45]. In addition, it is also significantly lower than the average seroprevalence of N. caninum in cattle in China (13.69%) [46]. The lower seroprevalence of N. caninum in cattle in this study may be because the serum samples in this study were all from beef cattle. Some studies have shown that beef cattle are less susceptible to N. caninum infection than dairy cattle, so their seroprevalence rates tend to be lower [44, 47, 48]. The seroprevalence rate in goats (2%) is similar to that in beef cattle in this study and is also significantly lower than that of France (33%), Spain (6%) and Malaysia (3.9%) and significantly lower than that in Qinghai Province (7.2%), China [31, 32, 45, 49]. Furthermore, our study showed that the difference in N. caninum prevalence between beef cattle and goats was not significant, similar to a previous study [31]. The low N. caninum seroprevalence rates in beef cattle and goat in Hunan Province may be related to climate, geographical environment, the good management mode of the farm or different study designs and diagnostic methods [31, 43, 44, 50].
In addition to the study of seroprevalence, we also analyzed risk factors that can help formulate relevant prevention and control strategies. Some of the risk factors for T. gondii infection in beef cattle and goats demonstrated statistical significance. Among the beef cattle in this study, higher T. gondii seroprevalence was observed in cows, in semi-intensive management and on farms with cats. Our study demonstrated that cows exhibited higher seroprevalence than bulls, which could potentially be attributed to hormonal differences related to gender that influence immune responses against T. gondii [33, 41, 51, 52]. Semi-intensive raising and the presence of cats around the farm have been reported in many previous studies as risk factors for T. gondii infection [45, 53,54,55], and these factors increase the probability and frequency/chances of exposure of cattle to oocysts that are widely distributed in the environment. In addition, the seroprevalence of T. gondii in beef cattle in central Hunan was about two times lower than that in other areas of Hunan Province. There is a statistically significant difference in the seroprevalence of T. gondii in goats between western and other regions Hunan. Also, we observed a statistically significant difference in the seroprevalence of N. caninum between central Hunan and western Hunan compared to other regions. This variation may be due to differences in animal welfare, climate, investigation methods, animal husbandry practices and geography [41]. Among the goats in this study, significant differences in T. gondii seroprevalence were observed among goats of different ages. Taking the age group of 2 to 3 years as a reference, the T. gondii seroprevalences in goats aged 0–1, 1–2 and > 3 years old are nearly twice that in goats aged 2 to 3 years old, showing a trend of lowering first and then getting higher. This trend is different from most studies, which indicate that seroprevalence increases with age [33, 38, 41, 56]. The trend may be due to the following reasons. For newborn lambs from younger ewes, vertical transmission may be one of the reasons for the relatively high T. gondii antibody levels at this age. On the one hand, T. gondii is transmitted from the ewe to the fetus via the placenta, and lambs are born with circulating antibodies. On the other hand, tachyzoites of T. gondii may be present in breast milk, which may cause infection after suckling. Of course, among the possible reasons for the high antibody levels, it cannot be ruled out that lambs received maternal antibodies against T. gondii from colostrum [57, 58]. For older goats, longer exposure time to oocysts in the environment may be related to their higher antibody levels, and the same phenomenon was observed in our study of beef cattle > 3 years old infected with N. caninum [33, 59].
Previous studies have identified ToxoDB#225 and ToxoDB#10 in cattle in Henan and Jilin Provinces [37] and ToxoDB#9 and ToxoDB#10 in goats in Yunnan Province in China [60]. Considering previous molecular reports in Hunan Province, ToxoDB#10 was also identified from wild birds [61]. However, in this study, only one genotype (ToxoDB#10) was identified in one beef sample. The genotyping results from chevon samples were only suggestive because of the uncertainty of two genetic markers. ToxoDB#9 (Chinese 1) and ToxoDB#10 (Type I) are considered the predominant genotypes of T. gondii in China, and our detection of ToxoDB#10 in beef provides new evidence supporting this view [62, 63]. In addition, our study only detected one positive sample of N. caninum collected from a farmer's market in Changsha City. This is the first report of amplification of N. caninum Nc-5 gene in beef cattle muscle tissue from Hunan Province. The number and sources of samples need to be further expanded in the future to obtain more information on the genotypes of T. gondii and N. caninum in infected beef cattle and goats.
Conclusions
This study represents the first extensive serological and molecular examination of T. gondii and N. caninum infection and associated risk factors in beef cattle and goat populations in Hunan Province, China. Our results may provide baseline information for the development of control measures against these parasitic infections in beef cattle and goats in this province and elsewhere. Future studies are necessary to elucidate the potential effect of both T. gondii and N. caninum on reproduction of ruminants. It is recommended to implement good management measures on ruminant farms to decrease the T. gondii infection in cattle and goats.
Availability of data and materials
The datasets supporting the findings of this article are included within the paper and its supplementary materials. The Nc-5 sequence of Neospora caninum obtained in this study is available at GenBank (accession no. MW590805).
Abbreviations
- IHAT:
-
Indirect hemagglutination test
- cELISA:
-
Competitive-inhibition enzyme-linked immunoassay assay
- OD:
-
Optical density
- S/N%:
-
Competition percentage
- PCR:
-
Polymerase chain reaction
- Mn-PCR–RFLP:
-
Multilocus nested polymerase chain reaction-restriction fragment length polymorphism technology
- OR:
-
Odds ratio
- CI:
-
Confidence interval
References
De Barros LD, Garcia JL, Bresciani KDS, Cardim ST, Storte VS, Headley SA. A review of toxoplasmosis and neosporosis in water buffalo (Bubalus bubalis). Front Vet Sci. 2020;7:455.
Smith NC, Goulart C, Hayward JA, Kupz A, Miller CM, van Dooren GG. Control of human toxoplasmosis. Int J Parasitol. 2021;51:95–121.
Ansari-Lari M. Bovine neosporosis in Iran: a systematic review and meta-analysis. Prev Vet Med. 2020;176:104913.
Odeniran PO, Omolabi KF, Ademola IO. A meta-analysis of Toxoplasma gondii seroprevalence, genotypes and risk factors among food animals in West African countries from public health perspectives. Prev Vet Med. 2020;176:104925.
Lindsay DS, Dubey JP. Neosporosis, toxoplasmosis, and sarcocystosis in ruminants: an update. Vet Clin North Am Food Anim Pract. 2020;36:205–22.
Gong Q-L, Li J, Li D, Tian T, Leng X, Li J-M, et al. Seroprevalence of Toxoplasma gondii in cattle in China from 2010 to 2019: a systematic review and meta-analysis. Acta Trop. 2020;211:105439.
Wei X-Y, Gong Q-L, Zeng A, Wang W, Wang Q, Zhang X-X. Seroprevalence and risk factors of Toxoplasma gondii infection in goats in China from 2010 to 2020: a systematic review and meta-analysis. Prev Vet Med. 2021;186:105230.
Wei X-Y, An Q, Xue N-Y, Chen Y, Chen Y-Y, Zhang Y, et al. Seroprevalence and risk factors of Neospora caninum infection in cattle in China from 2011 to 2020: a systematic review and meta-analysis. Pre Vet Med. 2022;203:105620.
Rodrigues AA, Reis SS, de Sousa ML, da Silva ME, Garcia JL, Nascimento TVC, et al. A systematic literature review and meta-analysis of risk factors for Neospora caninum seroprevalence in goats. Prev Vet Med. 2020;185:105176.
Torgerson PR, de Silva NR, Fevre EM, Kasuga F, Rokni MB, Zhou X-N, et al. The global burden of foodborne parasitic diseases: an update. Trends Parasitol. 2014;30:20–6.
Nayeri T, Sarvi S, Moosazadeh M, Amouei A, Hosseininejad Z, Daryani A. The global seroprevalence of anti-Toxoplasma gondii antibodies in women who had spontaneous abortion: a systematic review and meta-analysis. PLoS Negl Trop Dis. 2020;14:e0008103.
Ahmadpour E, Daryani A, Sharif M, Sarvi S, Aarabi M, Mizani A, et al. Toxoplasmosis in immunocompromised patients in Iran: a systematic review and meta-analysis. J Infect Dev Ctries. 2014;8:1503–10.
Dubey J, Schares G, Ortega-Mora L. Epidemiology and control of neosporosis and Neospora caninum. Clin Microbiol Rev. 2007;20:323–67.
Dubey J, Schares G. Neosporosis in animals—the last five years. Vet Parasitol. 2011;180:90–108.
Sánchez-Sánchez R, Vázquez P, Ferre I, Ortega-Mora LM. Treatment of toxoplasmosis and neosporosis in farm ruminants: state of knowledge and future trends. Curr Top Med Chem. 2018;18:1304–23.
Pitel P-H, Pronost S, Chatagnon G, Tainturier D, Fortier G, Ballet J-J. Neosporosis in bovine dairy herds from the west of France: detection of Neospora caninum DNA in aborted fetuses, seroepidemiology of N. caninum in cattle and dogs. Vet Parasitol. 2001;102:269–77.
Zhou P, Chen Z, Li HL, Zheng H, He S, Lin RQ, et al. Toxoplasma gondii infection in humans in China. Parasit Vectors. 2011;4:165.
Elsheikha HM, Marra CM, Zhu X-Q. Epidemiology, pathophysiology, diagnosis, and management of cerebral toxoplasmosis. Clin Microbiol Rev. 2021;34:e00115-e119.
Li F, Wang S-P, Wang C-J, He S-C, Wu X, Liu G-H. Seroprevalence of Toxoplasma gondii in goats in Hunan province China. Parasite. 2016;23:44.
Wang Y-G, Gui B-Z, Li R-C, Wang G-P, Ge M, Liu G-H. Seroprevalence and risk factors of Toxoplasma gondii infection in growth stages of pigs in Hunan province subtropical China. Vector Borne Zoonotic Dis. 2019;19:945–9.
Sun L-X, Liang Q-L, Nie L-B, Hu X-H, Li Z, Yang J-F, et al. Serological evidence of Toxoplasma gondii and Neospora caninum infection in black-boned sheep and goats in southwest China. Parasitol Int. 2020;75:102041.
Gui BZ, Lv QY, Ge M, Li RC, Zhu XQ, Liu GH. First report of Neospora caninum infection in pigs in China. Transbound Emerg Dis. 2020;67:29–32.
Burg JL, Grover CM, Pouletty P, Boothroyd J. Direct and sensitive detection of a pathogenic protozoan, Toxoplasma gondii, by polymerase chain reaction. J Clin Microbiol. 1989;27:1787–92.
Belaz S, Gangneux J-P, Dupretz P, Guiguen C, Robert-Gangneux F. A 10-year retrospective comparison of two target sequences, REP-529 and B1, for Toxoplasma gondii detection by quantitative PCR. J Clin Microbiol. 2015;53:1294–300.
Su C, Shwab E, Zhou P, Zhu X, Dubey J. Moving towards an integrated approach to molecular detection and identification of Toxoplasma gondii. Parasitology. 2010;137:1–11.
Müller N, Sager H, Hemphill A, Mehlhorn H, Heydorn A, Gottstein B. Comparative molecular investigation of Nc5-PCR amplicons from Neospora caninum NC-1 and Hammondia heydorni-Berlin-1996. Parasitol Res. 2001;87:883–5.
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 2011;28:2731–9.
Ge W, Sun H, Wang Z, Xu P, Wang W, Mu G, et al. Prevalence and genotype of Toxoplasma gondii infection in cattle from Jilin Province Northeastern China. Vector Borne Zoonotic Dis. 2014;14:399–402.
Amdouni Y, Rjeibi MR, Rouatbi M, Amairia S, Awadi S, Gharbi M. Molecular detection of Toxoplasma gondii infection in slaughtered ruminants (sheep, goats and cattle) in Northwest Tunisia. Meat Sci. 2017;133:180–4.
Zhou M, Zhang N, Zhang M, Ma G. Culture, eating behavior, and infectious disease control and prevention. J Ethn Food. 2020;7:40.
Moskwa B, Kornacka A, Cybulska A, Cabaj W, Reiterova K, Bogdaszewski M, et al. Seroprevalence of Toxoplasma gondii and Neospora caninum infection in sheep, goats, and fallow deer farmed on the same area. J Anim Sci. 2018;96:2468–73.
Liu Z-K, Li J-Y, Pan H. Seroprevalence and risk factors of Toxoplasma gondii and Neospora caninum infections in small ruminants in China. Prev Vet Med. 2015;118:488–92.
Abdallah M-C, Kamel M, Karima B, Samir A, Djamel K, Rachid K, et al. Cross-sectional survey on Toxoplasma gondii infection in cattle, sheep, and goats in Algeria: seroprevalence and risk factors. Vet Sci. 2019;6:63.
Bekele T, Kasali O. Toxoplasmosis in sheep, goats and cattle in central Ethiopia. Vet Res Commun. 1989;13:371–5.
Matsuo K, Husin D. A survey of Toxoplasma gondii antibodies in goats and cattle in Lampung province, Indonesia. Southeast Asian J Trop Med Public Health. 1996;27:554–5.
Azwai S, El-Gammoudi F, Gameel S. A serological survey of toxoplasmosis in some animal species in Libya. Alex J Vet Sci. 1993;9:133–5.
Dubey J, Murata F, Cerqueira-Cézar C, Kwok O, Yang Y. Public health significance of Toxoplasma gondii infections in cattle: 2009–2020. J Parasitol. 2020;106:772–88.
Mumtaz T, Awan UA, Mushtaq A, Afzal MS, Mahmood T, Wasif S, et al. Prevalence of toxoplasmosis in sheep and goats in Pakistan: a systematic review and meta-analysis. Pathogens. 2022;11:1331.
Selim A, Marzok M, Alshammari A, Al-Jabr OA, Salem M, Wakid MH. Toxoplasma gondii infection in Egyptian domestic sheep and goats: seroprevalence and risk factors. Trop Anim Health Prod. 2023;55:182.
Dubey JP, Murata F, Cerqueira-Cézar C, Kwok O. Public health and economic importance of Toxoplasma gondii infections in goats: The last decade. Res Vet Sc. 2020;132:292–307.
Symeonidou I, Sioutas G, Lazou T, Gelasakis AI, Papadopoulos E. A review of Toxoplasma gondii in animals in Greece: a foodborne pathogen of public health importance. Animals. 2023;13:2530.
Campero L, Basso W, Moré G, Fiorani F, Hecker Y, Echaide I, et al. Neosporosis in Argentina: past, present and future perspectives. Vet Parasitol Reg Stud Reports. 2023;41:100882. https://doi.org/10.1016/j.vprsr.2023.100882.
Klun I, Ćirković V, Maletić M, Bradonjić S, Djurković-Djaković O. Seroprevalence of Neospora caninum infection and associated risk factors in dairy cattle in Serbia. Parasitol Res. 2019;118:1875–83.
Samkange A, Chitanga S, Tjipura-Zaire GN, Mutjavikua VG, Smith JW, Neves L, et al. Seroprevalence and associated risk factors of bovine neosporosis in the Khomas region of Namibia. Onderstepoort J Vet Res. 2023;90:2077.
Sadiq MB, Muhamad AS, Hamdan SA, Ramanoon SZ, Zakaria Z, Aziz NAA, et al. Seroprevalence and factors associated with Toxoplasma gondii, Neospora caninum, and Besnoitia besnoiti infections in cattle and goats in Selangor. Malaysia Animals. 2023;13:948.
Ying Z, Zhu Z-F, Yang X, Liu J, Liu Q. Prevalence and associated risk factors of Neospora caninum infection among cattle in mainland China: a systematic review and meta-analysis. Prev Vet Med. 2022;201:105593.
Gharekhani J, Yakhchali M, Berahmat R. Neospora caninum infection in Iran (2004–2020): a review. J Parasit Dis. 2020;44:671–86.
Ribeiro CM, Soares IR, Mendes RG, de Santis Bastos PA, Katagiri S, Zavilenski RB, et al. Meta-analysis of the prevalence and risk factors associated with bovine neosporosis. Trop Anim Health Prod. 2019;51:1783–800.
Díaz P, Cabanelas E, Díaz JM, Viña M, Béjar JP, Pérez-Creo A, et al. Seroprevalence of Toxoplasma gondii and Neospora caninum in goats from north-western Spain. Ann Agric Environ Med. 2016;23:587–90.
Cao H, Zheng W-B, Wang Y, Gao W-W, Liu Q, Zhu X-Q, et al. Seroprevalence of Neospora caninum infection and associated risk factors in cattle in Shanxi province, North China. Front Vet Sci. 2022;9:1053270.
Walker W, Roberts CW, Ferguson D, Jebbari H, Alexander J. Innate immunity to Toxoplasma gondii is influenced by gender and is associated with differences in interleukin-12 and gamma interferon production. Infect Immun. 1997;65:1119–21.
Harding AT, Heaton NS. The impact of estrogens and their receptors on immunity and inflammation during infection. Cancers. 2022;14:909.
Tenter AM, Heckeroth AR, Weiss LM. Toxoplasma gondii: from animals to humans. Int J Parasitol. 2000;30:1217–58.
Dámek F, Swart A, Waap H, Jokelainen P, Le Roux D, Deksne G, et al. Systematic review and modelling of age-dependent prevalence of Toxoplasma gondii in livestock, wildlife and felids in Europe. Pathogens. 2023;12:97.
Stelzer S, Basso W, Silván JB, Ortega-Mora LM, Maksimov P, Gethmann J, et al. Toxoplasma gondii infection and toxoplasmosis in farm animals: risk factors and economic impact. Food Waterborne Parasitol. 2019;15:e00037.
Tilahun B, Tolossa YH, Tilahun G, Ashenafi H, Shimelis S. Seroprevalence and risk factors of Toxoplasma gondii infection among domestic ruminants in East Hararghe zone of Oromia Region. Ethiopia Vet Med Int. 2018;2018:4263470.
Costa FT, Nogueira DB, Oliveira MA, Silva SS, Silva RF, Sarmento WF, et al. Vertical transmission of Toxoplasma gondii in naturally infected ewes in the semiarid region of Brazil. Comp Immunol Microbiol Infect Dis. 2021;74:101595.
Sousa LN, Batista SP, Silva SS, da Silva RF, Sarmento WF, Mota RA, et al. Dynamics of natural infection by Toxoplasma gondii in goat herds in the semiarid region of northeastern Brazil: transitional antibody observation. Vet Res Commun. 2022;46:507–15.
Lopes AP, Dubey J, Neto F, Rodrigues A, Martins T, Rodrigues M, et al. Seroprevalence of Toxoplasma gondii infection in cattle, sheep, goats and pigs from the North of Portugal for human consumption. Vet Parasitol. 2013;193:266–9.
Miao Q, Huang S-Y, Qin S-Y, Yu X, Yang Y, Yang J-F, et al. Genetic characterization of Toxoplasma gondii in Yunnan black goats (Capra hircus) in southwest China by PCR-RFLP. Parasit Vectors. 2015;8:57.
Liu M-T, Jiang W-X, Gui B-Z, Jin Y-C, Yi J-N, Li F, et al. Molecular prevalence and genetic characterization of Toxoplasma gondii in wild birds in Hunan province. China Vector Borne Zoonotic Dis. 2019;19:378–83.
Shwab EK, Zhu X-Q, Majumdar D, Pena HF, Gennari SM, Dubey JP, et al. Geographical patterns of Toxoplasma gondii genetic diversity revealed by multilocus PCR-RFLP genotyping. Parasitology. 2014;141:453–61.
Dong H, Su R, Lu Y, Wang M, Liu J, Jian F, et al. Prevalence, risk factors, and genotypes of Toxoplasma gondii in food animals and humans (2000–2017) from China. Front Microbiol. 2018;9:2108.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Key Research and Development Program of China (grant nos. 2021YFC2300800 and 2021YFC2300802), Hunan Provincial Natural Science Foundation of China (grant no. 2023JJ5005) and the Scientific Research Fund of Hunan Provincial Education Department (grant nos. 19A218 and 20C0972).
Author information
Authors and Affiliations
Contributions
GHL and XQZ conceived the study, designed the experiments and critically revised the manuscript. XLY, WHY, HLZ and MLC performed the experiments, analyzed the data and drafted the manuscript. JX, WCC, YJZ and FL participated in the implementation of the study. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The animal study was reviewed and approved by the Ethics Committee of Hunan Agricultural University (no. 43321503).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests. The co-corresponding author Prof. Xing-Quan Zhu serves as the Subject Editor for the section “Parasite genetics, genomics and proteomics” of Parasites & Vectors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1.
Analysis of risk factors for Neospora caninum infection in beef cattle in Hunan Province, China. Table S2. Analysis of risk factors for Neospora caninum infection in goats in Hunan Province, China.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yi, XL., Yang, WH., Zheng, HL. et al. Seroprevalence and molecular detection of Toxoplasma gondii and Neospora caninum in beef cattle and goats in Hunan province, China. Parasites Vectors 17, 195 (2024). https://doi.org/10.1186/s13071-024-06283-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13071-024-06283-9