Abstract
Background
In the UK, routinely recorded data may benefit prospective studies including randomised controlled trials (RCTs). In an on-going study, we aim to assess the feasibility of access and agreement of routinely recorded clinical and non-clinical data compared to data collected during a RCT using standard prospective methods. This paper will summarise available UK routinely recorded data sources and discuss our experience with the feasibility of accessing routinely recorded data for participants of a RCT before finally proposing recommendations for improving the access and implementation of routinely recorded data in RCTs.
Methods
Setting: the case study RCT is the Standard and New Antiepileptic Drugs II (SANAD II) trial, a pragmatic, UK, multicentre, phase IV RCT assessing the clinical and cost-effectiveness of antiepileptic drug treatments for newly diagnosed epilepsy.
Participants: 98 participants have provided written consent to permit the request of routinely recorded data.
Study procedures: routinely recorded clinical and non-clinical data were identified and data requested through formal applications from available data holders for the duration that participants have been recruited into SANAD II. The feasibility of accessing routinely recorded data during a RCT is assessed and recommendations for improving access proposed.
Results
Secondary-care clinical and socioeconomic data is recorded on a national basis and can be accessed, although there are limitations in the application process. Primary-care data are recorded by a number of organisations on a de-identified basis but access for specific individuals has not been feasible. Access to data recorded by non-clinical sources, including The Department for Work and Pensions and The Driving and Vehicle Licensing Agency, was not successful.
Conclusions
Recommendations discussed include further research to assess the attributes of routinely recorded data, an assessment of public perceptions and the development of strategies to collaboratively improve access to routinely recorded data for research.
Trial registration
International Standard Randomised Controlled Trials, ISRCTN30294119. Registered on 3 July 2012. EudraCT No: 2012-001884-64. Registered on 9 May 2012.
Similar content being viewed by others
Background
There is a plethora of individual-level, routinely recorded data in the UK. These data are recorded to fulfil specific, defined purposes and are regulated for security, confidentiality and disclosure by The Data Protection Act 1998 [1] and The Freedom of Information Act 2000 [2]. Access to routinely recorded data for ‘secondary purposes’, such as clinical research, is permitted providing that there is demonstrable secondary benefit.
The potential for routinely recorded data to inform clinical research and Health Technology Assessment (HTA) has long been recognised [3]. Presently, there are a number of sources of routinely recorded primary and secondary-care clinical data with regional or national coverage. However, limitations with accuracy of coding, confidentiality, ownership and data access have been previously identified as significant barriers to using routinely recorded data in research [4].
There are numerous examples of retrospective, observational, record-linkage population studies where routine sources have proved a valid and efficient method for providing data for clinical research [5]. In the context of prospective research, such as randomised controlled trials (RCTs), routinely recorded data have been used to inform judgements about the feasibility of sample size and recruitment targets [6] and measuring participant outcomes [3, 7]. Pragmatic cluster RCTs have been coordinated through routine data sources including patient recruitment, randomisation, and administration of intervention and trial assessments, such as through the Clinical Practice Research Datalink (CPRD) [8]. The majority of RCTs incur costs as clinicians assess participants, record outcomes and complete Case Report Forms – hence, using routinely recorded data may provide an efficient alternative method for data collection in addition to reducing the burden on participants. Furthermore, data from non-clinical routine sources may inform outcomes beyond the standard RCT assessments of clinical efficacy and effectiveness. For example, cost data (such as use of health care resources) and socioeconomic data (such as employment and means-tested benefits data) may inform health economic analyses and the assessment of the broader societal impact of health care interventions.
The potential benefits of using routinely recorded data in clinical research have resulted in a political drive to increase implementation, detailed in The Plan for Growth [9] and The NHS Constitution [10], where research is presented as a core activity making the link explicit between the provision of NHS services and research. Consequently, initiatives, such as the Administrative Data Research Network [11], have been established to provide a method of access to individual-level data, linking clinical and non-clinical sources of routinely recorded data.
The objective of this paper is to review relevant sources of routinely recorded data for England, Scotland and Wales and to discuss our experience with the feasibility of accessing individual-level data for a subgroup of participants enrolled into a RCT before finally proposing recommendations for improving the access and implementation of routinely collected data in RCTs. This is an on-going study and in a future publication we aim to assess the agreement of routinely recorded data compared to paired data collected in a RCT using standard prospective methods.
Methods
The case study RCT is the Standard and New Antiepileptic Drugs (SANAD) II trial. SANAD II is a pragmatic, UK, multicentre, phase IV RCT funded by the National Institute for Health Research (NIHR) Health Technology Assessment (HTA) programme, assessing the clinical and cost-effectiveness of a number of antiepileptic drugs as first-line treatments for newly diagnosed epilepsy. Data for clinical outcomes, including seizure freedom and adverse events, are recorded on Case Report Forms by the treating clinical team during outpatient appointments. Data to inform cost-effectiveness analyses, including health care resource use and quality of life, are recorded through participant completion of questionnaires. SANAD II is currently recruiting and is expected to report in 2019.
Following research ethics and governance approvals, 470 participants enrolled in SANAD II were invited to provide written consent to permit the request of routinely recorded data for the duration of their participation in SANAD II. Ninety-eight (20.9%) participants provided consent and were included in the study. Relevant sources of routinely recorded data were identified and detailed scoping discussions ensued. Subsequently, where accessible, routinely recorded data for participants recruited into SANAD II were requested through formal applications. The routinely recorded data sources included in this study are as follows:
-
Clinical routine data sources: secondary care:
-
◦ The Health and Social Care Information Centre (HSCIC)
-
◦ The NHS Wales Informatics Service (NWIS)
-
◦ The NHS National Services Scotland; Information Services Division (ISD)
-
-
Clinical routine data sources: primary care:
-
◦ The Clinical Practice Research Datalink (CPRD)
-
◦ ResearchOne
-
◦ QResearch
-
◦ The Health Improvement Network (THIN) database
-
◦ North West eHealth (NWEH)
-
-
Non-clinical routine data sources:
-
◦ The Office for National Statistics (ONS)
-
◦ HM Revenue and Customs (HMRC)
-
◦ The Department for Work and Pensions (DWP)
-
◦ The Driver and Vehicle Licensing Authority (DVLA)
-
-
‘Linked’ routine data sources:
-
◦ The Secure Anonymised Information Linkage (SAIL) databank
-
◦ The Administrative Data Research Network (ADRN)
-
In a future publication, the agreement between routinely recorded data and data collected using standard prospective methods will be assessed for baseline variables such as gender, age and date of first seizure, and for outcome measures relevant to SANAD II such as time to 12-month remission from seizures. To assess agreement between paired continuous data, Bland-Altman methods will be employed. Acceptable clinical limits of agreement for each variable or SANAD II outcome will be specified a priori and compared to the 95% confidence limits of agreement. To assess agreement between paired, nominal categorical datasets, cross tabulations will be constructed followed by calculation of Cohen’s Kappa.
Results
Clinical routine data sources: secondary care
Electronic medical records of patients’ use of secondary-care services in the UK are routinely managed on a national basis. A number of public service organisations provide national information, data and IT systems for commissioners, analysts and clinicians in health and social care. Data are recorded to inform patient care, provide the data for remuneration for hospital trusts and are subsequently used to monitor and improve clinical services through clinical research. Table 1 summarises the data sources where access to individual-level data is possible.
Clinical routine data sources: primary care
Electronic medical records of patients’ use of primary-care services in the UK are recorded routinely by the general practitioner to inform patient care and remuneration, but are not currently available for clinical research on a national basis. A number of organisations represent collaborations between governmental bodies or academic institutions and providers of primary-care IT systems. Access on a regional basis is possible through a number of data sources summarised in Table 2.
Non-clinical routine data sources
Non-clinical, individual-level data are routinely recorded by a number of UK governmental departments for a variety of indications. Selected organisations record data that would be informative to prospective clinical research in epilepsy and other diseases, summarised in Table 3.
‘Linked’ routine data sources
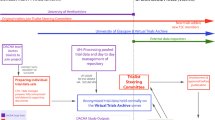
In order to provide a ‘complete’ dataset of the information required to meet research objectives, data from a number of organisations may need to be accessed. This is typically accomplished by linking data sources using identifiers such as patients’ name, date of birth, National Insurance number or NHS number. In response to the growing recognition of the potential of routinely recorded data, initiatives have been established to assist with the provision of linked, de-identified, aggregate data between data sources:
-
The Secure Anonymised Information Linkage (SAIL) Databank is an initiative developed by Swansea University and funded by the Welsh Government. SAIL provides a method of access to individual-level, routinely recorded, de-identified electronic data for patients across Wales to support research [12]. Access to clinical datasets provided by NWIS is complemented with numerous non-clinical administrative datasets including births, deaths and demographic data. Following the scoping process a formal application is submitted to the Information Governance Review Panel before access to data is granted. SAIL data have been accessed to measure clinical outcomes in retrospective research [13]
-
The Administrative Data Research Network (ADRN) is a UK-wide partnership between universities, government departments, national statistics authorities, funders and researchers, funded by the Economic and Social Research Council. ADRN provides a method of access to a number of non-clinical administrative routine datasets including employment, socioeconomic, crime and education data [11] in addition to clinical datasets detailed previously such as those recorded by HSCIC. Following development of a project proposal a formal application is reviewed by the Approvals Panel before access to data is granted
Challenges and feasibility of access
We have requested access to routinely recorded data for individuals enrolled in the SANAD II RCT, resident in England and Wales, who have provided written consent. There were insufficient participants meeting the eligibility criteria resident in Scotland. Data sources were identified and scoping discussions informed the initial assessment of feasibility. Data sources were deemed feasible if individual-level data could be provided for specified individuals providing consent. Resources required including cost and researcher time were also factors important in the assessment of feasibility. Including the preparation, research ethics and governance approval and submission of the applications for data access, significant researcher time and a period of 18 months were required. The feasibility, timeline and key milestones involved for each data source are summarised in Table 4.
Clinical routine data sources
Routinely recorded secondary-care data can be requested on an individual-level, identifiable basis for patients in England and Wales through HSCIC and NWIS, accessed through SAIL and in our experience this process is feasible as part of a RCT, yet there are notable limitations. In England, HSCIC has set a target time to data access of sixty working days following submission for a complex application, involving bespoke data linkage from multiple datasets. From the date of submission of the Data Access Request Service online application, we have been granted access to the data within this timeframe. However, this positive experience following submission of the application is countered by limitations in the pre-application process. Acknowledging the significant update to online application and approval procedures that occurred during this period, there remains a considerable period of time required in the development of the application. The nature of the request for identifiable data necessitated participant consent as the valid legal basis. HSCIC require ethical and governance approval to be in place prior to DARS review and to prevent future amendments and delays, it was rational to ensure the consent materials had been reviewed by the HSCIC’s Information Governance Team, prior to submitting the documents for ethical and governance approval. HSCIC provide written guidance regarding the consent materials and advise that documents should be reviewed. However, in our experience there is no formalised process for providing this review. Following significant correspondence the consent materials were reviewed by the Data Access and Information Sharing Team. However, this feedback was provided following a formal submission and review by the Data Access Request Service. Formalising the process for the review of consent materials would likely improve the time and resource efficiency for both HSCIC and the researcher.
For participants in Wales, we have requested secondary-care data and, for a proportion of participants, primary-care data through SAIL databank. SAIL provided a streamlined pre-application service, including engaging in multiple discussions and completion of a scoping document outlining the study methods and costs involved. Consent materials were also promptly reviewed by a member of the Information Governance Team.
Common to both sources of secondary-care, routinely recorded data; there are stringent information governance requirements that must be in place prior to application. These include information security measures and assessments, specific inclusion regarding the ‘processing of health care data for the subjects of research’ in the institutional Data Protection Act registration and, in the case of HSCIC, an institutional Data Sharing Framework Contract. Adequate guidance is provided by the data sources and, if not addressed by the researcher, may cause delay. Furthermore, there is a time lag of approximately 3–6 months before data become available within each data source. This delay potentially limits the utility of such sources in prospective clinical research, such as drug trials, where prompt reporting is clinically important and a regulatory requirement.
Routinely recorded primary-care data for specific participants in England are less accessible. The majority of providers of primary-care data, such as ResearchOne and QResearch, provide data on a de-identified basis with no facility to re-identify individuals. Therefore, where specific participants need to be identified, as for RCTs such as SANAD II, these sources are not applicable. Following our correspondence, CPRD confirmed it may be possible to retrieve identifiable individual-level data linked to HSCIC data in the future, but the required approvals were not in place and the timescale to resolution was unclear. Furthermore, such primary-care sources provide data for only a proportion of the population and can be expensive.
North West eHealth employs an alternative methodology whereby primary-care data are extracted directly from the GP through a third party. This process requires participant and GP consent and installation of the required software but is an effective data-extraction method [14]. NWEH offers a number of primary-care research tools for the wider research community but does not currently routinely provide a bespoke primary-care data-extraction service for research.
Non-clinical routine data sources
Aggregate economic and societal statistics, provided by Lower Layer Super Output Area (LSOA), can be accessed through the ONS and are in the public domain. Such data may have additional benefits to the analyses of health and socioeconomic outcomes in RCTs. Individual-level, economic data from sources such as the DWP and HMRC would likely be informative to prospective clinical research as such data are often poorly or incompletely recorded using standard methods [15]. However, relevant to this study, there is no previous evidence of access to DWP or HMRC individual-level or aggregate data for clinical research.
During scoping discussions with DWP and HMRC, we were directed to ADRN but this network has not been successful in negotiating data access.
Finally, the outcomes of selected clinical studies may be measured using DVLA data. However, the DVLA declined the request for access, citing insufficient internal resources to process the request and more stringent data protection requirements than those employed in the NHS or academic institutions, without providing explicit details regarding these requirements.
Discussion
Routinely recorded data are valid for use in retrospective clinical research [3, 4] and have the potential to be used in prospective research including measuring the outcomes of RCTs [7] and providing additional benefits such as a method to address missing RCT data. Limitations, specifically with respect to accuracy and access have been recognised for some time. Academic, political [9] and health service [10] interest in UK sources of routinely recorded data has resulted in expansion and improvements, notably in the access to linked datasets. However, our experience with accessing individual-level data for specific participants providing written consent, to inform the outcomes of a RCT, highlights persisting limitations.
Clinical routine data sources are numerous and there is comprehensive national coverage of secondary-care data. In our experience, accessing individual-level data is feasible. However, inefficiencies in the application processes persist, particularly during the informal ‘pre-application’ phase. The notable limitation encountered was obtaining feedback on the Patient Information Sheet and Consent Form prior to ethical and governance review. Formalising an explicit review process for consent materials would improve the efficiency for both the data holders and the research team.
Access to routinely recorded, individual-level, primary-care data has not been feasible. Each primary-care data source has limited geographical coverage, often based on GP IT systems, which usually process de-identified data and may incur significant expense. The inception of the HSCIC General Practice Extraction Service, which records primary-care data nationally for England, represents the most optimistic national source; however, access is currently restricted to Department of Health initiatives such as research involving screening procedures [16].
The access to non-clinical data sources for clinical research has not been possible. ADRN has been established to act on behalf of the researcher in negotiating access to de-identified, linked, routinely recorded data from a number of organisations and the study proposal was promptly directed to ADRN. However, the decision whether to release data remains with the data holder. Ideologically, the next step would be the storage of de-identified linked data from participating organisations in a single repository, similar to those established for RCT data [17]. This would create a single point of access and remove the burden for each organisation to consider each study individually. This would, however, require significant information governance and security barriers to be cleared and, in light of recent developments within the research climate, individual consent. Including patients as stakeholders in the development of such data sources is essential [18].
Although there are examples of pragmatic RCTs being coordinated through routine data sources [8], there are likely to be limitations when accessing routinely recorded data to measure the outcomes of RCTs. Quality assurance is unclear and the level of agreement of routinely recorded data with data recorded through standard RCT methods remains uncertain, particularly when measuring clinical outcomes. The time delay before routinely recorded data become available may have implications for RCTs where prompt reporting is both clinically important and a regulatory requirement. Furthermore the pre-application and application process may introduce further delays. This will have implications for RCTs relying on routinely recorded data. The cost-efficiency of accessing routinely recorded data, compared to standard methods, is unclear. Further research is required to assess the agreement, additional benefits and cost-efficiency of routinely recorded data compared to data collected through standard RCT methods; it may be in the additional benefits, such as addressing missing RCT data, where routinely recorded data is most useful.
Conclusions
The failure of access to routinely recorded data for a purpose, such as this study with clear secondary benefit to clinical research methodology, seems inappropriate when the ‘public purse’ funds the research, the researcher and the public body holding the data. Perhaps a significant cause or contributor to the current limitations is the Care.Data initiative in 2014. The proposal to extract primary-care records from all patients was opposed publicly by a number of groups and, for example, resulted in an internal inquiry within HSCIC. Data applications were suspended during this period and our current experience may be explained by the concurrent revision of the HSCIC application and approval procedures. However, in the medium term, of more concern is the harm in public perception that resulted. Currently, more than 1.2 million individuals in the UK have submitted a ‘Type 2 objection’, meaning that their data will not be shared for purposes other than direct care [19]. Although the application procedures may improve, and in time we may be able to access data more efficiently, the loss of 2.2% of the population’s data will have implications for the routinely recorded data that will then be made available for research. Involving patients as important stakeholders and re-gaining their trust will be an essential factor in realising the individual and population health care benefits of routinely recorded data [20].
Recommendations
We propose recommendations to improve access and implementation of routinely recorded data during a RCT, summarised in Table 5.
Abbreviations
- ADRN:
-
Administrative Data Research Network
- CPRD:
-
Clinical Practice Research Datalink
- DVLA:
-
Driver and Vehicle Licensing Agency
- DWP:
-
Department for Work and Pensions
- HMRC:
-
HM Revenue and Customs
- HSCIC:
-
The Health and Social Care Information Centre
- ISD:
-
Information Services Division
- NHS:
-
National Health Service
- NWEH:
-
North West eHealth
- NWIS:
-
NHS Wales Information Centre
- ONS:
-
The Office for National Statistics
- RCT:
-
Randomised controlled trial
- SAIL:
-
The Secure Anonymised Information Linkage Databank
- SANAD II:
-
The Standard and New Antiepileptic Drugs II trial
- THIN:
-
The Health Improvement Network
References
UK Government. The Data Protection Act. 2016. Available from: https://www.gov.uk/data-protection/the-data-protection-act. Accessed Mar 2016.
UK Government. The Freedom of Information Act. 2016. Available from: http://www.legislation.gov.uk/ukpga/2000/36/contents. Accessed 4 Mar 2016.
Lewsey JD, et al. Using routine data to complement and enhance the results of randomised controlled trials. Health Technol Assess. 2000;4(22):1–45. i-iv.
Raftery J, Roderick P, Stevens A. Potential use of routine databases in health technology assessment. Health Technol Assess (Winch Eng). 2005;9(20):1–92. iii-iv.
Clarson LE, et al. Increased risk of vascular disease associated with gout: a retrospective, matched cohort study in the UK Clinical Practice Research Datalink. Ann Rheum Dis. 2015;74(4):642–7.
McGregor J, et al. The Health Informatics Trial Enhancement Project (HITE): using routinely collected primary care data to identify potential participants for a depression trial. Trials. 2010;11:39.
Williams JG, et al. Can randomised trials rely on existing electronic data? A feasibility study to explore the value of routine data in health technology assessment. Health Technol Assess (Winch Eng). 2003;7(26):1–117. iii, v-x.
Gulliford MC, et al. Cluster randomized trials utilizing primary care electronic health records: methodological issues in design, conduct, and analysis (eCRT Study). Trials. 2014;15(1):220.
UK Government. The Plan for Growth. 2011. Available from: http://www.gov.uk/government./publications/plan-for-growth. Accessed 3 Mar 2016.
UK Government. The NHS Constitution for England. 2013. Available from: http://www.gov.uk/government/publications/the-nhs-constitution-for-england. Accessed 3 Mar 2016.
ADRN. The Administrative Data Research Network. 2016. Available from: http://adrn.ac.uk. Accessed 16 Jun 2016.
SAIL. The Secure Anonymised Information Linkage Databank. 2016. Available from: http://www.saildatabank.com. Accessed 1 Jun 2016.
Sayers A, et al. Evidence for a persistent, major excess in all cause admissions to hospital in children with type-1 diabetes: results from a large Welsh national matched community cohort study. BMJ Open. 2015;5(4):e005644.
New JP, et al. Obtaining real-world evidence: The Salford Lung Study. Thorax. 2014;69(12):1152–4.
(US) National Research Council. The prevention and treatment of missing data in clinical trials. National Academies Press, Washington DC (US); 2010.
HSCIC. General Practice Extraction Service. 2016. Available from: http://www.hscic.gov.uk/gpes. Accessed 5 Apr 2016.
Clinical-Study-Data-Request. Clinical Study Data Request. 2016. Available from: https://www.clinicalstudydatarequest.com. Accessed 5 Apr 2016.
Nelson EC, Dixon-Woods M, Batalden PB, Homa K, Van Citters AD, Morgan TS, Eftimovska E, Fisher ES, Ovretveit J, Harrison W, Lind C, Lindblad S. Patient focused registries can improve health, care and science. BMJ. 2016;354:i3319.
HSCIC. Press release: patient opt out. 2016. Available from: http://www.hscic.gov.uk/catalogue/PUB20527. Accessed 1 May 2016.
Van Staa TP, Goldacre B, Buchan I, Smeeth L. Big health data: the need to earn public trust after past management. BMJ. 2016;354:95–7.
HSCIC. The Health and Social Care Information Centre. 2016. Available from: http://www.hscic.gov.uk. Accessed 16 Jul 2016.
Bouras G, et al. Linked hospital and primary care database analysis of the incidence and impact of psychiatric morbidity following gastrointestinal cancer surgery in England. Ann Surg. 2016;264(1):93–9.
Turner EL, et al. Design and preliminary recruitment results of the Cluster randomised triAl of PSA testing for Prostate cancer (CAP). Br J Cancer. 2014;110(12):2829–36.
NWIS. NHS Wales Informatics Service. 2016. Available from: http://www.wales.nhs.uk/nwis/page/52490. Accessed 4 May 2016.
Ismail SI, Puyk B. The rise of obstetric anal sphincter injuries (OASIS): 11-year trend analysis using Patient Episode Database for Wales (PEDW) data. J Obstet Gynaecol. 2014;34(6):495–8.
ISD Scotland. The Information Services Division. 2016. Available from: http://www.isdscotland.org/Products-and-Services/index.asp. Accessed 5 May 2016.
Ahmed A, et al. Upper gastrointestinal bleeding in Scotland 2000–2010: improved outcomes but a significant weekend effect. World J Gastroenterol. 2015;21(38):10890–7.
CPRD. The Clinical Practice Research Datalink. 2016. Available from: http://www.cprd.com/intro.asp. Accessed 4 May 2016.
TPP. ResearchOne. 2016. Available from: http://www.tpp-uk.com/products/systmone. Accessed 4 May 2016.
Herrett E, et al. Text messaging reminders for influenza vaccine in primary care: protocol for a cluster randomised controlled trial (TXT4FLUJAB). BMJ Open. 2014;4(5):e004633.
QResearch. QResearch. 2016. Available from: http://www.qresearch.org/SitePages/Home.aspx. Accessed 6 May 2016.
Hill T, et al. Antidepressant use and risk of epilepsy and seizures in people aged 20 to 64 years: cohort study using a primary care database. BMC Psychiatry. 2015;15:315.
THIN. The Health Improvement Network. 2016. Available from: http://www.thin-uk.net. Accessed 5 Apr 2016.
González-Pérez A, et al. Incidence and predictors of hemorrhagic stroke in users of low-dose acetylsalicylic acid. J Stroke Cerebrovasc Dis. 2015;24(10):2321–8.
NWEH. North West eHealth. 2016. Available from: http://www.nweh.org.uk. Accessed 5 May 2016.
ApolloMedical. Apollo Data Extraction. 2016. Available from: http://www.apollo-medical.com/. Accessed 6 Apr 2016.
GraphnetHealth. Graphnet. 2016. Available from: http://www.graphnethealth.com/what-we-do/overview/what-we-do. Accessed 6 Apr 2016.
ONS. The Office for National Statistics. 2016. Available from: https://www.ons.gov.uk. Accessed 4 Apr 2016.
ONS. Official Labour Market Statistics. 2016. Available from: http://www.nomisweb.co.uk. Accessed 4 Apr 2016.
OCSI. Data 4 Neighbourhoods and Regeneration. 2016. Available from: http://www.data4nr.net/introduction. Accessed 7 Apr 2016.
UK Government. HM Revenue and Customs. 2016. Available from: https://www.gov.uk/government/organisations/hm-revenue-customs. Accessed 4 Apr 2016.
UK Government. The Department for Work and Pensions. 2016. Available from: https://www.gov.uk/government/organisations/department-for-work-pensions. Accessed 1 May 2016.
UK Government. Driver and Vehicle Licensing Agency. 2016. Available from: https://www.gov.uk/government/organisations/driver-and-vehicle-licensing-agency. Accessed 2 May 2016.
Clarke M. Standardising outcomes for clinical trials and systematic reviews. Trials. 2007;8:39.
Acknowledgements
Not Applicable
Funding
This report is independent research arising from a Clinical Training Fellowship (GA Powell) awarded by the Medical Research Council Hubs for Trials Methodology Research (Reference: P16-2014-GPC). LJ Bonnett is funded by a Post-Doctoral Fellowship (PDF-2015-08-044) from the National Institute for Health Research and AG Marson is part funded by the National Institute for Health Research Collaboration for Leadership in Applied Health Research and Care North West Coast. The views expressed are those of the authors and not necessarily those of the NHS, the Medical Research Council, the National Institute for Health Research or the routine data sources detailed in the report.
Availability of data and materials
There is no dataset available for this study and, therefore, data sharing is not applicable.
Author information
Authors and Affiliations
Contributions
GAP performed the background research, prepared the protocol, liaised with the routine data sources, prepared and submitted the routine data applications and drafted and redrafted the manuscript. LJB, CTS, DAH, PRW and AGM provided input into the development of the protocol and drafted and redrafted the manuscript. AGM is the guarantor for the report. The University of Liverpool was the sponsor for the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
To request identifiable data from routine data sources, written participant consent was required. The North of Scotland Research Ethics Service (16/NS/0007) and Health Research Authority (IRAS 189002) approved the study.
Consent for publication
Not applicable
Competing interests
All authors declare that (1) GAP, LJB, CTS, DAH, PRW and AGM do not have support from any company for the submitted work, (2) GP, LJB, CTS, DAH, PRW and AGM have no financial relationship with any company that might have an interest in the submitted work in the previous 3 years and (3) GP, LJB, CTS, DAH, PRW and AGM have no other relationships or activities that could appear to have influenced the submitted work.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Powell, G.A., Bonnett, L.J., Tudur-Smith, C. et al. Using routinely recorded data in the UK to assess outcomes in a randomised controlled trial: The Trials of Access. Trials 18, 389 (2017). https://doi.org/10.1186/s13063-017-2135-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13063-017-2135-9